Abstract
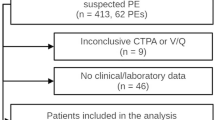
The accuracy of the classic scores that help stratify the pretest clinical probability of pulmonary embolism (PE) in SARS-CoV-2 infection (COVID-19) is low. Therefore, to estimate the risk of PE in these patients, a new set of guidelines must be established. The recently published CHEDDAR score proposes a new diagnostic strategy to reduce the use of computed tomography pulmonary angiography (CTPA) in non-critically ill SARS-COV-2 patients with suspected PE. According to the nomogram, patients are segregated into low-risk (< 182 points) or high-risk (≥ 182 points) based on the best cut-off value to discard PE in the original cohort. We aimed to externally validate this diagnostic strategy in an independent cohort. We analyzed data from two retrospective cohorts of hospitalized non-critically ill COVID-19 patients who underwent a CTPA due to suspicion for PE. CHEDDAR score was applied. As per the CHEDDAR nomogram, patients were classified as having a low or high clinical pre-test probability. Of the 270 patients included, 69 (25.5%) had PE. Applying the CHEDDAR score, 182 (67.4%) patients could have had PE excluded without imaging. Among 58 patients classified as having high clinical pre-test probability, 39 (67.2%) had PE. Sensitivity, specificity, positive and negative predictive values, and AUC were 56%, 90%, 67%, 85%, and 0.783 (95% CI 0.71–0.85), respectively. We provide external validation of the CHEDDAR score in an independent cohort. Even though the CHEDDAR score showed good discrimination capacity, caution is required in patients classified as having low clinical pre-test probability with a D-dimer value > 3000 ng/mL, and a RALE score ≥ 4.


Similar content being viewed by others
Abbreviations
- AUC:
-
Area under the receiver operating characteristic curve
- CRP:
-
C-reactive protein
- CTPA:
-
Computed tomography pulmonary angiogram
- NPV:
-
Negative predictive value
- PE:
-
Pulmonary embolism
- PPV:
-
Positive predictive value
- RALE:
-
Radiographic assessment of lung edema
References
Klok FA, Kruip MJHA, van der Meer NJM et al (2020) Confirmation of the high cumulative incidence of thrombotic complications in critically ill ICU patients with COVID-19: an updated analysis. Thromb Res 191:148–150. https://doi.org/10.1016/j.thromres.2020.04.041
Gong X, Yuan B, Yuan Y (2022) Incidence and prognostic value of pulmonary embolism in COVID-19: a systematic review and meta-analysis. PLoS ONE 17:e0263580. https://doi.org/10.1371/journal.pone.0263580
Gonzalez-Fajardo JA, Ansuategui M, Romero C et al (2021) Mortality of COVID-19 patients with vascular thrombotic complications. Med Clin (Engl Ed) 156:112–117. https://doi.org/10.1016/j.medcle.2020.10.008
Fabre O, Rebet O, Carjaliu I et al (2020) Severe acute proximal pulmonary embolism and COVID-19: a word of caution. Ann Thorac Surg 110:e409-411. https://doi.org/10.1016/j.athoracsur.2020.04.005
Zuin M, Barco S, Giannakoulas G et al (2023) Risk of venous thromboembolic events after COVID-19 infection: a systematic review and meta-analysis. J Thromb Thrombolysis 55(3):490–498. https://doi.org/10.1007/s11239-022-02766-7
Gul MH, Htun ZM, de Jesus PV et al (2023) Predictors and outcomes of acute pulmonary embolism in COVID-19; insights from US National COVID cohort collaborative. Respir Res 24(1):59. https://doi.org/10.1186/s12931-023-02369-7
Van Dam LF, Kroft LJM, van der Wal LI et al (2020) Clinical and computed tomography characteristics of COVID-19 associated acute pulmonary embolism: a different phenotype of thrombotic disease? Thromb Res 193:86–89. https://doi.org/10.1016/j.thromres.2020.06.010
Tan BK, Mainbourg S, Friggeri A et al (2021) Arterial and venous thromboembolism in COVID-19: a study-level meta-analysis. Thorax 76(10):970–979. https://doi.org/10.1136/thoraxjnl-2020-215383
Suh YJ, Hong H, Ohana M et al (2021) Pulmonary embolism and deep vein thrombosis in COVID-19: a systematic review and meta-analysis. Radiology 298:70–80. https://doi.org/10.1148/radiol.2020203557
Ackermann M, Verleden SE, Kuehnel M et al (2020) Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. N Engl J Med 383(2):120–128. https://doi.org/10.1056/NEJMoa2015432
Kahn SR, de Wit K (2022) Pulmonary embolism. N Engl J Med 387:45–57. https://doi.org/10.1056/NEJMcp2116489
Righini M, Robert-Ebadi H, Le Gal G (2017) Diagnosis of acute pulmonary embolism. J Thromb Haemost 15(7):1251–1261. https://doi.org/10.1111/jth.13694
Franco-Moreno AI, Bustamante-Fermosel A, Ruiz-Giardin JM et al (2023) Utility of probability scores for the diagnosis of pulmonary embolism in patients with SARS-CoV-2 infection: a systematic review. Rev Clin Esp (Barc) 223(1):40–49. https://doi.org/10.1016/j.rceng.2022.07.004
Rostami M, Mansouritorghabeh H (2020) D-dimer level in COVID-19 infection: a systematic review. Expert Rev Hematol 13:1265–1275. https://doi.org/10.1080/17474086.2020.1831383
Franco-Moreno A, Brown-Lavalle D, Rodríguez-Ramírez N et al (2023) Clinical prediction model for pulmonary thrombosis diagnosis in hospitalized patients with SARS-CoV-2 infection. J Clin Transl Res 9(2):59–68
von Elm E, Altman DG, Egger M et al (2007) STROBE initiative. strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. BMJ 335:806–808. https://doi.org/10.1136/bmj.39335.541782.AD
Swets JA (1988) Measuring the accuracy of diagnostic systems. Science 240(4857):1285–1293. https://doi.org/10.1126/science.3287615
Dronkers CEA, van der Hulle T, Le Gal G et al (2017) Subcommittee on predictive and diagnostic variables in thrombotic disease. Towards a tailored diagnostic standard for future diagnostic studies in pulmonary embolism: communication from the SSC of the ISTH. J Thromb Haemost 15(5):1040–1043. https://doi.org/10.1111/jth.13654
Acknowledgements
CHEDDAR Research Group: David Brown-Lavalle, Nuria Muñoz-Rivas, Eva Moya-Mateo, María Teresa Bellver-Álvarez, Ana Bustamante-Fermosel, Carmen Campos-Rebollo, María García-Lorente, Idoia Pagai-Valcárcel, Roberto Pedrero-Tomé.
Funding
No specific funding was received from any bodies in the public, commercial or not-for-profit sectors to carry out the work described in this manuscript.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared no conflicts of interest.
Ethical approval
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000. Ethics approval was obtained from the Hospital Universitario Clínico San Carlos (code 22/569-E). The need for Informed Consent was waived by the Ethics Committee of the Hospital Universitario Clínico San Carlos due to the retrospective nature of the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The members of the CHEDDAR Research Group has been included in “Acknowledgements” section.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Franco-Moreno, A., Palma-Huerta, E., Fernández-Vidal, E. et al. External validation of the CHEDDAR score for suspected pulmonary embolism in patients with SARS-CoV-2 infection in an independent cohort. J Thromb Thrombolysis 57, 352–357 (2024). https://doi.org/10.1007/s11239-023-02918-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-023-02918-3