Abstract
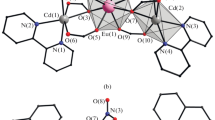
It was shown that refluxing of europium(III) acetate hydrate with trifluoroacetic acid in a dioxane–acetonitrile mixture gives the polymer {[Eu(μ-OOCCF3)3(OH2)2]}n (I) containing dioxane solvate molecules. The reaction of I with [phen(µ-OOCtBu)2(OOCtBu)2] (II) (phen = 1,10-phenanthroline) in CH2Cl2 at room temperature gives a precipitate, the recrystallization of which from acetonitrile affords an unusual trinuclear heterometallic heteroanionic phen2Zn2Eu(µ3-OH)(OOCtBu)4(OOCCF3)2 complex (III). The structure of the products was established from X-ray diffraction data (CCDC no. 2235937–2235939). The optical properties of complex III were studied.






Similar content being viewed by others

REFERENCES
Shi, Q., Zhang, S., Wang, Q., et al., J. Mol. Struct., 2007, vol. 837, p. 185.
Binnemans, K., Chem. Rev., 2009, vol. 109, p. 4283.
Lu, X.-Q., Feng, W.-X., Hui, Y.-N., et al., Eur. J. Inorg. Chem., 2010, vol. 18, p. 2714.
Yang, X., Jones, R.A., Wu, Q., et al., Polyhedron, 2006, vol. 25, p. 271.
Lo, W.-K., Wong, W.-K., Wong, W.-Y., et al., Inorg. Chem., 2006, vol. 45, p. 9315.
Lu, X., Bi, W., Chai, W., et al., New J. Chem., 2008, vol. 32, p. 127.
Belousov, Y.A., Kiskin, M.A., Sidoruk, A.V., et al., Aust. J. Chem., 2022, vol. 75, p. 572.
Colis, J.C.F., Staples, R., Tripp, C., et al., J. Phys. Chem. B, 2005, vol. 109, p. 102.
Yang, X., Jones, R.A., Lynch, V., et al., Dalton Trans, 2005, no. 5, p. 849.
Xu, H.B., Shi, L.X., Ma, E., et al., Chem. Commun., 2006, vol. 15, p. 1601.
Chietal, Y.-X., J. Lumin., 2011, vol. 131, p. 1707.
Xu, H.B., Zhong, Y.T., Zhang, W.X., et al., Dalton Trans., 2010, vol. 39, p. 5676.
Shmelev, M.A., Gogoleva, N.V., Sidorov, A.A., et al., Inorg. Chim. Acta, 2021, p. 120050.
Nikolaevskii, S.A., Yambulatov, D.S., Starikova, A.A., et al., Russ. J. Coord. Chem., 2020, vol. 46, no. 4, p. 260. https://doi.org/10.1134/S1070328420040053
Kiskin, M.A., Varaksina, E.A., Taydakov, I.V., et al., Inorg. Chim. Acta, 2018, vol. 482, p. 85. https://doi.org/10.1016/j.ica.2018.05.037
Egorov, E.N., Kiskin, M.A., Sidorov, A.A., et al., Rus. Chem. Bull., 2013, vol. 62, no. 10, p. 2141. https://doi.org/10.1007/s11172-013-0313-9
Nikolaevskii, S.A., Evstifeev, I.S., and Kiskin, M.A., et al., Polyhedron, 2018, vol. 152, p. 61. https://doi.org/6110.1016/j.poly.2018.06.021
Kiskin, M.A., Dobrokhotova, Zh.V., Bogomyakov, A.S., et al., Russ. Chem. Bull., 2016, vol. 65, p. 1488. https://doi.org/10.1007/s11172-016-1475-z
Rastorguev, A.A., Remova, A.A., Romanenko, G.V., et al., J. Struct. Chem., 2001, vol. 42, p. 907.
Munasinghe, H.N., Szlag, R.G., Imer, M.R., and Rabuffetti, F.A., Inorg. Chem., 2022, vol. 61, p. 5588. https://doi.org/10.1021/acs.inorgchem.2c00196
Sokolova, N.P., Varand, V.L., Romanenko, G.V., et al., Russ. J. Coord. Chem., 2003, vol. 29, p. 362. https://doi.org/10.1023/A:1023636120532
Belyi, V.I., Rastorguev, A.A., Remov, A.A., et al., J. Struct. Chem., 2004, vol. 45, p. 130.
Uvarova, M.A. and Nefedov, S.E., Russ. J. Inorg. Chem., 2021, vol. 66, no. 12, p. 1837. https://doi.org/10.1134/S0036023621120202
Boyle, T.J., Yonemoto, D.T., Sears, J.M., and Treadwell, L.J., Polyhedron, 2017, vol. 131, p. 59.
Barja, B., Baggio, R., Garland, M.T., and Aramendia, P.F., Inorg. Chim. Acta, 2003, vol. 346, p. 187.
Uvarova, M.A. and Nefedov, S.E., Russ. J. Inorg. Chem., 2021, vol. 66, no. 6, p. 839. https://doi.org/10.1134/S0036023621060206
Golubnichaya, M.A., Sidorov, A.A., Fomina, I.G., et al., Russ. Chem. Bull., 1999, vol. 48, p. 1751.
SMART (control) and SAINT (integration). Software. Version 5.0, Madison: Bruker AXS Inc., 1997.
Krause, L., Herbst-Irmer, R., Sheldrick, G.M., and Stalke, D., J. Appl. Crystallogr., 2015, vol. 48, p. 3.
Spek, A.L., Acta Crystallogr., Sect. D: Biol. Crystallogr., 2009, vol. 65, no. 2, p. 148.
Dolomanov, O.V., Bourhis, L.J., Gildea, R.J., et al., J. Appl. Crystallogr., 2009, vol. 42, p. 339.
Casanova, D., Llunell, M., Alemany, P., and Alvarez, S., Chem.-Eur. J., 2005, vol. 11, p. 1479.
Uvarova, M.A., Lutsenko, I.A., Nikiforova, M.E., et al., Russ. J. Coord. Chem., 2022, vol. 48, p. 457. https://doi.org/10.1134/S1070328422080073
Uvarova, M.A. and Nefedov, S.E., Russ. J. Coord. Chem., 2020, vol. 46, no. 9, p. 608. https://doi.org/10.1134/S1070328420090079
Xiao, Y., Huang, P., and Wang, W., J. Clust. Sci., 2015, vol. 26, no. 4, p. 1091.
Dey, D., Roy, S., Purkayastha, R.D., et al., J. Mol. Struct., 2013, vol. 1053, p. 127.
Bochkarev, M.N., Vitukhnovskii, A.G., and Katkova, M.A., Organicheskie svetoizluchayushchie diody (OLED) (Organic Light-Emitting Diodes (OLED)), Nizhny Novgorod: Dekom, 2011.
ACKNOWLEDGMENTS
X-ray diffraction studies, elemental analysis, and IR spectroscopy were performed using equipment of the Center of Molecular Structure Investigation of the Kurnakov Institute of General and Inorganic Chemistry, Russian Academy of Sciences, and was supported by the Ministry of Science and Higher Education of the Russian Federation. Study of the optical properties was carried out using the equipment of the Lebedev Physical Institute, Russian Academy of Sciences.
Funding
This study was supported by the Ministry of Science and Higher Education of the Russian Federation within the framework of the state assignment for the Kurnakov Institute of General and Inorganic Chemistry, Russian Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors of this work declare that they have no conflicts of interest.
Additional information
Translated by Z. Svitanko
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Uvarova, M.A., Taidakov, I.V., Shmelev, M.A. et al. Synthesis and Optical Properties of a Heterocarboxylate Complex with the Zn2Eu Metal Core. Russ J Coord Chem 49, 784–791 (2023). https://doi.org/10.1134/S1070328423600882
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328423600882