Abstract
Purpose
PiwiL1 has been reported to be over-expressed in many cancers. However, the molecular mechanism by which these proteins contribute to tumorigenesis and their regulation in cancer cells is still unclear. We intend to understand the role of PiwiL1 in tumorigenesis and also its regulation in cervical cells.
Methods
We studied the effect of loss of PiwiL1 function on tumor properties of cervical cancer cells in vitro and in vivo. Also we have looked into the effect of PiwiL1 overexpression in the malignant transformation of normal cells both in vitro and in vivo. Further RNA-seq and RIP-seq analyses were done to get insight of the direct and indirect targets of PiwiL1 in the cervical cancer cells.
Results
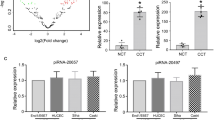
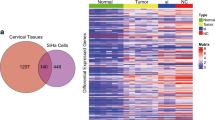
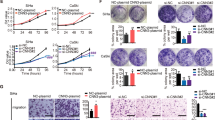
Here, we report that PiwiL1 is not only over-expressed, but also play a major role in tumor induction and progression. Abolition of PiwiL1 in CaSki cells led to a decrease in the tumor-associated properties, whereas, its upregulation conferred malignant transformation of normal HaCaT cells. Our study delineates a new link between HPV oncogenes, E6 and E7 with PiwiL1. p53 and E2F1 directly bind and differentially regulate PiwiL1 promoter in a context-dependant manner. Further, RNA-seq together with RIP-RNA-seq suggested a strong and direct role for PiwiL1 in promoting metastasis in cervical cancer cells.
Conclusion
Our study demonstrates that PiwiL1 act as an oncogene in cervical cancer by inducing tumor-associated properties and EMT pathway. The finding that HPV oncogenes, E6/E7 can positively regulate PiwiL1 suggests a possible mechanism behind HPV-mediated tumorigenesis in cervical cancer.








Similar content being viewed by others
Data availability
All data that support the findings of this study are available from the corresponding authors upon reasonable request.
References
R. Suzuki, S. Honda, Y. Kirino, Front. Genet. 3, 204 (2012). https://doi.org/10.3389/fgene.2012.00204
Y. Liu, M. Dou, X. Song, Y. Dong, S. Liu, H. Liu, J. Tao, W. Li, X. Yin, W. Xu, Mol. Cancer 18, 123 (2019). https://doi.org/10.1186/s12943-019-1052-9
Y.W. Iwasaki, S. Sriswasdi, Y. Kinugasa, J. Adachi, Y. Horikoshi, A. Shibuya, W. Iwasaki, S. Tashiro, T. Tomonaga, H. Siomi, EMBO J. 40, e108345 (2021). https://doi.org/10.15252/embj.2021108345
A.K. Sharma, M.C. Nelson, J.E. Brandt, M. Wessman, N. Mahmud, K.P. Weller, R. Hoffman, Blood 97, 426–434 (2001). https://doi.org/10.1182/blood.v97.2.426
W. Deng, H. Lin, Dev. Cell 2, 819–830 (2002). https://doi.org/10.1016/s1534-5807(02)00165-x
A. Grimson, M. Srivastava, B. Fahey, B.J. Woodcroft, H.R. Chiang, N. King, B.M. Degnan, D.S. Rokhsar, D.P. Bartel, Nature 455, 1193–1197 (2008). https://doi.org/10.1038/nature07415
Y. Mei, D. Clark, L. Mao, Cancer Lett. 336, 46–52 (2013). https://doi.org/10.1016/j.canlet.2013.04.008
T. Sasaki, A. Shiohama, S. Minoshima, N. Shimizu, Genomics 82, 323–330 (2003). https://doi.org/10.1016/s0888-7543(03)00129-0
J.H. Lee, D. Schutte, G. Wulf, L. Fuzesi, H.J. Radzun, S. Schweyer, W. Engel, K. Nayernia, Hum. Mol. Genet. 15, 201–211 (2006). https://doi.org/10.1093/hmg/ddi430
X. Liu, Y. Sun, J. Guo, H. Ma, J. Li, B. Dong, G. Jin, J. Zhang, J. Wu, L. Meng, C. Shou, Int. J. Cancer 118, 1922–1929 (2006). https://doi.org/10.1002/ijc.21575
G. He, L. Chen, Y. Ye, Y. Xiao, K. Hua, D. Jarjoura, T. Nakano, S.H. Barsky, R. Shen, J.X. Gao, Am. J. Transl. Res. 2, 156–169 (2010)
D. Li, X. Sun, D. Yan, J. Huang, Q. Luo, H. Tang, Z. Peng, Exp. Biol. Med. (Maywood) 237, 1231–1240 (2012). https://doi.org/10.1258/ebm.2012.011380
A. Janic, L. Mendizabal, S. Llamazares, D. Rossell, C. Gonzalez, Science 330, 1824–1827 (2010). https://doi.org/10.1126/science.1195481
D. Qiao, A.M. Zeeman, W. Deng, L.H. Looijenga, H. Lin, Oncogene 21, 3988–3999 (2002). https://doi.org/10.1038/sj.onc.1205505
G. Sun, Y. Wang, L. Sun, H. Luo, N. Liu, Z. Fu, Y. You, Brain Res. 1373, 183–188 (2011). https://doi.org/10.1016/j.brainres.2010.11.097
L.F. Grochola, T. Greither, H. Taubert, P. Moller, U. Knippschild, A. Udelnow, D. Henne-Bruns, P. Wurl, Br. J. Cancer 99, 1083–1088 (2008). https://doi.org/10.1038/sj.bjc.6604653
Y. Zeng, L.K. Qu, L. Meng, C.Y. Liu, B. Dong, X.F. Xing, J. Wu, C.C. Shou, Chin. Med. J. (Engl) 124, 2144–2149 (2011)
W. He, Z. Wang, Q. Wang, Q. Fan, C. Shou, J. Wang, K.E. Giercksky, J.M. Nesland, Z. Suo, BMC Cancer 9, 426 (2009). https://doi.org/10.1186/1471-2407-9-426
Y. Wang, Y. Liu, X. Shen, X. Zhang, X. Chen, C. Yang, H. Gao, Int. J. Clin. Exp. Pathol. 5, 315–325 (2012)
Y. Tan, L. Liu, M. Liao, C. Zhang, S. Hu, M. Zou, M. Gu, X. Li, Acta Biochim. Biophys. Sin. (Shanghai) 47, 315–324 (2015). https://doi.org/10.1093/abbs/gmv018
H. Jeong, K.H. Park, Y. Lee, A. Jeong, S. Choi, K.W. Kim, Processes 9, 1208 (2021)
M. Kunnummal, M. Angelin, A.V. Das, Hum. Cell 34, 1629–1641 (2021). https://doi.org/10.1007/s13577-021-00590-4
W. Liu, Q. Gao, K. Chen, X. Xue, M. Li, Q. Chen, G. Zhu, Y. Gao, Oncol. Rep. 32, 1853–1860 (2014). https://doi.org/10.3892/or.2014.3401
C. Li, X. Zhou, J. Chen, Y. Lu, Q. Sun, D. Tao, W. Hu, X. Zheng, S. Bian, Y. Liu, Y. Ma, Oncotarget 6, 27794–27804 (2015). https://doi.org/10.18632/oncotarget.4533
G. Pei, B. Li, A. Ma, Oncol. Lett. 16, 3874–3880 (2018). https://doi.org/10.3892/ol.2018.9056
D. Feng, C. Peng, C. Li, Y. Zhou, M. Li, B. Ling, H. Wei, Z. Tian, Oncol. Rep. 22, 1129–1134 (2009). https://doi.org/10.3892/or_00000545
D. Feng, K. Yan, Y. Zhou, H. Liang, J. Liang, W. Zhao, Z. Dong, B. Ling, Oncotarget 7, 64575–64588 (2016). https://doi.org/10.18632/oncotarget.11810
M. Arbyn, E. Weiderpass, L. Bruni, S. de Sanjose, M. Saraiya, J. Ferlay, F. Bray, Lancet Glob. Health 8, e191–e203 (2020). https://doi.org/10.1016/S2214-109X(19)30482-6
H.G. Ahmed, S.H. Bensumaidea, F.D. Alshammari, F.S.H. Alenazi, B.A. ALmutlaq, M.Z. Alturkstani, I.A. Aladani, Asian Pac. J. Cancer Prev. 18, 1543–1548 (2017). https://doi.org/10.22034/APJCP.2017.18.6.1543
W.K. Liu, X.Y. Jiang, Z.X. Zhang, Arch. Virol. 155, 657–663 (2010). https://doi.org/10.1007/s00705-010-0635-y
S. Shi, Z.Z. Yang, S. Liu, F. Yang, H. Lin, Proc. Natl. Acad. Sci. U. S. A. 117, 22390–22401 (2020). https://doi.org/10.1073/pnas.2008724117
S. Sreekanth, V.A. Rasheed, L. Soundararajan, J. Antony, M. Saikia, K.C. Sivakumar, A.V. Das, Mol. Neurobiol. 54, 8033–8049 (2017). https://doi.org/10.1007/s12035-016-0237-0
K.J. Livak, T.D. Schmittgen, Methods 25, 402–408 (2001). https://doi.org/10.1006/meth.2001.1262
J. Issac, P.S. Raveendran, M. Kunnummal, M. Angelin, S. Ravindran, B. Basu, A.V. Das, Biochim. Biophys. Acta Mol. Cell Res. 1870, 119510 (2023). https://doi.org/10.1016/j.bbamcr.2023.119510
L.C. Crowley, B.J. Marfell, A.P. Scott, N.J. Waterhouse, Cold Spring Harb. Protoc., 2016 (2016). https://doi.org/10.1101/pdb.prot087288
K. Xie, K. Zhang, J. Kong, C. Wang, Y. Gu, C. Liang, T. Jiang, N. Qin, J. Liu, X. Guo, R. Huo, M. Liu, H. Ma, J. Dai, Z. Hu, Cancer Med. 7, 157–166 (2018). https://doi.org/10.1002/cam4.1248
Y. Hu, G.K. Smyth, J. Immunol. Methods 347, 70–78 (2009). https://doi.org/10.1016/j.jim.2009.06.008
A.V. Das, J. James, S. Bhattacharya, A.N. Imbalzano, M.L. Antony, G. Hegde, X. Zhao, K. Mallya, F. Ahmad, E. Knudsen, I. Ahmad, J. Biol. Chem. 282, 35187–35201 (2007). https://doi.org/10.1074/jbc.M706742200
A. Subramanian, P. Tamayo, V.K. Mootha, S. Mukherjee, B.L. Ebert, M.A. Gillette, A. Paulovich, S.L. Pomeroy, T.R. Golub, E.S. Lander, J.P. Mesirov, Proc. Natl. Acad. Sci. U. S. A. 102, 15545–15550 (2005). https://doi.org/10.1073/pnas.0506580102
B.A. Van Tine, J.C. Kappes, N.S. Banerjee, J. Knops, L. Lai, R.D. Steenbergen, C.L. Meijer, P.J. Snijders, P. Chatis, T.R. Broker, P.T. Moen Jr., L.T. Chow, J. Virol. 78, 11172–11186 (2004). https://doi.org/10.1128/JVI.78.20.11172-11186.2004
C.A. Burmeister, S.F. Khan, G. Schafer, N. Mbatani, T. Adams, J. Moodley, S. Prince, Tumour Virus Res. 13, 200238 (2022). https://doi.org/10.1016/j.tvr.2022.200238
P. Dong, Y. Xiong, Y. Konno, K. Ihira, D. Xu, N. Kobayashi, J. Yue, H. Watari, Front. Cell Dev. Biol. 9, 656993 (2021). https://doi.org/10.3389/fcell.2021.656993
K.J. Sales, Human papillomavirus and cervical cancer. In Cancer and Inflammation Mechanisms: Chemical, Biological, and Clinical Aspects. 165–180 (2014)
S. Li, X. Hong, Z. Wei, M. Xie, W. Li, G. Liu, H. Guo, J. Yang, W. Wei, S. Zhang, Front. Microbiol. 10, 2483 (2019). https://doi.org/10.3389/fmicb.2019.02483
E.K. Yim, J.S. Park, Cancer Res. Treat. 37, 319–324 (2005). https://doi.org/10.4143/crt.2005.37.6.319
X. Wang, X. Tong, H. Gao, X. Yan, X. Xu, S. Sun, Q. Wang, J. Wang, Int. J. Oncol. 45, 2385–2392 (2014). https://doi.org/10.3892/ijo.2014.2673
H. Huang, X. Yu, X. Han, J. Hao, J. Zhao, G. Bebek, S. Bao, R.A. Prayson, A.M. Khalil, E. Jankowsky, J.S. Yu, Cell Rep. 34, 108522 (2021). https://doi.org/10.1016/j.celrep.2020.108522
S. Siddiqi, M. Terry, I. Matushansky, PLoS One 7, e33711 (2012). https://doi.org/10.1371/journal.pone.0033711
L. Yang, L. Bi, Q. Liu, M. Zhao, B. Cao, D. Li, J. Xiu, Dis. Markers 2015, 383056 (2015). https://doi.org/10.1155/2015/383056
Z. Chen, Q. Che, F.Z. Jiang, H.H. Wang, F.Y. Wang, Y. Liao, X.P. Wan, Biochem. Biophys. Res. Commun. 463, 876–880 (2015). https://doi.org/10.1016/j.bbrc.2015.06.028
Acknowledgements
We acknowledge the University Grants Commission (UGC), Government of India, for the Senior Research Fellowship to MK and PSR. We want to thank all Neuro Stem Cell Biology Lab (RGCB) members and Jayasree R for their various technical supports. We thank Dr. Zhi-Ming Zheng for providing the pZMZ70 plasmid. We acknowledge Dr. Arumugam Rajavelu and Dr. Tessy Thomas Maliekkal for their valuable suggestions in experiment design. We thank Ms. Jayalekshmi V S and Ms. Neethu Krishnan for their help in histological work. We are extremely thankful to Dr. Sumitra Shankar for the critical reading of the manuscript. We acknowledge Clevergene Bicorp Pvt. Ltd, Bangalore for total and RIP RNA sequencing.
Funding
This work was supported by Institutional intramural funding. M.K [961/[CSIR-UGC NET DEC 2016] and P.S.R [888/[CSIR-UGC NET DEC 2018]. have received research support from UGC, Govt. of India.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. The first draft of the manuscript was written by MK and all authors commented on previous versions of the manuscript; MK: Data curation, writing–original draft, Data visualization; PSR: assisted with the in vitro experiments; BB: Bioinformatics, Data visualization and helped in in vivo experiments; SVR: performed qRT-PCR analysis; RAP: helped in in vivo experiments; KK: histopathology data analysis and interpretation; MA: assisted with western blot; JI: assisted with immunocytochemistry; JJ: Intellectually contributed to experimental design AVD: Conceptualization, Designing experiments, Interpreted data, Critical revision. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All in vivo experiments followed the institutional animal ethical committee (IAEC/815/ANI/2020) guidelines.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
13402_2023_904_MOESM6_ESM.csv
Supplementary file6 (CSV 543 KB) Supplementary Table 5: Significant upregulated genes in PIWIL1 overexpressed HaCaT cells
13402_2023_904_MOESM7_ESM.csv
Supplementary file7 (CSV 515 KB) Supplementary Table 6: Significant downregulated genes in PIWIL1 overexpressed HaCaT cells
13402_2023_904_MOESM10_ESM.csv
Supplementary file10 (CSV 80 KB) Supplementary Table 9: Gene Set Enrichment Analysis between HaCaT control and HaCaT-PIWIL1 overexpressed cells
13402_2023_904_MOESM11_ESM.csv
Supplementary file11 (CSV 1 KB) Supplementary Table 10: The list of genes common in PIWIL1 RIP- Seq data and PIWIL1 positively regulated transcripts
13402_2023_904_MOESM12_ESM.csv
Supplementary file12 (CSV 4 KB) Supplementary Table 11: The list of genes common in PIWIL1 RIP- Seq data and PIWIL1 negatively regulated transcripts
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kunnummal, M., Raveendran, P.S., Basu, B. et al. HPV16 E6/E7-mediated regulation of PiwiL1 expression induces tumorigenesis in cervical cancer cells. Cell Oncol. (2023). https://doi.org/10.1007/s13402-023-00904-8
Accepted:
Published:
DOI: https://doi.org/10.1007/s13402-023-00904-8