Abstract
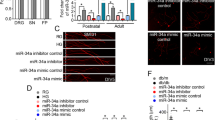
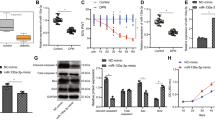
Asprosin, a hormone secreted from adipose tissue, has been implicated in the modulation of cell viability. Current studies suggest that neurological impairments are increased in individuals with obesity-linked diabetes, likely due to the presence of excess adipose tissue, but the precise molecular mechanism behind this association remains poorly understood. In this study, our hypothesis that asprosin has the potential to mitigate neuronal damage in a high glucose (HG) environment while also regulating the expression of microRNA (miRNA)-181a, which is involved in critical biological processes such as cellular survival, apoptosis, and autophagy. To investigate this, dorsal root ganglion (DRG) neurons were exposed to asprosin in a HG (45 mmol/L) environment for 24 hours, with a focus on the role of the protein kinase A (PKA) pathway. Expression of miRNA-181a was measured by using real-time polymerase chain reaction (RT-PCR) in diabetic DRG. Our findings revealed a decline in cell viability and an upregulation of apoptosis under HG conditions. However, pretreatment with asprosin in sensory neurons effectively improved cell viability and reduced apoptosis by activating the PKA pathway. Furthermore, we observed that asprosin modulated the expression of miRNA-181a in diabetic DRG. Our study demonstrates that asprosin has the potential to protect DRG neurons from HG-induced damage while influencing miRNA-181a expression in diabetic DRG. These findings provide valuable insights for the development of clinical interventions targeting neurotoxicity in diabetes, with asprosin emerging as a promising therapeutic target for managing neurological complications in affected individuals.






Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Ayar A, Ozcan M, Alcin E, Serhatlioglu I, Ozcan S, Kutlu S, Kelestimur H (2014) Oxytocin activates calcium signaling in rat sensory neurons through a protein kinase C-dependent mechanism. J Physiol Biochem 70(1):43–8. https://doi.org/10.1007/s13105-013-0278-z
Bischoff A (1973) Ultrastructural pathology of periperal nervous system in early diabetes. Adv Metab Disord 2. Suppl 2:441–449. https://doi.org/10.1016/b978-0-12-027362-1.50052-2
Chen S, Wang X, Qiu CM, Hou JN, Wei XY, Xiang CX, Tang MY, Zhang R, Pei HF (2019) Study of the role and mechanism of asprosin/spartin pathway in cardiac microvascular endothelial injury induced by diabete mellitus. Sichuan da xue xue bao. Yi xue ban. J Sichuan Univ. Med Sci Ed 50(6):827–834
Cnop M, Welsh N, Jonas JC, Jorns A, Lenzen S, Eizirik DL (2005) Mechanisms of pancreatic beta-cell death in type 1 and type 2 diabetes: many differences, few similarities. Diabetes 54(Suppl 2):S97-107. https://doi.org/10.2337/diabetes.54.suppl2.s97
Hekim MG, Kelestemur MM, Bulmus FG, Bilgin B, Bulut F, Gokdere E, Ozdede MR, Kelestimur H, Canpolat S, Ozcan M (2021) Asprosin, a novel glucogenic adipokine: a potential therapeutic implication in diabetes mellitus. Arch Physiol Biochem 1–7. https://doi.org/10.1080/13813455.2021.1894178
Kantharidis P, Wang B, Carew RM, Lan HY (2011) Diabetes complications: the microRNA perspective. Diabetes 60(7):1832–1837. https://doi.org/10.2337/db11-0082
Klöting N, Berthold S, Kovacs P, Schön MR, Fasshauer M, Ruschke K, Stumvoll M, Blüher M (2009) MicroRNA expression in human omental and subcutaneous adipose tissue. PLoS ONE 4(3):e4699. https://doi.org/10.1371/journal.pone.0004699
Lee T, Yun S, Jeong JH, Jung TW (2019) Asprosin impairs insulin secretion in response to glucose and viability through TLR4/JNK-mediated inflammation. Mol Cell Endocrinol 486:96–104. https://doi.org/10.1016/j.mce.2019.03.001
Leong SY, Rao VT, Bin JM, Gris P, Sangaralingam M, Kennedy TE et al (2014) Heterogeneity of oligodendrocyte progenitor cells in adult human brain. Ann Clin Transl Neurol 1:272–283. https://doi.org/10.1002/acn3.55
Li QJ, Chau J, Ebert PJ, Sylvester G, Min H, Liu G, Braich R, Manoharan M, Soutschek J, Skare P, Klein LO, Davis MM, Chen CZ (2007) miR-181a is an intrinsic modulator of T cell sensitivity and selection. Cell 129(1):147–161. https://doi.org/10.1016/j.cell.2007.03.008
Liu Y, Song Y, Zhu X (2017) MicroRNA-181a regulates apoptosis and autophagy process in Parkinson's disease by inhibiting p38 mitogen-activated protein kinase (MAPK)/c-Jun N-terminal kinases (JNK) signaling pathways. Med Sci Monit 23:1597–1606. https://doi.org/10.12659/msm.900218
Liu Y, Zhang S, Xue J, Wei Z, Ao P, Shen B, Ding L (2019) CGRP reduces apoptosis of DRG cells induced by high-glucose oxidative stress injury through PI3K/AKT induction of heme oxygenase-1 and Nrf-2 expression. Oxid Med Cell Longev 2019:2053149. https://doi.org/10.1155/2019/2053149
Liu Y, Zhao Z, Yang F, Gao Y, Song J, Wan Y (2013) microRNA-181a is involved in insulin-like growth factor-1-mediated regulation of the transcription factor CREB1. J Neurochem 126(6):771–780. https://doi.org/10.1111/jnc.12370
Lonze BE, Riccio A, Cohen S, Ginty DD (2002) Apoptosis, axonal growth defects, and degeneration of peripheral neurons in mice lacking CREB. Neuron 34(3):371–385. https://doi.org/10.1016/s0896-6273(02)00686-4
Lorenzen J, Kumarswamy R, Dangwal S, Thum T (2012) MicroRNAs in diabetes and diabetes-associated complications. RNA Biol 9(6):820–827. https://doi.org/10.4161/rna.20162
Lozano-Bartolomé J, Llauradó G, Portero-Otin M, Altuna-Coy A, Rojo-Martínez G, Vendrell J, Jorba R, Rodríguez-Gallego E, Chacón MR (2018) Altered expression of miR-181a-5p and miR-23a-3p is associated with obesity and TNFα-induced insulin resistance. J Clin Endocrinol Metab 103(4):1447–1458. https://doi.org/10.1210/jc.2017-01909
Maranta F, Cianfanelli L, Cianflone D (2021) Glycaemic control and vascular complications in diabetes mellitus type 2. Adv Exp Med Biol 1307:129–152. https://doi.org/10.1007/5584_2020_514
Miska EA, Alvarez-Saavedra E, Townsend M, Yoshii A, Šestan N, Rakic P, Constantine-Paton M, Horvitz HR (2004) Microarray analysis of microRNA expression in the developing mammalian brain. Genome Biol 5(9):1–13
Moon JM, Xu L, Giffard RG (2013) Inhibition of microRNA-181 reduces forebrain ischemia-induced neuronal loss. J Cereb Blood Flow Metab 33(12):1976–1982. https://doi.org/10.1038/jcbfm.2013.157
Ozcan M, Ayar A, Serhatlioglu I, Alcin E, Sahin Z, Kelestimur H (2010) Orexins activates protein kinase C-mediated Ca(2+) signaling in isolated rat primary sensory neurons. Physiol Res 59(2):255–262. https://doi.org/10.33549/physiolres.931739
Ozcan M, Gok ZB, Kacar E, Serhatlioglu I, Kelestimur H (2016) Nesfatin-1 increases intracellular calcium concentration by protein kinase C activation in cultured rat dorsal root ganglion neurons. Neurosci Lett 619:177–181. https://doi.org/10.1016/j.neulet.2016.03.018
Ozcan M, Canpolat S, Bulmus O, Ulker N, Tektemur A, Tekin S, Ozcan S, Serhatlioglu I, Kacar E, Ayar A, Kelestimur H (2019) Agomelatine pretreatment prevents development of hyperglycemia and hypoinsulinemia in streptozotocin-induced diabetes in mice. Fundam Clin Pharmacol 33(2):170–180. https://doi.org/10.1111/fcp.12413
Ozcan S, Kelestemur MM, Hekim MG, Bulmus O, Bulut F, Bilgin B, Canpolat S, Ozcan M (2022a) Asprosin, a novel therapeutic candidate for painful neuropathy: an experimental study in mice. Naunyn Schmiedebergs Arch Pharmacol 395(3):325–335. https://doi.org/10.1007/s00210-021-02197-w
Ozcan S, Ulker N, Bulmus O, Yardimci A, Ozcan M, Canpolat S (2022b) The modulatory effects of irisin on asprosin, leptin, glucose levels and lipid profile in healthy and obese male and female rats. Arch Physiol Biochem 128(3):724–731. https://doi.org/10.1080/13813455.2020.1722706
Pek SL, Sum CF, Lin MX, Cheng AK, Wong MT, Lim SC, Tavintharan S (2016) Circulating and visceral adipose miR-100 is down-regulated in patients with obesity and type 2 diabetes. Mol Cell Endocrinol 427:112–123. https://doi.org/10.1016/j.mce.2016.03.010
Peng L, Ma W, Yi F, Yang YJ, Lin W, Chen H, Zhang X, Zhang LH, Zhang F, Du Q (2014) MicroRNA profiling in Chinese patients with primary Sjögren syndrome reveals elevated miRNA-181a in peripheral blood mononuclear cells. J Rheumatol 41(11):2208–2213. https://doi.org/10.3899/jrheum.131154
Percie du Sert N, Hurst V, Ahluwalia A, Alam S, Avey MT, Baker M, Browne WJ, Clark A, Cuthill IC, Dirnagl U, Emerson M, Garner P, Holgate ST, Howells DW, Karp NA, Lazic SE, Lidster K, MacCallum CJ, Macleod M, Pearl EJ, Petersen OH, Rawle F, Reynolds P, Rooney K, Sena ES, Silberberg SD, Steckler T, Würbel H (2020) The ARRIVE guidelines 2.0: updated guidelines for reporting animal research. PLoS Biol 18(7):e3000410. https://doi.org/10.1371/journal.pbio.3000410
Pope JE, Deer TR, Kramer J (2013) A systematic review: current and future directions of dorsal root ganglion therapeutics to treat chronic pain. Pain Med 14(10):1477–1496. https://doi.org/10.1111/pme.12171
Romere C, Duerrschmid C, Bournat J, Constable P, Jain M, Xia F, Saha PK, Del Solar M, Zhu B, York B, Sarkar P, Rendon DA, Gaber MW, LeMaire SA, Coselli JS, Milewicz DM, Sutton VR, Butte NF, Moore DD, Chopra AR (2016) Asprosin, a fasting-induced glucogenic protein hormone. Cell 165(3):566–579. https://doi.org/10.1016/j.cell.2016.02.063
Rother KI (2007) Diabetes treatment–bridging the divide. N Engl J Med 356(15):1499–1501. https://doi.org/10.1056/NEJMp078030
Russell JW, Golovoy D, Vincent AM, Mahendru P, Olzmann JA, Mentzer A, Feldman EL (2002) High glucose-induced oxidative stress and mitochondrial dysfunction in neurons. FASEB Journal: Official Publication of the Federation of American Societies for Experimental Biology 16:1738–1748. https://doi.org/10.1096/fj.01-1027com
Russell JW, Sullivan KA, Windebank AJ, Herrmann DN, Feldman EL (1999) Neurons undergo apoptosis in animal and cell culture models of diabetes. Neurobiol Dis 6(5):347–363. https://doi.org/10.1006/nbdi.1999.0254
Shi L, Cheng Z, Zhang J, Li R, Zhao P, Fu Z, You Y (2008) hsa-mir-181a and hsa-mir-181b function as tumor suppressors in human glioma cells. Brain Res 1236:185–193. https://doi.org/10.1016/j.brainres.2008.07.085
Shi TJ, Zhang MD, Zeberg H, Nilsson J, Grünler J, Liu SX, Xiang Q, Persson J, Fried KJ, Catrina SB, Watanabe M, Arhem P, Brismar K, Hökfelt TG (2013) Coenzyme Q10 prevents peripheral neuropathy and attenuates neuron loss in the db-/db- mouse, a type 2 diabetes model. Proc Natl Acad Sci U S A 110(2):690–695. https://doi.org/10.1073/pnas.1220794110
Shillo P, Sloan G, Greig M, Hunt L, Selvarajah D, Elliott J, Gandhi R, Wilkinson ID, Tesfaye S (2019) Painful and painless diabetic neuropathies: what is the difference? Curr DiabRep 19(6):1–13
Shin D, Shin JY, McManus MT, Ptacek LJ, Fu YH (2009) Dicer ablation in oligodendrocytes provokes neuronal impairment in mice. Ann Neurol 66:843–857. https://doi.org/10.1002/ana.21927
Sui BD, Xu TQ, Liu JW, Wei W, Zheng CX, Guo BL, Wang YY, Yang YL (2013) Understanding the role of mitochondria in the pathogenesis of chronic pain. Postgrad Med J 89(1058):709–714. https://doi.org/10.1136/postgradmedj-2012-131068
Ugur K, Aydin S (2019) Saliva and blood asprosin hormone concentration associated with obesity. Int J Endocrinol 2019:2521096. https://doi.org/10.1155/2019/2521096
Vincent AM, Olzmann JA, Brownlee M, Sivitz WI, Russell JW (2004) Uncoupling proteins prevent glucose-induced neuronal oxidative stress and programmed cell death. Diabetes 53:726–734. https://doi.org/10.2337/diabetes.53.3.726
Vincent AM, McLean LL, Backus C, Feldman EL (2005) Short-term hyperglycemia produces oxidative damage and apoptosis in neurons. FASEB J 19:638–640. https://doi.org/10.1096/fj.04-2513fje
Vyklický L, Knotková-Urbancovä H (1996) Can sensory neurones in culture serve as a model of nociception? Physiol Res 45(1):1–9
Wang CY, Lin TA, Liu KH, Liao CH, Liu YY, Wu VC, Wen MS, Yeh TS (2019) Serum asprosin levels and bariatric surgery outcomes in obese adults. Int J Obes (lond) 43(5):1019–1025. https://doi.org/10.1038/s41366-018-0248-1
Wang R, Hu W (2021) Asprosin promotes β-cell apoptosis by inhibiting the autophagy of β-cell via AMPK-mTOR pathway. J Cell Physiol 236(1):215–221
Wang X, Tang X, Li M, Marshall J, Mao Z (2005) Regulation of neuroprotective activity of myocyte-enhancer factor 2 by cAMP-protein kinase A signaling pathway in neuronal survival. J Biol Chem 280(17):16705–16713. https://doi.org/10.1074/jbc.M501819200
Wiggins AM, Sorge RE (2022) Corrigendum to “An improved model of type 2 diabetes with effects on glucose tolerance, neuropathy and retinopathy with and without obesity”(✰). Physiol Behav 251:113823. https://doi.org/10.1016/j.physbeh.2022.113823
Yang H, Yang L (2016) Targeting cAMP/PKA pathway for glycemic control and type 2 diabetes therapy. J Mol Endocrinol 57(2):R93-r108. https://doi.org/10.1530/jme-15-0316
Yuan M, Li W, Zhu Y, Yu B, Wu J (2020) Asprosin: a novel player in metabolic diseases. Front Endocrinol (lausanne) 11:64. https://doi.org/10.3389/fendo.2020.00064
Zhang H, Hu W, Zhang G (2020) Circulating asprosin levels are increased in patients with type 2 diabetes and associated with early-stage diabetic kidney disease. Int Urol Nephrol 52(8):1517–1522. https://doi.org/10.1007/s11255-020-02509-8
Zhang Z, Tan Y, Zhu L, Zhang B, Feng P, Gao E, Xu C, Wang X, Yi W, Sun Y (2019) Asprosin improves the survival of mesenchymal stromal cells in myocardial infarction by inhibiting apoptosis via the activated ERK1/2-SOD2 pathway. Life Sci 231:116554. https://doi.org/10.1016/j.lfs.2019.116554
Zochodne DW, Verge VM, Cheng C, Sun H, Johnston J (2001) Does diabetes target ganglion neurones? Progressive sensory neurone involvement in long-term experimental diabetes. Brain 124(Pt 11):2319–2334. https://doi.org/10.1093/brain/124.11.2319
Funding
This study was supported by Turkish Scientific Technical Research Organization (TUBITAK) Project no: 119S138.
Author information
Authors and Affiliations
Contributions
MA, SO performed the in vivo study, planned experiments, analyzed data, and the manuscript; NKT and MGH performed Tunel assay; SD was performed qRt-PCR experiments; ST, BB, FB, MMK, and SC contributed to experiment design, prepared the figures and analyzed the data; and MO planned the experiments, analyzed and interpreted data, and wrote the manuscript. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
This study protocol was approved by the local animal experimentation ethics committee of Fırat University (FUHADEK Decision No: 9, Protocol No: 2019/06).
Consent to Participate
Not applicable.
Competing Interests
The authors declare that there are no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Adam, M., Ozcan, S., Dalkilic, S. et al. Modulation of Neuronal Damage in DRG by Asprosin in a High-Glucose Environment and Its Impact on miRNA181-a Expression in Diabetic DRG. Neurotox Res 42, 5 (2024). https://doi.org/10.1007/s12640-023-00678-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12640-023-00678-9