Abstract
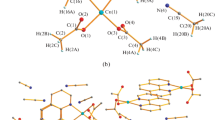
A series of new Pd–Mn bimetallic acetate and pivalate complexes were synthesized and structurally characterized. The starting [Pd(OOCMe)4Mn] complex reacts with N‑donor ligands such as 1,10-phenanthroline (phen) to give [Pd(OOCMe)4Mn(phen)]⋅MeCN (I) (CCDC no. 2217716). The replacement of acetate bridges in the heterometallic Pd–Mn carboxylate complexes with pivalate bridges was investigated. It was shown that complete replacement of all acetate bridges with pivalate ones can occur both in heterometallic complex I, which contains a ligand coordinated to the second metal atom, to give the compound [Pd(Piv)4Mn(phen)]⋅C6H6 (II) (CCDC no. 2217717), structurally related to the initial acetate complex, and in the acetate complex [Pd(OOCMe)4Mn]. The heterometallic co-crystallized pivalate [Pd(Piv)4Mn⋅2HPiv] (III) (CCDC no. 2217718) formed in the latter case can react with 5-nitro-1,10-phenanthroline to give the complex [Pd(Piv)4Mn(Nphen)] (IV) (CCDC no. 2217719).







Similar content being viewed by others
REFERENCES
Buchwalter, P., Rosé, J., and Braunstein, P., Chem. Rev., 2015, vol. 115, no. 1, p. 28.
Kozitsyna, N.Yu., Nefedov, S.E., Yakushev, I.A., et al., Mendeleev Commun., 2007, vol. 17, no. 5, p. 261.
Khramov, E., Belyakova, O., Murzin, V., et al., Z. Anorg. Allg. Chem., 2014, vol. 640, nos. 12−13, p. 2577.
Ershov, B.G., Anan’ev, A.V., Abkhalimov, E.V., et al., Nanotechnologies Russ., 2011, vol. 6, nos. 5–6, p. 323.
Cherkashina, N.V., Churakov, A.V., Yakushev, I.A., et al., Russ. J. Coord. Chem., 2019, vol. 45, no. 4, p. 253. https://doi.org/10.1134/S107032841904002X
Garkul, I.A., Zadesenets, A.V., Korolkov, I.V., et al., J. Struct. Chem., 2020, vol. 61, no. 5, p. 719.
Garkul’, I.A., Zadesenets, A.V., Plyusnin, P.E., et al., Russ. J. Inorg. Chem., 2020, vol. 65, no. 10, p. 1571. https://doi.org/10.1134/S003602362010006X
Smirnova, N.S., Khramov, E.V., Stolarov, I.P., et al., Intermetallics, 2021, vol. 132, p. 107160.
Nefedov, S.E., Vargaftik, M.N., and Moiseev, I.I., Inorg. Chem. Commun., 2006, vol. 9, no. 7, p. 755.
Yakushev, I.A., Sosunov, E.A., Makarevich, Yu.E., et al., J. Struct. Chem., 2022, vol. 63, no. 12, p. 103431.
Pasynskii, A.A., Shapovalov, S.S., Skabitskii, I.V., et al., Russ. J. Inorg. Chem., 2016, vol. 42, no. 9, p. 608. https://doi.org/10.31857/S0132344X22030069
Nefedov, S.E., Yakushev, I.A., Kozitsyna, N.Yu., et al., Inorg. Chem. Commun., 2007, vol. 10, no. 8, p. 948.
Nefedov, S.E., Kozitsyna, N.Yu., Akhmadullina, N.S., et al., Inorg. Chem. Commun., 2011, vol. 14, no. 4, p. 554.
Nefedov, S.E., Kozitsyna, N.Yu., Vargaftik, M.N., et al., Polyhedron, 2009, vol. 28, no. 1, p. 172.
Ho, P.-H., Hung, C.-C., Wang, Y.-H., et al., Asian J. Org. Chem., 2019, vol. 8, no. 2, p. 275.
Pasynskii, A.A. and Shapovalov, S.S., Russ. J. Inorg. Chem., 2016, vol. 42, no. 9, p. 574. https://doi.org/10.1134/S1070328416090050
Shapovalov, S.S., Gordienko, A.V., Pasynskii, A.A., et al., Russ. J. Inorg. Chem., 2011, vol. 37, no. 6, p. 447. https://doi.org/10.1134/S1070328411050125
Yakushev, I.A., Dyuzheva, M.A., Stebletsova, I.A., et al., Russ. J. Inorg. Chem., 2022, vol. 48, no. 3, p. 153. https://doi.org/10.1134/S107032842203006X
Popova, A.S., Ogarkova, N.K., Shapovalov, S.S., et al., Mendeleev Commun., 2022, vol. 32, no. 5, p. 576.
Shmelev, M.A., Gogoleva, N.V., Makarov, D.A., et al., Russ. J. Inorg. Chem., 2020, vol. 46, no. 1, p. 1. https://doi.org/10.1134/S1070328420010078
Nefedov, S.E., Perova, E.V., Yakushev, I.A., et al., Inorg. Chem. Commun., 2009, vol. 12, no. 6, p. 454.
Nefedov, S.E., Kushan, E.V., Uvarova, M.A., et al., Inorg. Chim. Acta, 2013, vol. 395, p. 104.
Nefedov, S.E., Uvarova, M.A., Golubnichaya, M.A., et al., Russ. J. Inorg. Chem., 2014, vol. 59, no. 7, p. 733. https://doi.org/10.1134/S0036023614070171
Kozitsyna, N.Yu., Nefedov, S.E., Dolgushin, F.M., et al., Inorg. Chim. Acta, 2006, vol. 359, no. 7, p. 2072.
Perrin, D.D. and Armarego, W.L.F., Purification of Laboratory Chemicals, Oxford: Pergamon, 1988.
APEX3, SAINT and SADABS, Madison: Bruker AXS Inc., 2016.
Krause, L., Herbst-Irmer, R., Sheldrick, G.M., et al., J. Appl. Crystallogr., 2015, vol. 48, no. 1, p. 3.
Sheldrick, G.M., Acta Crystallogr., Sect. A: Found. Adv., 2015, vol. 71, no. 1, p. 3.
Sheldrick, G.M., Acta Crystallogr., Sect. C: Struct. Chem., 2015, vol. 71, no. 1, p. 3.
Dolomanov, O.V., Bourhis, L.J., Gildea, R.J., et al., J. Appl. Crystallogr., 2009, vol. 42, no. 2, p. 339.
Huang, G.-H., Li, J.-M., Huang, J.-J., et al., Chem.- Eur. J., 2014, vol. 20, no. 18, p. 5240.
Akhmadullina, N.S., Cherkashina, N.V., Kozitsyna, N.Yu., et al., Inorg. Chim. Acta, 2009, vol. 362, no. 6, p. 1943.
Pinsky, M. and Avnir, D., Inorg. Chem., 1998, vol. 37, no. 21, p. 5575.
Cirera, J., Ruiz, E., and Alvarez, S., Chem. Eur. J., 2006, vol. 12, no. 11, p. 3162.
Janjić, G.V., Petrović, P.V., Ninković, D.B., et al., J. Mol. Model., 2011, vol. 17, no. 8, p. 2083.
Yakushev, I.A., Stebletsova, I.A., Cherkashina, N.V., et al., J. Struct. Chem., 2021, vol. 62, no. 9, p. 1411.
Lord, R.C. and Merrifield, R.E., J. Chem. Phys., 1953, vol. 21, no. 1, p. 166.
Funding
This study was supported by the Russian Science Foundation (project no. 18-73-10206-P).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors of this work declare that they have no conflicts of interest.
Additional information
Dedicated to the memory of Professor Sergey Evgen’evich Nefedov
Translated by Z. Svitanko
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sosunov, E.A., Maksimova, A.D., Yakushev, I.A. et al. Chemical Transformations of [Pd(OOCMe)4Mn] in the Reactions with 1,10-Phenanthroline and Pivalic Acid. Russ J Coord Chem 49, 812–821 (2023). https://doi.org/10.1134/S1070328423600754
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328423600754