Abstract
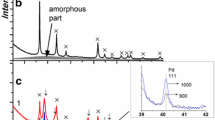
The powders of Rh and CeO2 are synthesized by pulsed laser ablation in liquid. The Rh–CeO2 model catalysts are prepared by the calcination of these powders in a wide temperature range from 450 °C to 1000 °C. The formation of individual and mixed (rhodium- and cerium-containing) phases with increasing temperature of catalyst calcination is studied by powder XRD and Raman spectroscopy. The redox properties of prepared catalysts are tested in a temperature-programmed reaction of CO reduction; their catalytic properties are studied on the example of CO oxidation. It is shown that the catalysts remain stable during catalytic tests due to the formation of a nano-heterophase system consisting of rhodium oxide (Rh2O3) and cerium oxide (CeO2) nanoparticles. The discovered high stability is most likely explained by the formation of the Rh3+–CeO2 species with the localization of Rh3+ ions in subsurface CeO2 layers due to the contacts between rhodium oxide and cerium oxide nanoparticles. Introducing Rh3+ ions into Ce4+ positions of the CeO2 lattice distorts the cerium oxide structure and leads to the formation of active oxygen species interacting with CO at low temperatures. The catalysts are shown to preserve high activity in the reaction of low-temperature CO oxidation even after the calcination at 1000 °C.









Similar content being viewed by others
REFERENCES
M. Shelef and G. W. Graham. Why rhodium in automotive three-way catalysts? Catal. Rev. Sci. Eng., 1994, 36, 433-457. https://doi.org/10.1080/01614949408009468
W. Alsalahi and A. M. Trzeciak. Rhodium-catalyzed hydroformylation under green conditions: Aqueous/organic biphasic, “on water”, solventless and Rh nanoparticle based systems. Coord. Chem. Rev., 2021, 430, 213732. https://doi.org/10.1016/j.ccr.2020.213732
K. Shimura and T. Fujitani. Effects of rhodium catalyst support and particle size on dry reforming of methane at moderate temperatures. Mol. Catal., 2021, 509, 111623. https://doi.org/10.1016/j.mcat.2021.111623
M. Alisha, R. M. Philip, and G. Anilkumar. An overview of rhodium-catalysed heck type reactions. J. Organomet. Chem., 2022, 959, 122207. https://doi.org/10.1016/j.jorganchem.2021.122207
L. Zhu, C. Li, Q. Yun, S. Han, Y. Lv, Q. Lu, and J. Chen. Recent advances of Rh-based intermetallic nanomaterials for catalytic applications. Chin. Chem. Lett., 2023, 108515. https://doi.org/10.1016/j.cclet.2023.108515
M. V. Twigg. Progress and future challenges in controlling automotive exhaust gas emissions. Appl. Catal., B, 2007, 70, 2-15. https://doi.org/10.1016/j.apcatb.2006.02.029
R. J. Farrauto, M. Deeba, and S. Alerasool. Gasoline automobile catalysis and its historical journey to cleaner air. Nat. Catal., 2019, 2, 603-613. https://doi.org/10.1038/s41929-019-0312-9
M. Zammit, C. Dimaggio, C. Kim, C. Lambert, G. Muntean, C. Peden, J. Parks, and K. Howden. Future automotive aftertreatment solutions: the challenge workshop report (No. PNNL-22815). Richland, Washington, USA: Pacific Northwest National Lab.(PNNL), 2013. https://doi.org/10.2172/1097340
C. K. Lambert. Current state of the art and future needs for automotive exhaust catalysis. Nat. Catal., 2019, 2, 554-557. https://doi.org/10.1038/s41929-019-0303-x
T. Montini, M. Melchionna, M. Monai, and P. Fornasiero. Fundamentals and catalytic applications of CeO2-based materials. Chem. Rev., 2016, 116, 5987-6041. https://doi.org/10.1021/acs.chemrev.5b00603
M. Kurnatowska and L. Kepinski. Structure and thermal stability of nanocrystalline Ce1–xRhxO2–y in reducing and oxidizing atmosphere. Mater. Res. Bull., 2013, 48, 852-862. https://doi.org/10.1016/j.materresbull.2012.11.076
E. A. Derevyannikova, T. Y. Kardash, L. S. Kibis, E. M. Slavinskaya, V. A. Svetlichnyi, O. A. Stonkus, A. S. Ivanova, and A. I. Boronin. The structure and catalytic properties of Rh-doped CeO2 catalysts. Phys. Chem. Chem. Phys., 2017, 19, 31883-31897. https://doi.org/10.1039/C7CP06573F
K. A. Ledwa, L. Kępiński, and M. Pawlyta. Thermal stability and propane combustion activity of RhxCe1–xO2–y nanoparticles deposited on functionalized alumina. Catal. Sci. Technol., 2019, 9, 4633-4644. https://doi.org/10.1039/C9CY01316D
E. A. Fedorova, T. Y. Kardash, L. S. Kibis, O. A. Stonkus, E. M. Slavinskaya, V. A. Svetlichnyi, S. Pollastri, and A. I. Boronin. Unraveling the low-temperature activity of Rh–CeO2 catalysts in CO oxidation: Probing the local structure and redox transformation of Rh3+ species. Phys. Chem. Chem. Phys., 2023, 25, 2862-2874. https://doi.org/10.1039/D2CP04503F
S. Imamura, T. Yamashita, R. Hamada, Y. Saito, Y. Nakao, N. Tsuda, and C. Kaito, Strong interaction between rhodium and ceria. J. Mol. Catal. A: Chem., 1998, 129, 249-256. https://doi.org/10.1016/S1381-1169(97)00183-0
A. Gayen, K. R. Priolkar, P. R. Sarode, V. Jayaram, M. S. Hegde, G. N. Subbanna, and S. Emura. Ce1–xRhxO2–δ solid solution formation in combustion-synthesized Rh/CeO2 catalyst studied by XRD, TEM, XPS, and EXAFS. Chem. Mater., 2004, 16, 2317-2328. https://doi.org/10.1021/cm040126l
L. S. Kibis, T. Y. Kardash, E. A. Derevyannikova, O. A. Stonkus, E. M. Slavinskaya, V. A. Svetlichnyi, and A. I. Boronin. Redox and catalytic properties of RhxCe1–xO2–δ solid solution. J. Phys. Chem. C, 2017, 121, 26925-26938. https://doi.org/10.1021/acs.jpcc.7b09983
H. Jeong, G. Lee, B. Kim, J. Bae, J. W. Han, and H. Lee. Fully dispersed Rh ensemble catalyst to enhance low-temperature activity. J. Am. Chem. Soc., 2018, 140, 9558-9565. https://doi.org/10.1021/jacs.8b04613
L. S. Kibis, D. A. Svintsitskiy, E. A. Derevyannikova, T. Yu. Kardash, E. M. Slavinskaya, O. A. Stonkus, V. A. Svetlichnyi, and A. I. Boronin. From highly dispersed Rh3+ to nanoclusters and nanoparticles: Probing the low-temperature NO + CO activity of Rh-doped CeO2 catalysts. Appl. Surf. Sci., 2019, 493, 1055-1066. https://doi.org/10.1016/j.apsusc.2019.07.043
K. Khivantsev, C. G. Vargas, J. Tian, L. Kovarik, N. R. Jaegers, J. Szanyi, and Y. Wang. Economizing on precious metals in three-way catalysts: Thermally stable and highly active single-atom rhodium on ceria for NO abatement under dry and industrially relevant conditions. Angew. Chem., Int. Ed., 2021, 60, 391-398. https://doi.org/10.1002/anie.202010815
K. Sevcikova, V. Nehasil, M. Vorokhta, S. Haviar, V. Matolín, I. Matolínová, K. Mašek, I. Píš, K. Kobayashi, M. Kobata, T. Nagata, Y. Matsushita, and H. Yoshikawa. Altering properties of cerium oxide thin films by Rh doping. Mater. Res. Bull., 2015, 67, 5-13. https://doi.org/10.1016/j.materresbull.2015.02.059
H. Yoshida, K. Koizumi, M. Boero, M. Ehara, S. Misumi, A. Matsumoto, Y. Kuzuhara, T. Sato, J. Ohyama, and M. Machida. High turnover frequency CO–NO reactions over Rh overlayer catalysts: A comparative study using Rh nanoparticles. J. Phys. Chem. C, 2019, 123, 6080-6089. https://doi.org/10.1021/acs.jpcc.9b00383
E. D. Fakhrutdinova, A. V. Shabalina, M. A. Gerasimova, A. L. Nemoykina, O. V. Vodyankina, and V. A. Svetlichnyi. Highly defective dark nano titanium dioxide: Preparation via pulsed laser ablation and application. Materials, 2020, 13, 2054. https://doi.org/10.3390/ma13092054
A. Wazeer, A. Das, A. Sinha, and A. Karmakar. Nanomaterials synthesis via laser ablation in liquid: A review. J. Inst. Eng. (India): Ser. D, 2022, 104, 413-426. https://doi.org/10.1007/s40033-022-00370-w
E. M. Slavinskaya, T. Y. Kardash, O. A. Stonkus, R. V. Gulyaev, I. N. Lapin, V. A. Svetlichnyi, and A. I. Boronin. Metal-support interaction in Pd/CeO2 model catalysts for CO oxidation: From pulsed laser-ablated nanoparticles to highly active state of the catalyst. Catal. Sci. Technol., 2016, 6, 6650-6666. https://doi.org/10.1039/c6cy00319b
E. M. Slavinskaya, A. I. Stadnichenko, V. V. Muravyov, T. Y. Kardash, E. A. Derevyannikova, V. I. Zaikovskii, O. A. Stonkus, I. N. Lapin, V. A. Svetlichnyi, and A. I. Boronin. Transformation of a Pt–CeO2 mechanical mixture of pulsed-laser-ablated nanoparticles to a highly active catalyst for carbon monoxide oxidation. ChemCatChem, 2018, 10, 2232-2247. https://doi.org/10.1002/cctc.201702050
A. I. Stadnichenko, E. M. Slavinskaya, E. A. Fedorova, D. A. Goncharova, V. I. Zaikovskii, T. Y. Kardash, V. A. Svetlichnyi, and A. I. Boronin. Activation of Au–CeO2 composites prepared by pulsed laser ablation in the reaction of low-temperature CO oxidation. J. Struct. Chem., 2021, 62, 1918-1934. https://doi.org/10.1134/S0022476621120118
E. M. Slavinskaya, A. V. Zadesenets, O. A. Stonkus, A. I. Stadnichenko, A. V. Shchukarev, Y. V. Shubin, S. V. Korenev, and A. I. Boronin. Thermal activation of Pd/CeO2–SnO2 catalysts for low-temperature CO oxidation. Appl. Catal., B, 2020, 277, 119275. https://doi.org/10.1016/j.apcatb.2020.119275
V. A. Svetlichny and I. N. Lapin. Poluchenie nanochastits CeO2 metodom lazernoi ablyatsii ob′emnykh mishenei metallicheskogo tseriya v zhidkosti (Production of CeO2 nanoparticles by laser ablation of volumetric targets of metallic cerium in liquid). Izv. Vyssh. Uchebn. Zaved., Fiz., 2015, 55, 106-112.
M. Romeo, K. Bak, J. El Fallah, F. Le Normand, and L. Hilaire. XPS study of the reduction of cerium dioxide. Surf. Interface Anal., 1993, 20, 508-512. https://doi.org/10.1002/sia.740200604
J. W. M. Biesterbos and J. Hornstra. The crystal structure of the high-temperature, low-pressure form of Rh2O3. J. Less Common Met., 1973, 30, 121-125. https://doi.org/10.1016/0022-5088(73)90013-1
A. Wold, R. J. Arnott, and W. J. Croft. The reaction of rare earth oxides with a high temperature form of rhodium(III) oxide. Inorg. Chem., 1963, 2, 972-974. https://doi.org/10.1021/ic50009a023.
S. Hosokawa, M. Taniguchi, K. Utani, H. Kanai, and S. Imamura. Affinity order among noble metals and CeO2. Appl. Catal., A, 2005, 289, 115-120. https://doi.org/10.1016/j.apcata.2005.04.048
S. Askrabic, Z. Dohcevic-Mitrovic, A. Kremenovic, N. Lazarevic, V. Kahlenberg, and Z. V Popovic. Oxygen vacancy-induced microstructural changes of annealed CeO2–x nanocrystals. J. Raman Spectrosc., 2012, 43, 76-81. https://doi.org/10.1002/jrs.2987
T. Taniguchi, T. Watanabe, N. Sugiyama, A. K. Subramani, H. Wagata, N. Matsushita, and M. Yoshimura. Identifying defects in ceria-based nanocrystals by UV resonance Raman spectroscopy. J. Phys. Chem. C, 2009, 113, 19789-19793. https://doi.org/10.1021/jp9049457
L. Li, F. Chen, J.-Q. Lu, and M.-F. Luo. Study of defect sites in Ce1–xMxO2–δ (x = 0.2) solid solutions using Raman spectroscopy. J. Phys. Chem. A, 2011, 115, 7972-7977. https://doi.org/10.1021/jp203921m
S. Musić, A. Šarić, S. Popović, and M. Ivanda. Formation and characterisation of nanosize α-Rh2O3 particles. J. Mol. Struct., 2009, 924-926, 221-224. https://doi.org/10.1016/j.molstruc.2008.10.017
W. H. Weber, R. J. Baird, and G. W. Graham. Raman investigation of palladium oxide, rhodium sesquioxide and palladium rhodium dioxide. J. Raman Spectrosc., 1988, 19, 239-244. https://doi.org/10.1002/jrs.1250190404
O. A. Stonkus, A. V. Zadesenets, E. M. Slavinskaya, A. I. Stadnichenko, V. A. Svetlichnyi, Y. V. Shubin, S. V. Korenev, and A. I. Boronin. Pd/CeO2–SnO2 catalysts with varying tin content: Promotion of catalytic properties and structure modification. Catal. Commun., 2022, 172, 106554. https://doi.org/10.1016/j.catcom.2022.106554
L. Marot, D. Mathys, G. De Temmerman, and P. Oelhafen. Characterization of sub-stoichiometric rhodium oxide deposited by magnetron sputtering. Surf. Sci., 2008, 602, 3375-3380. https://doi.org/10.1016/j.susc.2008.09.012
A. A. Tolia, R. J. Smiley, W. N. Delgass, C. G. Takoudis, and M. J. Weaver. Surface oxidation of rhodium at ambient pressures as probed by surface-enhanced Raman and X-ray photoelectron spectroscopies. J. Catal., 1994, 150, 56-70. https://doi.org/10.1006/jcat.1994.1322
L. S. Kibis, A. I. Stadnichenko, S. V. Koscheev, V. I. Zaikovskii, and A. I. Boronin. XPS study of nanostructured rhodium oxide film comprising Rh4+ species. J. Phys. Chem. C, 2016, 120, 19142-19150. https://doi.org/10.1021/acs.jpcc.6b05219
M. Kurnatowska, M. E. Schuster, W. Mista, and L. Kepinski. Self-regenerative property of nanocrystalline Ce0.89M0.11O2–y (M = Pd, Rh) mixed oxides. ChemCatChem, 2014, 6, 3125-3131. https://doi.org/10.1002/cctc.201402480
Funding
This work was funded by the Ministry of Science and Higher Education of the Russian Federation within the State Assignment for BIC SB RAS (project No. AAAA-A21-121011390053-4). The TEM study was conducted using the equipment of the Center of Collective Use “National Center of Catalyst Research”.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interests.
Additional information
Russian Text © The Author(s), 2023, published in Zhurnal Strukturnoi Khimii, 2023, Vol. 64, No. 11, 118600.https://doi.org/10.26902/JSC_id118600
Supplementary material
Rights and permissions
About this article
Cite this article
Kibis, L.S., Krotova, A.I., Fedorova, E.A. et al. Preparation of Model Rh–CeO2 Catalysts by Pulsed Laser Ablation in Liquid. J Struct Chem 64, 2187–2199 (2023). https://doi.org/10.1134/S0022476623110161
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476623110161