Abstract
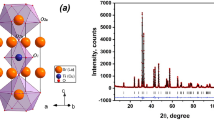
Layered perovskite-like niobate CsBa2Nb3O10 has been synthesized in a pure single-phase state for the first time using both nitrates and carbonates of cesium and barium. Unlike its Ca-, Sr- and Pb-containing analogs, the niobate obtained was shown not to undergo substitution of interlayer alkali cations with protons (protonation) upon acid treatments under various conditions. A potential reason for its chemical inactivity may consist in partial disordering of cesium and barium cations between the interlayer space and perovskite slab, hindering the interlayer ion exchange. Optical bandgap energy of CsBa2Nb3O10, being equal to 2.8 eV, potentially allows using visible light (λ < 443 nm) for driving photocatalytic reactions. However, the photocatalytic potential of this niobate towards hydrogen production remains untapped since the activity of the interlayer space in protonation and hydration reactions, as shown earlier, is a fundamentally important factor determining the photocatalytic performance of ion-exchangeable layered perovskite-like oxides.





Similar content being viewed by others
REFERENCES
M. Dion, M. Ganne, and M. Tournoux, Mater. Res. Bull. 16, 1429 (1981). https://doi.org/10.1016/0025-5408(81)90063-5
K. Domen, Y. Ebina, T. Sekine, et al., Catal. Today 16, 479 (1993). https://doi.org/10.1016/0920-5861(93)80088-I
A. J. Jacobson, J. T. Lewandowski, and J. W. Johnson, J. Less-Common Met. 116, 137 (1986). https://doi.org/10.1016/0022-5088(86)90224-9
T. Kawaguchi, K. Horigane, Y. Itoh, et al., Phys Condens. Matter 536, 830 (2018). https://doi.org/10.1016/j.physb.2017.09.060
L. Fang, H. Zhang, and R. Yuan 17, 3 (2002).
C. H. Mahler, B. L. Cushing, J. N. Lalena, et al., Mater. Res. Bull. 33, 1581 (1998). https://doi.org/10.1016/S0025-5408(98)00166-4
M. Fang, C. H. Kim, and T. E. Mallouk, Chem. Mater. 11, 1519 (1999). https://doi.org/10.1021/cm981065s
J. Yoshimura, Y. Ebina, J. Kondo, et al., J. Phys. Chem. 97, 1970 (1993). https://doi.org/10.1021/j100111a039
Y. Liou and C. M. Wang, J. Electrochem. Soc. 143, 1492 (1996). https://doi.org/10.1149/1.1836668
C. Ziegler, T. Dennenwaldt, D. Weber, et al., Z. Anorg. Allg. Chem. 643, 1668 (2017). https://doi.org/10.1002/zaac.201700269
H. Fukuoka, T. Isami, and S. Yamanaka, J. Solid State Chem. 151, 40 (2000). https://doi.org/10.1006/jssc.2000.8619
R. E. Schaak and T. E. Mallouk, Chem. Mater. 14, 1455 (2002). https://doi.org/10.1021/cm010689m
S. Tahara and Y. Sugahara, Langmuir 19, 9473 (2003). https://doi.org/10.1021/la0343876
S. Tahara, Y. Takeda, and Y. Sugahara, Chem. Mater. 17, 6198 (2005). https://doi.org/10.1021/cm0514793
A. Shimada, Y. Yoneyama, S. Tahara, et al., Chem. Mater. 21, 4155 (2009). https://doi.org/10.1021/cm900228c
A. D. Khramova, O. I. Silyukov, S. A. Kurnosenko, et al., Molecules 28, 4807 (2023). https://doi.org/10.3390/molecules28124807
V. V. Voytovich, S. A. Kurnosenko, O. I. Silyukov, et al., Front. Chem. 8 (2020). https://doi.org/10.3389/fchem.2020.00300
V. V. Voytovich, S. A. Kurnosenko, O. I. Silyukov, et al., Catalysts 11, 897 (2021). https://doi.org/10.3390/catal11080897
Y. Ebina, T. Sasaki, and M. Watanabe, Solid State Ionics 151, 177 (2002). https://doi.org/10.1016/S0167-2738(02)00707-5
T. Sasaki, J. Ceram. Soc. Jpn. 115, 9 (2007). https://doi.org/10.2109/jcersj.115.9
V. Nicolosi, M. Chhowalla, M. G. Kanatzidis, et al., Science 340, 6139 (2013). https://doi.org/10.1126/science.1226419
T. H. Wang, C. N. Henderson, T. I. Draskovic, et al., Chem. Mater. 26, 898 (2014). https://doi.org/10.1021/cm401803d
H. Gao, S. Shori, X. Chen, et al., J. Colloid Interface Sci. 392, 226 (2013). https://doi.org/10.1016/j.jcis.2012.09.079
M. Sakaki, Y. Q. Feng, and K. Kajiyoshi, J. Solid State Chem. 277, 253 (2019). https://doi.org/10.1016/j.jssc.2019.06.018
Y.-S. Han, I. Park, and J.-H. Choy, J. Mater. Chem. 11, 1277 (2001). https://doi.org/10.1039/b006045n
W.-J. Lee, H. J. Yeo, D.-Y. Kim, et al., Bull. Korean Chem. Soc. 34, 2041 (2013). https://doi.org/10.5012/bkcs.2013.34.7.2041
F. Hashemzadeh, Water Sci. Technol. 73, 1378 (2016). https://doi.org/10.2166/wst.2015.610
S.-H. Kweon, M. Im, W.-H. Lee, et al., J. Mater. Chem. C 4, 178 (2016). https://doi.org/10.1039/C5TC03815D
V. Thangadurai, P. Schmid-Beurmann, and W. Weppner, J. Solid State Chem. 158, 279 (2001). https://doi.org/10.1006/jssc.2001.9108
E. Zahedi, M. Hojamberdiev, and M. F. Bekheet, RSC Adv. 5, 88725 (2015). https://doi.org/10.1039/c5ra13763b
J. R. Reddy, S. Kurra, R. Guje, et al., Ceram. Int. 41, 2869 (2015). https://doi.org/10.1016/j.ceramint.2014.10.109
C. N. Henderson, Studies on the Exfoliation, Reassembly and Applications of Layered Materials, The Pennsylvania State University, 2013.
I. A. Rodionov, E. A. Maksimova, A. Y. Pozhidaev, et al., Front. Chem. 7, 1 (2019). https://doi.org/10.3389/fchem.2019.00863
I. A. Rodionov, E. O. Gruzdeva, A. S. Mazur, et al., Catalysts 12, 1556 (2022). https://doi.org/10.3390/catal12121556
S. A. Kurnosenko, V. V. Voytovich, O. I. Silyukov, et al., Catalysts 13, 749 (2023). https://doi.org/10.3390/catal13040749
S. A. Kurnosenko, V. V. Voytovich, O. I. Silyukov, et al., Catalysts 11, 1279 (2021). https://doi.org/10.3390/catal13040749
S. A. Kurnosenko, V. V. Voytovich, O. I. Silyukov, et al., Catalysts 13, 614 (2023). https://doi.org/10.3390/catal13030614
I. A. Zvereva, O. I. Silyukov, and M. V. Chislov, Russ. J. Gen. Chem. 81, 1434 (2011). https://doi.org/10.1134/S1070363211070061
S. A. Kurnosenko, A. A. Burov, O. I. Silyukov, et al., Glas. Phys. Chem. 49, 160 (2023). https://doi.org/10.1134/S1087659622600971
L. V. Yafarova, O. I. Silyukov, T. D. Myshkovskaya, et al., J. Therm. Anal. Calorim. 143, 87 (2021). https://doi.org/10.1007/s10973-020-09276-9
J. -M. Jehng and I. E. Wachs, Chem. Mater. 3, 100 (1991). https://doi.org/10.1021/cm00013a025
Y. Hong and S.-J. Kim, Bull. Korean Chem. Soc. 17, 730 (1996).
I. Zvereva, Y. Smimov, V. Gusarov, et al., Solid State Sci. 5, 343 (2003). https://doi.org/10.1016/S1293-2558(02)00021-3
E. A. Tugova, Russ. J. Inorg. Chem. 67, 874 (2022). https://doi.org/10.1134/S0036023622060237
D. S. Shtarev, A. V. Shtareva, and A. Y. Petrova, Russ. J. Inorg. Chem. 67, 1368 (2022). https://doi.org/10.1134/S0036023622090145
H. Shibata, Y. Ogura, Y. Sawa, et al., Boisci. Biotechnol. Biochem. 62, 2306 (1998). https://doi.org/10.1271/bbb.62.2306
Y. Nosaka and A. Nosaka, ACS Energy Lett. 1, 356 (2016). https://doi.org/10.1021/acsenergylett.6b00174
W. Cui, L. Liu, S. Ma, et al., Catal. Today 207, 44 (2013). https://doi.org/10.1016/j.cattod.2012.05.009
N. Xiao, S. Li, X. Li, et al., Chinese J. Catal. 41, 642 (2020). https://doi.org/10.1016/S1872-2067(19)63469-8
ACKNOWLEDGMENTS
The research was conducted using the equipment of the St. Petersburg State University Research Park, namely Center for X-ray Diffraction Studies, Interdisciplinary Center for Nanotechnology, Center for Optical and Laser Research, Center for Chemical Analysis and Materials Research, and Center for Thermal Analysis and Calorimetry. This article is dedicated to the 300th anniversary of St. Petersburg State University.
Funding
The research was supported by the Russian Science Foundation (grant no. 19-13-00184).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kurnosenko, S.A., Silyukov, O.I., Rodionov, I.A. et al. Synthesis, Ion-Exchange and Photocatalytic Properties of Layered Perovskite-Like Niobate CsBa2Nb3O10: Comparative Analysis with Related Dion–Jacobson Phases A\({\text{A}}_{2}^{'}\)Nb3O10 (A = K, Rb, Cs; A' = Ca, Sr, Pb). Russ. J. Inorg. Chem. 68, 1903–1912 (2023). https://doi.org/10.1134/S0036023623602842
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023623602842