Abstract
Background
Diesel particulate matter (DPM) constitutes a significant air pollutant that adversely affects neurological health through the olfactory pathway. Although extensive human epidemiological and animal research exists, the specific mechanisms underlying DPM-induced olfactory dysfunction have not been definitively elucidated.
Objective
This study aimed to conduct a comprehensive analysis of the behavioral, histological, and molecular changes in the olfactory bulb (OB) of mice following intranasal exposure to 10 mg/kg DPM for a duration of four weeks.
Results
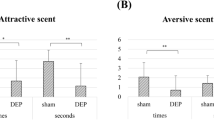
Exposure to DPM led to notable olfactory impairment in the mice, characterized by an elevation in Iba-1-positive microglia, though without inducing neuronal cell death. Transcriptomic evaluation revealed 84 differentially expressed genes (DEGs) in the OB that met the criteria of fold change greater than 1.5 and a p value less than 0.05. Within this set, 55 genes were upregulated and 29 were downregulated. Gene ontology-based functional analysis revealed that these DEGs were primarily related to sensory organ morphogenesis, energy homeostasis, and the regulation of monocyte aggregation. Subsequent investigation using the Kyoto Encyclopedia of Genes and Genomes database identified enriched pathways connected to neuroactive ligand-receptor interactions and calcium signaling.
Conclusion
Our findings suggest a plausible association between DPM-induced olfactory dysfunction and disruptions in a range of molecular pathways. This hypothesis is supported by observed alterations in gene expression and the presence of mild neuroinflammation, primarily driven by microglial activation.





Similar content being viewed by others
Availability of data and materials
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- BP:
-
Biological processes
- Bmp15 :
-
Bone morphogenetic protein 15
- cDNA:
-
Complementary DNA
- CC:
-
Cellular component
- CON:
-
Control
- DEGs:
-
Differentially expressed genes
- DPM:
-
Diesel particulate matter
- FPKM:
-
Fragments per kilobase of transcript per million mapped reads
- GO:
-
Gene ontology
- H&E:
-
Hematoxylin–eosin
- Iba-1:
-
Ionized calcium-binding adapter molecule 1
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
- Mas1 :
-
Mas1 proto-oncogene, G protein-coupled receptor
- MF:
-
Molecular function
- NeuN:
-
Neuronal nuclei
- NF-κB:
-
Nuclear factor kappa B
- OB:
-
Olfactory bulb
- PBS:
-
Phosphate-buffered saline
- PBS-T:
-
PBS containing 0.1% tween 20
- PFA:
-
Paraformaldehyde
- PPI:
-
Protein–protein interaction
- RAS:
-
Renin-angiotensin system
- RNA-seq:
-
RNA-sequencing
- RT-qPCR:
-
Reverse transcription-quantitative real-time polymerase chain reaction
- STRING:
-
Search Tool for the retrieval of interacting genes/proteins
- Tnfsf13b :
-
Tumor necrosis factor superfamily member 13b
References
Ahn JH et al (2016) Long-term observation of neuronal degeneration and microgliosis in the gerbil dentate gyrus after transient cerebral ischemia. J Neurol Sci 363:21–26
Ang MJ et al (2020) Transcriptome profiling reveals novel candidate genes related to hippocampal dysfunction in SREBP-1c knockout mice. Int J Mol Sci 23:4131
Balasubramanian N, James TD, Selvakumar GP, Reinhardt J, Marcinkiewcz CA (2023) Repeated ethanol exposure and withdrawal alters angiotensin-converting enzyme 2 expression in discrete brain regions: implications for SARS-CoV-2 neuroinvasion. Alcohol Clin Exp Res (Hoboken) 47:219–239
Blum JL, Chen LC, Zelikoff JT (2017) Exposure to ambient particulate matter during specific gestational periods produces adverse obstetric consequences in mice. Environ Health Perspect 125:077020
Bos I et al (2012) Changed gene expression in brains of mice exposed to traffic in a highway tunnel. Inhal Toxicol 24:676–686
Brann JH, Firestein SJ (2014) A lifetime of neurogenesis in the olfactory system. Front Neurosci 8:182
Brook RD et al (2010) Particulate matter air pollution and cardiovascular disease: an update to the scientific statement from the American Heart Association. Circulation 121:2331–2378
Calderon-Garciduenas L et al (2002) Air pollution and brain damage. Toxicol Pathol 30:373–389
Chen Z et al (2021) Glial activation and inflammation in the NTS in a rat model after exposure to diesel exhaust particles. Environ Toxicol Pharmacol 83:103584
Chung DW et al (2010) Systemic administration of lipopolysaccharide induces cyclooxygenase-2 immunoreactivity in endothelium and increases microglia in the mouse hippocampus. Cell Mol Neurobiol 30:531–541
Daiber A et al (2020) Effects of air pollution particles (ultrafine and fine particulate matter) on mitochondrial function and oxidative stress—implications for cardiovascular and neurodegenerative diseases. Arch Biochem Biophys 696:108662
Ehsanifar M et al (2021) Hippocampal inflammation and oxidative stress following exposure to diesel exhaust nanoparticles in male and female mice. Neurochem Int 145:104989
Fukuda S, Taga T (2006) Roles of BMP in the development of the central nervous system. Clin Calcium 16:781–785
Ge SX, Jung D, Yao R (2020) ShinyGO: a graphical gene-set enrichment tool for animals and plants. Bioinformatics 36:2628–2629
Genc S, Zadeoglulari Z, Fuss SH, Genc K (2012) The adverse effects of air pollution on the nervous system. J Toxicol 2012:782462
Gu G et al (2023) Ang-(1–7)/MasR axis promotes functional recovery after spinal cord injury by regulating microglia/macrophage polarization. Cell Biosci 13:23
Hajbabaei M, Johnson KC, Guthrie J, Durbin TD (2013) Assessment of the emissions from the use of California Air Resources Board qualified diesel fuels in comparison with Federal diesel fuels. Int J Engine Res 14:138–150
Hoffmann B et al (2009) Chronic residential exposure to particulate matter air pollution and systemic inflammatory markers. Environ Health Perspect 117:1302–1308
Hoffmann BR et al (2017) Mechanisms of Mas1 receptor-mediated signaling in the vascular endothelium. Arterioscler Thromb Vasc Biol 37:433–445
Hong S, Weerasinghe-Mudiyanselage PDE, Kang S, Moon C, Shin T (2023) Retinal transcriptome profiling identifies novel candidate genes associated with visual impairment in a mouse model of multiple sclerosis. Anim Cells Syst (seoul) 27:219–233
Huang JS et al (2022) Immature olfactory sensory neurons provide behaviourally relevant sensory input to the olfactory bulb. Nat Commun 13:6194
Jensen GS, Leon-Palmer NE, Townsend KL (2021) Bone morphogenetic proteins (BMPs) in the central regulation of energy balance and adult neural plasticity. Metabolism 123:154837
Jeong SY, Kim J, Park EK, Baek MC, Bae JS (2020) Inhibitory functions of maslinic acid on particulate matter-induced lung injury through TLR4-mTOR-autophagy pathways. Environ Res 183:109230
Ji X et al (2022) Olfactory bulb microglia activation mediated neuronal death in real-ambient particulate matter exposure mice with depression-like behaviors. Sci Total Environ 821:153456
Juaid N et al (2021) Anti-hepatocellular carcinoma biomolecules: molecular targets insights. Int J Mol Sci 22:10774
Kim J et al (2019) Gene Expression profile of olfactory transduction signaling in an animal model of human multiple sclerosis. Exp Neurobiol 28:74–84
LaFever BJ, Imamura F (2022) Effects of nasal inflammation on the olfactory bulb. J Neuroinflammation 19:294
Lee J et al (2023) Particulate matter exposure and neurodegenerative diseases: a comprehensive update on toxicity and mechanisms. Ecotoxicol Environ Saf 266:115565
Li N, Xia T, Nel AE (2008) The role of oxidative stress in ambient particulate matter-induced lung diseases and its implications in the toxicity of engineered nanoparticles. Free Radic Biol Med 44:1689–1699
Li P et al (2014) Association between individual PM2.5 exposure and DNA damage in traffic policemen. J Occup Environ Med 56:e98–e101
Liu X et al (2023) Traumatic brain injury-induced inflammatory changes in the olfactory bulb disrupt neuronal networks leading to olfactory dysfunction. Brain Behav Immun 114:22–45
Lv H et al (2021) TAK-242 ameliorates olfactory dysfunction in a mouse model of allergic rhinitis by inhibiting neuroinflammation in the olfactory bulb. Int Immunopharmacol 92:107368
Ma Z et al (2023) TMEM59 ablation leads to loss of olfactory sensory neurons and impairs olfactory functions via interaction with inflammation. Brain Behav Immun 111:151–168
Machado CF, Reis-Silva TM, Lyra CS, Felicio LF, Malnic B (2018) Buried food-seeking test for the assessment of olfactory detection in mice. Bio Protoc 8:e2897
Morris RH, Counsell SJ, McGonnell IM, Thornton C (2021) Early life exposure to air pollution impacts neuronal and glial cell function leading to impaired neurodevelopment. BioEssays 43:e2000288
Nelson DR et al (2022) Molecular mechanisms behind safranal’s toxicity to HepG2 cells from dual omics. Antioxidants (Basel) 11:1125
Peixoto MS, de Oliveira Galvao MF, Batistuzzo de Medeiros SR (2017) Cell death pathways of particulate matter toxicity. Chemosphere 188:32–48
Roberts A, Trapnell C, Donaghey J, Rinn JL, Pachter L (2011) Improving RNA-Seq expression estimates by correcting for fragment bias. Genome Biol 12:R22
Song SJ, Park B, Jo K, Kim CS (2021) Damage to olfactory organs of adult zebrafish induced by diesel particulate matter. Int J Mol Sci 23:407
Urist MR (1965) Bone: formation by autoinduction. Science 150:893–899
van Eeden SF, Hogg JC (2002) Systemic inflammatory response induced by particulate matter air pollution: the importance of bone-marrow stimulation. J Toxicol Environ Health A 65:1597–1613
Wada M et al (2021) Behavioral characterization in MPTP/p mouse model of Parkinson’s disease. J Integr Neurosci 20:307–320
Wang H et al (2017) The acute airway inflammation induced by PM(2.5) exposure and the treatment of essential oils in Balb/c mice. Sci Rep 7:44256
Weerasinghe-Mudiyanselage PDE, Ang MJ, Kang S, Kim JS, Moon C (2022) Structural plasticity of the hippocampus in neurodegenerative diseases. Int J Mol Sci 23:3349
Werder EJ, Shrestha S, O'Brien KM, Chen H, Sandler DP (2021) Traffic-related air pollution and olfactory impairment among women in a nationwide US cohort. In: ISEE Conference Abstracts
Win-Shwe TT, Yamamoto S, Fujitani Y, Hirano S, Fujimaki H (2008) Spatial learning and memory function-related gene expression in the hippocampus of mouse exposed to nanoparticle-rich diesel exhaust. Neurotoxicology 29:940–947
Acknowledgements
Not applicable.
Funding
This work was supported by a grant from the National Research Foundation (NRF) of Korea funded by the Korean Government (NRF-2020R1A4A1019395; NRF-2022R1A2C100402212).
Author information
Authors and Affiliations
Contributions
JL and CM designed the research plan. JL, PDEW-M, BK, SK, J-SK, and CM performed the experiments and analyzed the data. JL, PDEW-M, and CM wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Jeongmin Lee: declares no conflict of interest. Poornima D.E. Weerasinghe-Mudiyanselage: declares no conflict of interest. Bohye Kim: declares no conflict of interest. Sohi Kang: declares no conflict of interest. Joong-Sun Kim: declares no conflict of interest. Changjong Moon: declares no conflict of interest.
Ethical approval
All the mice experimental protocols were approved by the Animal Care and Use Committee of Chonnam university, all animals were treated according to the guidelines for animal experimentation of Chonnam university in Gwangju, Republic of Korea (8 November 2022; CNU IACUC-YB-2022–136).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lee, J., Weerasinghe-Mudiyanselage, P.D.E., Kim, B. et al. Impact of diesel particulate matter on the olfactory bulb of mice: insights from behavioral, histological, and molecular assessments. Mol. Cell. Toxicol. (2023). https://doi.org/10.1007/s13273-023-00414-6
Accepted:
Published:
DOI: https://doi.org/10.1007/s13273-023-00414-6