Abstract
A substantial portion of dementia risk can be attributed to modifiable risk factors that can be affected by lifestyle changes. Identifying the contributors to dementia risk could prove valuable. Recently, machine learning methods have been increasingly applied to healthcare data. Several studies have attempted to predict dementia progression by using such techniques. This study aimed to compare the performance of different machine-learning methods in modeling associations between known cognitive risk factors and future dementia cases. A subset of the AGES-Reykjavik Study dataset was analyzed using three machine-learning methods: logistic regression, random forest, and neural networks. Data were collected twice, approximately five years apart. The dataset included information from 1,491 older adults who underwent a cognitive screening process and were considered to have healthy cognition at baseline. Cognitive risk factors included in the models were based on demographics, MRI data, and other health-related data. At follow-up, participants were re-evaluated for dementia using the same cognitive screening process. Various performance metrics for all three machine learning algorithms were assessed. The study results indicate that a random forest algorithm performed better than neural networks and logistic regression in predicting the association between cognitive risk factors and dementia. Compared to more traditional statistical analyses, machine-learning methods have the potential to provide more accurate predictions about which individuals are more likely to develop dementia than others.




Similar content being viewed by others
Notes
The work performed for this study falls under the machine learning approach to logistic regression, but there is, however, also a reference to a previous publication [28] that applies logistic regression using traditional statistics.
References
Smith GE, Bondi MW. Mild Cognitive Impairment and Dementia: Definitions, Diagnosis, and Treatment. Oxford, New York: Oxford University Press; 2013.
Cahill S, Pierce M, Werner P, Darley A, Bobersky A. A systematic review of the public’s knowledge and understanding of Alzheimer’s disease and dementia. Alzheimer Dis Assoc Disord. 2015;29(3):255–75. https://doi.org/10.1097/WAD.0000000000000102.
Livingston G, et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. The Lancet. 2020;396(10248):413–46. https://doi.org/10.1016/S0140-6736(20)30367-6.
Qiu C, Fratiglioni L. Aging without dementia is achievable: Current evidence from epidemiological research. J Alzheimers Dis. 2018;62(3):933–42. https://doi.org/10.3233/JAD-171037.
Gatz M, et al. Role of genes and environments for explaining Alzheimer disease. Arch Gen Psychiatry. 2006;63(2):168–74. https://doi.org/10.1001/archpsyc.63.2.168.
World Health Organization. Global status report on the public health response to dementia,” World Health Organization, Geneva, 2021. Accessed: Jul. 26, 2022. [Online]. Available: https://www.who.int/publications-detail-redirect/9789240033245
Clare L, et al. Potentially modifiable lifestyle factors, cognitive reserve, and cognitive function in later life: A cross-sectional study. PLoS Med. 2017;14(3):e1002259. https://doi.org/10.1371/journal.pmed.1002259.
Román GC, Jackson RE, Gadhia R, Román AN, Reis J. Mediterranean diet: The role of long-chain ω-3 fatty acids in fish; polyphenols in fruits, vegetables, cereals, coffee, tea, cacao and wine; probiotics and vitamins in prevention of stroke, age-related cognitive decline, and Alzheimer disease. Revue Neurologique. 2019;175(10):724–41. https://doi.org/10.1016/j.neurol.2019.08.005.
Antoniou M. The advantages of bilingualism debate. Annu Rev Linguist. 2019;5(1):395–415. https://doi.org/10.1146/annurev-linguistics-011718-011820.
Bubu OM et al. Sleep, cognitive impairment, and Alzheimer’s disease: A systematic review and meta-analysis. Sleep (2017); 40(1). https://doi.org/10.1093/sleep/zsw032
Shi L, et al. Sleep disturbances increase the risk of dementia: A systematic review and meta-analysis. Sleep Med Rev. 2018;40:4–16. https://doi.org/10.1016/j.smrv.2017.06.010.
Sindi S, et al. Sleep disturbances and dementia risk: A multicenter study. Alzheimer’s & Dementia. 2018;14(10):1235–42. https://doi.org/10.1016/j.jalz.2018.05.012.
Beauchet O, et al. Poor gait performance and prediction of dementia: Results from a meta-analysis. J Am Med Dir Assoc. 2016;17(6):482–90. https://doi.org/10.1016/j.jamda.2015.12.092.
Blankevoort CG, et al. Physical predictors of cognitive performance in healthy older adults: A cross-sectional analysis. PLoS One. 2013;8(7):e70799. https://doi.org/10.1371/journal.pone.0070799.
Cooper R, et al. Objective measures of physical capability and subsequent health: a systematic review. Age Ageing. 2011;40(1):14–23. https://doi.org/10.1093/ageing/afq117.
Cui M, Zhang S, Liu Y, Gang X, Wang G. Grip strength and the risk of cognitive decline and dementia: A systematic review and meta-analysis of longitudinal cohort studies. Front Aging Neurosci (2021); 13 Accessed: Apr. 10, 2023. [Online]. Available: https://www.frontiersin.org/articles/https://doi.org/10.3389/fnagi.2021.625551
Duchowny KA, et al. Associations between handgrip strength and dementia risk, cognition, and neuroimaging outcomes in the UK Biobank Cohort Study. JAMA Netw Open. 2022;5(6):e2218314. https://doi.org/10.1001/jamanetworkopen.2022.18314.
Alanazi HO, Abdullah AH, Qureshi KN. A critical review for developing accurate and dynamic predictive models using machine learning methods in medicine and health care. J Med Syst. 2017;41(4):69. https://doi.org/10.1007/s10916-017-0715-6.
Wiens J, Shenoy ES. Machine learning for healthcare: on the verge of a major shift in healthcare epidemiology. Clin Infect Dis. 2018;66(1):149–53. https://doi.org/10.1093/cid/cix731.
Hashimoto DA, Rosman G, Rus D, Meireles OR. Artificial intelligence in surgery: promises and perils. Ann Surg. 2018;268(1):70–6. https://doi.org/10.1097/SLA.0000000000002693.
Wallace ML, et al. Multidimensional sleep and mortality in older adults: A machine-learning comparison with other risk factors. J Gerontol: Series A. 2019;74(12):1903–9. https://doi.org/10.1093/gerona/glz044.
Seligman B, Tuljapurkar S, Rehkopf D. Machine learning approaches to the social determinants of health in the health and retirement study. SSM - Population Health. 2018;4:95–9. https://doi.org/10.1016/j.ssmph.2017.11.008.
Javeed A, Dallora AL, Berglund JS, Ali A, Ali L, Anderberg P. Machine learning for dementia prediction: A systematic review and future research directions. J Med Syst. 2023;47(1):17. https://doi.org/10.1007/s10916-023-01906-7.
Spooner A, et al. A comparison of machine learning methods for survival analysis of high-dimensional clinical data for dementia prediction. Sci Rep. 2020;10(1):1. https://doi.org/10.1038/s41598-020-77220-w.
Casanova R, Saldana S, Lutz MW, Plassman BL, Kuchibhatla M, Hayden KM. Investigating predictors of cognitive decline using machine learning. J Gerontol B Psychol Sci Soc Sci. 2020;75(4):733–42. https://doi.org/10.1093/geronb/gby054.
Qiu S, et al. Development and validation of an interpretable deep learning framework for Alzheimer’s disease classification. Brain. 2020;143(6):1920–33. https://doi.org/10.1093/brain/awaa137.
Garcia-Gutierrez F, Delgado-Alvarez A, Delgado-Alonso C, et al. Diagnosis of Alzheimer’s disease and behavioural variant frontotemporal dementia with machine learning-aided neuropsychological assessment using feature engineering and genetic algorithms. Int J Geriatric Psychiatry. 2021:1–13. https://doi.org/10.1002/gps.5667.
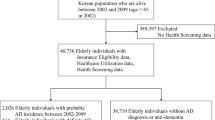
Valsdóttir V, et al. Exposure factors associated with dementia among older adults in Iceland: the AGES-Reykjavik study. GeroScience. 2023;45(3):1953–65. https://doi.org/10.1007/s11357-023-00804-7.
Harris TB, et al. Age, gene/environment susceptibility–Reykjavik study: Multidisciplinary applied phenomics. Am J Epidemiol. 2007;165(9):1076–87. https://doi.org/10.1093/aje/kwk115.
Valsdóttir V, et al. Cognition and brain health among older adults in Iceland: the AGES-Reykjavik study. Geroscience. 2022;44(6):2785–800. https://doi.org/10.1007/s11357-022-00642-z.
Abete P, et al. Cognitive impairment and cardiovascular diseases in the elderly. A heart–brain continuum hypothesis. Ageing Res Rev. 2014;18:41–52. https://doi.org/10.1016/j.arr.2014.07.003.
Cheng S-T. Cognitive reserve and the prevention of dementia: The role of physical and cognitive activities. Curr Psychiatry Rep. 2016;18(9):85. https://doi.org/10.1007/s11920-016-0721-2.
Feinkohl I, Price JF, Strachan MWJ, Frier BM. The impact of diabetes on cognitive decline: potential vascular, metabolic, and psychosocial risk factors. Alzheimer’s Research & Therapy. 2015;7(1):46. https://doi.org/10.1186/s13195-015-0130-5.
Pugazhenthi S, Qin L, Reddy PH. Common neurodegenerative pathways in obesity, diabetes, and Alzheimer’s disease. Biochimica et Biophysica Acta (BBA) - Mol Basis Dis. 2017;1863(5):1037–45. https://doi.org/10.1016/j.bbadis.2016.04.017.
Walker KA, Power MC, Gottesman RF. Defining the relationship between hypertension, cognitive decline, and dementia: A review. Curr Hypertens Rep. 2017;19(3):24. https://doi.org/10.1007/s11906-017-0724-3.
Weiss J, Puterman E, Prather AA, Ware EB, Rehkopf DH. A data-driven prospective study of dementia among older adults in the United States. PLoS ONE. 2020;15(10):e0239994. https://doi.org/10.1371/journal.pone.0239994.
Zaninotto P, Batty GD, Allerhand M, Deary IJ. Cognitive function trajectories and their determinants in older people: 8 years of follow-up in the English Longitudinal Study of Ageing. J Epidemiol Community Health. 2018;72(8):685–94. https://doi.org/10.1136/jech-2017-210116.
McCarrey AC, An Y, Kitner-Triolo MH, Ferrucci L, Resnick SM. Sex differences in cognitive trajectories in clinically normal older adults. Psychol Aging. 2016;31(2):166–75. https://doi.org/10.1037/pag0000070.
Singh-Manoux A, et al. Timing of onset of cognitive decline: results from Whitehall II prospective cohort study. BMJ. 2012;344:d7622. https://doi.org/10.1136/bmj.d7622.
Podsiadlo D, Richardson S. The timed ‘Up & Go’: a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc. 1991;39(2):142–8. https://doi.org/10.1111/j.1532-5415.1991.tb01616.x.
Yesavage JA, et al. Development and validation of a geriatric depression screening scale: A preliminary report. J Psychiatr Res. 1982;17(1):37–49. https://doi.org/10.1016/0022-3956(82)90033-4.
Folstein MF, Folstein SE, McHugh PR. ‘Mini-mental state’: A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189–98. https://doi.org/10.1016/0022-3956(75)90026-6.
Wechsler DW. WAIS-III: Wechsler adult intelligence scale. Manual. New York: Psychological Corporation; 1955.
Pedregosa F, et al. Scikit-learn: Machine Learning in Python. J Mach Learn Res. 2011;12:2825–30.
Cox DR. The Regression Analysis of Binary Sequences. J Royal Stat Soc Series B (Methodological). 1958;20(2):215–42.
Breiman L. Random Forests. Mach Learn. 2001;45(1):5–32. https://doi.org/10.1023/A:1010933404324.
McCulloch WS, Pitts WH. A logical calculus of the ideas immanent in nervous activity. Bull Math Biophys. 1943;5(4):115–33. https://doi.org/10.1007/BF02478259.
Stehman SV. Selecting and interpreting measures of thematic classification accuracy. Remote Sens Environ. 1997;62(1):77–89. https://doi.org/10.1016/S0034-4257(97)00083-7.
Brodersen KH, Ong CS, Stephan KE, Buhmann JM. The Balanced Accuracy and Its Posterior Distribution. In 2010 20th International Conference on Pattern Recognition (2010); 3121–3124. https://doi.org/10.1109/ICPR.2010.764
Chinchor N. “MUC-4 Evaluation Metrics,” in Fourth Message Uunderstanding Conference (MUC-4): Proceedings of a Conference Held in McLean, Virginia (1992); 1992. Accessed: Mar. 09, 2023. [Online]. Available: https://aclanthology.org/M92-1002
Bradley AP. The use of the area under the ROC curve in the evaluation of machine learning algorithms. Pattern Recogn. 1997;30(7):1145–59. https://doi.org/10.1016/S0031-3203(96)00142-2.
Baldi P, Brunak S, Chauvin Y, Andersen CAF, Nielsen H. Assessing the accuracy of prediction algorithms for classification: an overview. Bioinformatics. 2000;16(5):412–24. https://doi.org/10.1093/bioinformatics/16.5.412.
Chicco D, Jurman G. The advantages of the Matthews correlation coefficient (MCC) over F1 score and accuracy in binary classification evaluation. BMC Genomics. 2020;21:6. https://doi.org/10.1186/s12864-019-6413-7.
Luque A, Carrasco A, Martín A, de las Heras A. The impact of class imbalance in classification performance metrics based on the binary confusion matrix. Pattern Recognition. 2019;91:216–31. https://doi.org/10.1016/j.patcog.2019.02.023.
Hanczar B, Hua J, Sima C, Weinstein J, Bittner M, Dougherty ER. Small-sample precision of ROC-related estimates. Bioinformatics. 2010;26(6):822–30. https://doi.org/10.1093/bioinformatics/btq037.
Speiser JL, et al. Machine learning in aging: An example of developing prediction models for serious fall injury in older adults. J Gerontol: Series A. 2021;76(4):647–54. https://doi.org/10.1093/gerona/glaa138.
Powers DMW. Evaluation: from precision, recall and F-measure to ROC, informedness, markedness and correlation. Int J Machine Learning Technol. 2011;2(1):37–63. https://doi.org/10.48550/arXiv.2010.16061.
Funding
This work was supported by The Foundation of St. Josef’s Hospital in cooperation with The Icelandic Gerontological Research Center, National University Hospital of Iceland; Landspítali – University Hospital Research Fund; the Icelandic Gerontological Society; the Council on Aging in Iceland; Helga Jónsdóttir and Sigurliði Kristjánsson Memorial Fund and the Sustainability Institute and Forum (SIF) at Reykjavik University.
The AGES-Reykjavik Study received funding from the National Institutes of Health, Intramural Research Programs of the National Institute of Aging and the National Eye Institute (grant number ZIAEY00401); National Institutes of Health (contract number N01-AG-1–2100); the Icelandic Heart Association and the Icelandic Parliament.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Valsdóttir, V., Jónsdóttir, M.K., Magnúsdóttir, B.B. et al. Comparative study of machine learning methods for modeling associations between risk factors and future dementia cases. GeroScience 46, 737–750 (2024). https://doi.org/10.1007/s11357-023-01040-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-023-01040-9