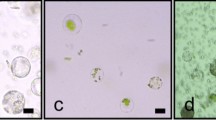
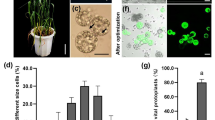
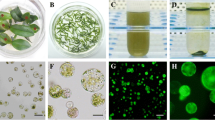
Abstract
Pepper (Capsicum annuum L.) is one of the most widely cultivated species and is highly valued for its pungency. The pungency of pepper is primarily attributed to a group of chemical compounds known as capsaicinoids. These compounds are synthesized in the placental tissue of pepper fruits through the activation of specific genes and enzymes, thereby contributing to their pungency. Protoplast-based gene expression systems have been considered an efficient method for gene function studies, protein-protein interactions, promoter analysis, and subcellular localization. Here, we optimized an efficient protocol for isolating protoplasts from pepper placental tissues and the polyethylene glycol (PEG)-mediated transient expression of green fluorescent proteins (GFP). Several factors affecting GFP expression in intact protoplasts were evaluated and optimized in this study. Protoplast isolation was carried out using 2.0% cellulase “onozuka” R-10 and 0.3% macerozyme R-10 solution. Different amounts of plasmid DNA and various incubation times for transfection with 40% PEG resulted in different transfection efficiencies (78–85%). The highest GFP transformation efficiency was observed when 120 µL protoplast suspension (3 × 107 to 5 × 107/mL) was mixed with 15 µg plasmid DNA and incubated for 25 min with an equal volume of 40% PEG. The improved protocol described in this study can be helpful for the isolation and transfection of pepper placenta-originated protoplasts and for the rapid investigation of pepper gene functions.




Similar content being viewed by others
References
Anilkumar C, Rao AM, Ramesh S, Bhavani B, Pranesh, (2019) Genetics of fruit yield and its component traits under different fruiting habit backgrounds in chilli (Capsicum annuum L.). J Genet 98:1–10
Armstrong CL, Petersen WL, Buchholz WG, Bowen BA, Sulc SL (1990) Factors affecting PEG-mediated stable transformation of maize protoplasts. Plant Cell Rep 9(6):335–339. https://doi.org/10.1007/BF00232864
Belletti P, Marzachi C, Lanteri S (1998) Flow cytometric measurement of nuclear DNA content in Capsicum (Solanaceae). Plant Syst Evol 209(1–2):85–91. https://doi.org/10.1007/BF00991526
Beris D, Kotsaridis K, Vakirlis N, Termentzi A, Theologidis I, Moury B, Vassilakos N (2020) The plasma membrane Cation binding protein 1 affects accumulation of potato virus Y in pepper both at the systemic level and in protoplasts. Virus Res 280:197899. https://doi.org/10.1016/j.virusres.2020.197899
Chen S, Tao L, Zeng L, Vega-Sanchez ME, Umemura K, Wang GL (2006) A highly efficient transient protoplast system for analyzing defence gene expression and protein-protein interactions in rice. Mol Plant Pathol 7(5):417–427. https://doi.org/10.1111/j.1364-3703.2006.00346.x
Csilléry G (2006) Pepper taxonomy and the botanical description of the species. Acta Agron Hung 54(2):151–166. https://doi.org/10.1556/AAgr.54.2006.2.5
Datta K, Datta SK (1999) Transformation of rice via PEG-mediated DNA uptake into protoplasts. Methods Mol Biol 111:335–347. https://doi.org/10.1385/1-59259-583-9:335
de Ruijter NCA, Verhees J, van Leeuwen W, van der Krol AR (2003) Evaluation and comparison of the GUS, LUC and GFP reporter system for gene expression studies in plants. Plant Biol 5(2):103–115. https://doi.org/10.1055/s-2003-40722
Egea C, Dickinson MJ, Candela M, Candela ME (1999) β-1, 3-Glucanase isoenzymes and genes in resistant and susceptible pepper (Capsicum annuum) cultivars infected with Phytophthora capsici. Physiol Plant 107(3):312–318. https://doi.org/10.1034/j.1399-3054.1999.100308.x
Gou YJ, Li YL, Bi PP, Wang DJ, Ma YY, Hu Y, Zhou H, Wen Y, Feng JY (2020) Optimization of the protoplast transient expression system for gene functional studies in strawberry (Fragaria vesca). Plant Cell Tissue Organ Cult (PCTOC) 141(1):41–53. https://doi.org/10.1007/s11240-020-01765-x
Huang H, Wang Z, Cheng J, Zhao W, Li X, Wang H, Zhang Z, Sui X (2013) An efficient cucumber (Cucumis sativus L.) protoplast isolation and transient expression system. Sci Hortic 150:206–212. https://doi.org/10.1016/j.scienta.2012.11.011
Jeon JM, Ahn NY, Son BH, Kim CY, Han CD, Kim GD, Gal SW, Lee SH (2007) Efficient transient expression and transformation of PEG-mediated gene uptake into mesophyll protoplasts of pepper (Capsicum annuum L.). Plant Cell Tissue Organ Cult 88(2):225–232. https://doi.org/10.1007/s11240-006-9194-z
Jung HI, Yan J, Zhai Z, Vatamaniuk OK (2015) Gene functional analysis using protoplast transient assays. Plant functional genomics. Methods Protoc 433–452
Kang JM, Kojima K, Ide Y, Sasaki S (1996) Plantlet regeneration from leaf protoplasts of Populus euphratica. J For Res 1(2):99–102. https://doi.org/10.1007/BF02348311
Kim H, Choi J, Won KH (2020) A stable DNA-free screening system for CRISPR/RNPs-mediated gene editing in hot and sweet cultivars of Capsicum annuum. BMC Plant Biol 20(1):449. https://doi.org/10.1186/s12870-020-02665-0
Li Q, Zhang D, Chen M, Liang W, Wei J, Qi Y, Yuan Z (2016) Development of japonica photo-sensitive genic male sterile rice lines by editing carbon starved anther using CRISPR/Cas9. J Genet Genom 43(6):415–419. https://doi.org/10.1016/j.jgg.2016.04.011
Lin HY, Chen JC, Fang SC (2018) A protoplast transient expression system to enable molecular, cellular, and functional studies in Phalaenopsis orchids. Front Plant Sci 9:843. https://doi.org/10.3389/fpls.2018.00843
Lin W (1983) Isolation of mesophyll protoplasts from mature leaves of soybeans. Plant Physiol 73(4):1067–1069. https://doi.org/10.1104/pp.73.4.1067
Lin Z, Huang LJ, Yu P, Chen J, Du S, Qin G, Zhang L, Li N, Yuan D (2023) Development of a protoplast isolation system for functional gene expression and characterization using petals of Camellia oleifera. Plant Physiol Biochem 201:107885. https://doi.org/10.1016/j.plaphy.2023.107885
Liu S, Chen C, Chen G, Cao B, Chen Q, Lei J (2012) RNA-sequencing tag profiling of the placenta and pericarp of pungent pepper provides robust candidates contributing to capsaicinoid biosynthesis. Plant Cell Tissue Organ Cult (PCTOC) 110(1):111–121. https://doi.org/10.1007/s11240-012-0135-8
Mishra R, Mohanty JN, Mahanty B, Joshi RK (2021) A single transcript CRISPR/Cas9 mediated mutagenesis of CaERF28 confers anthracnose resistance in chilli pepper (Capsicum annuum L.). Planta 254(1):5. https://doi.org/10.1007/s00425-021-03660-x
Ogawa K, Murota K, Shimura H, Furuya M, Togawa Y, Matsumura T, Masuta C (2015) Evidence of capsaicin synthase activity of the Pun1-encoded protein and its role as a determinant of capsaicinoid accumulation in pepper. BMC Plant Biol 15(1):93. https://doi.org/10.1186/s12870-015-0476-7
Pindel A (2007) Optimization of isolation conditions of Cymbidium protoplasts. Folia Hortic 19(2):79–88
Prakash AH, Rao KS, Kumar MU (1997) Plant regeneration from protoplasts of Capsicum annuum L. cv. California Wonder. J Biosci 22(3):339–344. https://doi.org/10.1007/BF02703236
Samanta MK, Dey A, Gayen S (2016) CRISPR/Cas9: an advanced tool for editing plant genomes. Transgen Res 25(5):561–573. https://doi.org/10.1007/s11248-016-9953-5
Schönknecht G, Brown JE, Verchot-Lubicz J (2008) Plasmodesmata transport of GFP alone or fused to potato virus X TGBp1 is diffusion driven. Protoplasma 232(3–4):143–152. https://doi.org/10.1007/s00709-008-0293-z
Sheen J (2001) Signal transduction in maize and Arabidopsis mesophyll protoplasts. Plant Physiol 127(4):1466–1475. https://doi.org/10.1104/pp.010820
Simonne AH, Simonne EH, Eitenmiller RR, Mills HA, Green NR (1997) Ascorbic acid and provitamin a contents in unusually colored bell peppers (Capsicum annuum L.). J Food Compos Anal 10(4):299–311. https://doi.org/10.1006/jfca.1997.0544
Son S, Kim S, Lee KS, Oh J, Choi I, Do JW, Yoon JB, Han J, Park SR (2021) The Capsicum baccatum-specific truncated NLR protein CbCN enhances the innate immunity against Colletotrichum acutatum. Int J Mol Sci 22(14):7672. https://doi.org/10.3390/ijms22147672
Stewart C Jr, Mazourek M, Stellari GM, O’Connell M, Jahn M (2007) Genetic control of pungency in C. chinense via the Pun1 locus. J Exp Bot 58(5):979–991. https://doi.org/10.1093/jxb/erl243
Suzuki K, Mori M (2003) Carotenoid composition of new cultivar of Capsicum annuum during maturation and its high capsanthin content. Nippon Shokuhin Kagaku Kogaku Kaishi 50(7):324–326. https://doi.org/10.3136/nskkk.50.324
Tai T, Staskawicz BJ (2000) Construction of a yeast artificial chromosome library of pepper (Capsicum annuum L.) and identification of clones from the Bs2 resistance locus. Theor Appl Genet 100(1):112–117. https://doi.org/10.1007/s001220050016
Taller J (2006) Gene functioning in pepper. Acta Agron Hung 54(2):233–269. https://doi.org/10.1556/AAgr.54.2006.2.10
Tan B, Xu M, Chen Y, Huang M (2013) Transient expression for functional gene analysis using Populus protoplasts. Plant Cell Tissue Organ Cult (PCTOC) 114(1):11–18. https://doi.org/10.1007/s11240-013-0299-x
Wang M, Zhang J, Wang L, Han L, Zhang X, Feng J (2018) Optimization of production conditions for protoplasts and polyethylene glycol-mediated transformation of Gaeumannomyces tritici. Molecules 23(6):1253. https://doi.org/10.3390/molecules23061253
Whiting S, Derbyshire E, Tiwari BK (2012) Capsaicinoids and capsinoids. A potential role for weight management? A systematic review of the evidence. Appetite 59(2):341–348. https://doi.org/10.1016/j.appet.2012.05.015
Yoo SD, Cho YH, Sheen J (2007) Arabidopsis mesophyll protoplasts: a versatile cell system for transient gene expression analysis. Nat Protoc 2(7):1565–1572. https://doi.org/10.1038/nprot.2007.199
Zhang H, Ma F, Wang X, Liu S, Saeed UH, Hou X, Zhang Y, Luo D, Meng Y, Zhang W, Abid K, Chen R (2020) Molecular and functional characterization of CaNAC035, an NAC transcription factor from pepper (Capsicum annuum L.). Front Plant Sci 11:14. https://doi.org/10.3389/fpls.2020.00014
Zhang J, Mei P, Wang Y, Liu M, Xing D, Wu W, Huang D, Xia W (2023) Efficient mesophyll protoplast isolation and PEG-mediated transient gene expression in greater yam (Dioscorea alata). Trop Plants 2:19. https://doi.org/10.48130/TP-2023-0019
Zhang K, Liu S, Fu Y, Wang Z, Yang X, Li W, Zhang C, Zhang D, Li J (2023) Establishment of an efficient cotton root protoplast isolation protocol suitable for single-cell RNA sequencing and transient gene expression analysis. Plant Methods 19(1):5. https://doi.org/10.1186/s13007-023-00983-6
Zhang Y, Su J, Duan S, Ao Y, Dai J, Liu J, Wang P, Li Y, Liu B, Feng D, Wang J, Wang H (2011) A highly efficient rice green tissue protoplast system for transient gene expression and studying light/chloroplast-related processes. Plant Methods 7(1):30. https://doi.org/10.1186/1746-4811-7-30
Funding
This work was supported by the International Cooperation Program (NRF-2022K2A9A2A06037703) and the Basic Science Research Program (RS-2023-00208020) managed by the National Research Foundation of Korea. Additionally, support was provided by the Korea Institute of Planning and Evaluation for Technology in Food Agriculture and Forestry (IPET) through the Digital Breeding Technology Development Program, funded by the Ministry of Agriculture, Food, and Rural Affairs (MAFRA) (3220683).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Akter, N., Shim, J. & Lee, S. Optimized protocol for isolation and transient expression of placenta-originated protoplast in pepper (Capsicum annuum L.). In Vitro Cell.Dev.Biol.-Plant (2023). https://doi.org/10.1007/s11627-023-10404-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11627-023-10404-x