Abstract
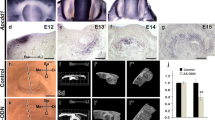
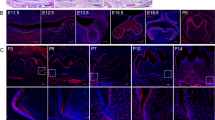
Glomerular epithelial protein-1 (Glepp1), a R3 subtype family of receptor-type protein tyrosine phosphatases, plays important role in the activation of Src family kinases and regulates cellular processes such as cell proliferation, differentiation, and apoptosis. In this study, we firstly examined the functional evaluation of Glepp1 in tooth development and morphogenesis. The precise expression level and developmental function of Glepp1 were examined by RT-qPCR, in situ hybridization, and loss and gain of functional study using a range of in vitro organ cultivation methods. Expression of Glepp1 was detected in the developing tooth germs in cap and bell stage of tooth development. Knocking down Glepp1 at E13 for 2 days showed the altered expression levels of tooth development-related signaling molecules, including Bmps, Dspp, Fgf4, Lef1, and Shh. Moreover, transient knock down of Glepp1 revealed alterations in cellular physiology, examined by the localization patterns of Ki67 and E-cadherin. Similarly, knocking down of Glepp1 showed disrupted enamel rod and interrod formation in 3-week renal transplanted teeth. In addition, due to attrition of odontoblastic layers, the expression signals of Dspp and the localization of NESTIN were almost not detected after knock down of Glepp1; however, their expressions were increased after Glepp1 overexpression. Thus, our results suggested that Glepp1 plays modulating roles during odontogenesis by regulating the expression levels of signaling molecules and cellular events to achieve the proper structural formation of hard tissue matrices in mice molar development.





Similar content being viewed by others
Data availability
All relevant data are included in the manuscript. Raw data can be made available by the author upon reasonable request.
References
Åberg T, Wozney J, Thesleff I (1997) Expression patterns of bone morphogenetic proteins (Bmps) in the developing mouse tooth suggest roles in morphogenesis and cell differentiation. Dev Dyn 210:383–396. https://doi.org/10.1002/(SICI)1097-0177(199712)210:4%3c383::AID-AJA3%3e3.0.CO;2-C
Abramyan J, Geetha-Loganathan P, Šulcová M, Buchtová M (2021) Role of cell death in cellular processes during odontogenesis. Front Cell Dev Biol 9. https://doi.org/10.3389/fcell.2021.671475
Adhikari N, Neupane S, Gwon G-J et al (2017) Grhl3 modulates epithelial structure formation of the circumvallate papilla during mouse development. Histochem Cell Biol 147:5–16. https://doi.org/10.1007/s00418-016-1487-7
Aryal YP, Neupane S, Kim T-Y et al (2020) Developmental roles of FUSE binding protein 1 (Fubp1) in tooth morphogenesis. Int J Mol Sci 21:8079. https://doi.org/10.3390/ijms21218079
Asbagh LA, Vazquez I, Vecchione L et al (2014) The tyrosine phosphatase PTPRO sensitizes colon cancer cells to anti-EGFR therapy through activation of SRC-mediated EGFR signaling. Oncotarget 5:10070–10083. https://doi.org/10.18632/oncotarget.2458
Avraham S, London R, Tulloch GA et al (1997) Characterization and chromosomal localization of PTPRO, a novel receptor protein tyrosine phosphatase, expressed in hematopoietic stem cells. Gene 204:5–16. https://doi.org/10.1016/S0378-1119(97)00420-4
Balic A, Thesleff I (2015) Tissue interactions regulating tooth development and renewal. Curr Top Dev Biol 115:157–186. https://doi.org/10.1016/BS.CTDB.2015.07.006
Biggs LC, Mikkola ML (2014) Early inductive events in ectodermal appendage morphogenesis. Semin Cell Dev Biol 25–26:11–21. https://doi.org/10.1016/j.semcdb.2014.01.007
Cai J, Cho S-W, Kim J-Y et al (2007) Patterning the size and number of tooth and its cusps. Dev Biol 304:499–507. https://doi.org/10.1016/j.ydbio.2007.01.002
Cho S-W, Lee H-A, Cai J et al (2007) The primary enamel knot determines the position of the first buccal cusp in developing mice molars. Differentiation 75:441–451. https://doi.org/10.1111/J.1432-0436.2006.00153.X
Dassule HR, Lewis P, Bei M et al (2000) Sonic hedgehog regulates growth and morphogenesis of the tooth. Development 127:4775–4785. https://doi.org/10.1186/1471-2199-12-38
Dressler GR (2006) The cellular basis of kidney development. Annu Rev Cell Dev Biol 22:509–529. https://doi.org/10.1146/annurev.cellbio.22.010305.104340
Jernvall J, Kettunen P, Karavanova I et al (1994) Evidence for the role of the enamel knot as a control center in mammalian tooth cusp formation: non-dividing cells express growth stimulating Fgf-4 gene. Int J Dev Biol 38:463–469
Jiang R, Xia Y, Li J et al (2010) High expression levels of IKKα and IKKβ are necessary for the malignant properties of liver cancer. Int J Cancer 126:1263–1274. https://doi.org/10.1002/ijc.24854
Jung JK, Gwon GJ, Neupane S et al (2017) Bortezomib facilitates reparative dentin formation after pulp access cavity preparation in mouse molar. J Endod 43:2041–2047. https://doi.org/10.1016/j.joen.2017.07.018
Kettunen P, Thesleff I (1998) Expression and function of FGFs-4, -8, and -9 suggest functional redundancy and repetitive use as epithelial signals during tooth morphogenesis. Dev Dyn 211:256–268. https://doi.org/10.1002/(SICI)1097-0177(199803)211:3%3c256::AID-AJA7%3e3.0.CO;2-G
Kim M, Kim H, Jho E, hoon, (2010) Identification of ptpro as a novel target gene of Wnt signaling and its potential role as a receptor for Wnt. FEBS Lett 584:3923–3928. https://doi.org/10.1016/j.febslet.2010.08.034
Kim J, Ahn Y, Adasooriya D et al (2019) Shh plays an inhibitory role in cusp patterning by regulation of Sostdc1. J Dent Res 98:98–106. https://doi.org/10.1177/0022034518803095
Kratochwil K, Galceran J, Tontsch S et al (2002) FGF4, a direct target of LEF1 and Wnt signaling, can rescue the arrest of tooth organogenesis in Lef1-/-mice. Genes Dev 16:3173–3185. https://doi.org/10.1101/gad.1035602
Lesot H, Brook AH (2009) Epithelial histogenesis during tooth development. Arch Oral Biol 54:S25–S33. https://doi.org/10.1016/j.archoralbio.2008.05.019
Liao WH, Cheng CH, Hung KS et al (2013) Protein tyrosine phosphatase receptor type O (Ptpro) regulates cerebellar formation during zebrafish development through modulating Fgf signaling. Cell Mol Life Sci 70:2367–2381. https://doi.org/10.1007/s00018-013-1259-7
Liu Z, Hou J, Ren L et al (2015) Protein tyrosine phosphatase receptor type O expression in the tumor niche correlates with reduced tumor growth, angiogenesis, circulating tumor cells and metastasis of breast cancer. Oncol Rep 33:1908–1914. https://doi.org/10.3892/or.2015.3772
MacKenzie A, Ferguson MW, Sharpe PT (1992) Expression patterns of the homeobox gene, Hox-8, in the mouse embryo suggest a role in specifying tooth initiation and shape. Development 115:403–420
Neupane S, Sohn WJ, Rijal G et al (2014) Developmental regulations of Perp in mice molar morphogenesis. Cell Tissue Res 358:109–121. https://doi.org/10.1007/s00441-014-1908-7
Neupane S, Sohn W-J, Gwon G-J et al (2015) The role of APCDD1 in epithelial rearrangement in tooth morphogenesis. Histochem Cell Biol 144:377–387. https://doi.org/10.1007/s00418-015-1345-z
Pispa J, Thesleff I (2003) Mechanisms of ectodermal organogenesis. Dev Biol 262:195–205. https://doi.org/10.1016/S0012-1606(03)00325-7
Sagai T, Amano T, Maeno A et al (2017) SHH signaling directed by two oral epithelium-specific enhancers controls tooth and oral development. Sci Rep 7:13004. https://doi.org/10.1038/s41598-017-12532-y
Seo H, Amano T, Seki R et al (2018) Upstream enhancer elements of Shh regulate oral and dental patterning. J Dent Res 97:1055–1063. https://doi.org/10.1177/0022034518758642
Thesleff I, Jernvall J (1997) The enamel knot: a putative signaling center regulating tooth development. Cold Spring Harb Symp Quant Biol 62:257–267. https://doi.org/10.1101/SQB.1997.062.01.032
Thesleff I, Keranen S, Jernvall J (2001a) Linking tooth morphogenesis and odontoblast differentiation
Thesleff I, Keränen S, Jernvall J (2001b) Enamel knots as signaling centers linking tooth morphogenesis and odontoblast differentiation. Adv Dent Res 15:14–18. https://doi.org/10.1177/08959374010150010401
Thomas PE, Wharram BL, Goyal M et al (1994) GLEPP1, a renal glomerular epithelial cell (podocyte) membrane protein-tyrosine phosphatase. Identification, molecular cloning, and characterization in rabbit. J Biol Chem 269:19953–19961. https://doi.org/10.1016/S0021-9258(17)32113-0
Tucker AS, Headon DJ, Courtney JM et al (2004) The activation level of the TNF family receptor, Edar, determines cusp number and tooth number during tooth development. Dev Biol 268:185–194. https://doi.org/10.1016/j.ydbio.2003.12.019
Vaahtokari A, Åberg T, Jernvall J et al (1996) The enamel knot as a signaling center in the developing mouse tooth. Mech Dev 54:39–43. https://doi.org/10.1016/0925-4773(95)00459-9
Wharram BL, Goyal M, Gillespie PJ et al (2000) Altered podocyte structure in GLEPP1 (Ptpro)-deficient mice associated with hypertension and low glomerular filtration rate. J Clin Invest 106:1281–1290. https://doi.org/10.1172/JCI7236
Yu M, Lin G, Arshadi N et al (2012) Expression profiling during mammary epithelial cell three-dimensional morphogenesis identifies PTPRO as a novel regulator of morphogenesis and ErbB2-mediated transformation. Mol Cell Biol 32:3913–3924. https://doi.org/10.1128/MCB.00068-12
Yuasa T, Kataoka H, Kinto N et al (2002) Sonic hedgehog is involved in osteoblast differentiation by cooperating with BMP-2. J Cell Physiol 193:225–232. https://doi.org/10.1002/jcp.10166
Funding
This work was supported by National Research Foundation of Korea (NRF) grant, funded by Korea government (NRF-2017R1A5A2015391, NRF-2018R1A2A3075600, NRF-2020R1A6A1A06046235, NRF-2022R1I1A2063745).
Author information
Authors and Affiliations
Contributions
Study concept and design; data acquisition, analysis, and interpretation; and drafting and critically revision of the manuscript: S.N., Y.P.A., S.J.C., and J.Y.K. Data interpretation, review, and editing: H.J.K., S.G.L., T.Y.K., E.P., J.Y.K., J.H.K., W.J.S., S.Y.A., C.H.A., J.K.J., J.H.H., H.Y., S.W.C., S.L., Y.L., K.K.P., B.K.M., C.P., and T.Y.K.
Corresponding authors
Ethics declarations
Ethical approval
All experiments involving animals were performed in accordance with the guidelines of the Kyungpook National University, School of Dentistry, Intramural Animal Use and Care Committee (KNU-2020–0107).
Informed consent
All authors gave their final approval and agree to be accountable for all aspects of the work.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Neupane, S., Aryal, Y.P., Kwak, HJ. et al. Developmental roles of glomerular epithelial protein-1 in mice molar morphogenesis. Cell Tissue Res 395, 53–62 (2024). https://doi.org/10.1007/s00441-023-03841-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-023-03841-y