Abstract
Surface drip fertigation has demonstrated promising results regarding the mitigation of nitrous oxide (N2O) emissions. The use of subsurface irrigation may offer the possibility of reducing these emissions further due to the modification of the soil moisture profile and N allocation, both of which affect the biochemical processes leading to N2O fluxes. However, the mitigation potential of subsurface irrigation combined with different mineral nitrogen (N) fertilizers (ammonium or nitrate-based, use of nitrification inhibitors) still needs to be evaluated. To respond to this need, a 2-year field experiment was set up in central Spain to test two different drip-fertigation systems (surface and subsurface at 30 cm depth) and four N fertilization treatments (control, calcium nitrate, and ammonium sulfate with or without the nitrification inhibitor 3,4-dimethylpyrazole phosphate, DMPP) in an irrigated maize (Zea mays L.) crop. Nitrous oxide emissions, mineral N concentrations (ammonium, NH4+, and nitrate, NO3−), and abundance of key N genes involved in nitrification and denitrification processes were measured in two soil layers (0–20 and 20–40 cm). Regardless of the irrigation system, ammonium sulfate gave the highest cumulative N2O losses in both campaigns, while calcium nitrate and the use of DMPP were the most effective strategies to abate N2O fluxes in the first and second years, respectively. Differences between irrigation systems were not statistically significant for cumulative N2O emissions, despite the clear effect on topsoil mineral N (higher NH4+ and NO3− concentrations in surface and subsurface drip, respectively). Nitrous oxide emissions were positively correlated with soil NH4+ concentrations. Gene abundances were not a trustworthy predictor of N2O losses in the 1st year, although a clear inhibitory effect of fertilization on microbial communities (i.e., ammonia oxidizers, nitrite reducers, and N2O reducers) was observed during this campaign. During the second year, nitrifying and denitrifying genes were affected by irrigation (with higher abundances in the 20–40 cm layer in subsurface than in surface drip) and by the addition of DMPP (which had a detrimental effect on gene abundances in both irrigation systems that disappeared after the fertigation period). In conclusion, the use of DMPP or calcium nitrate instead of ammonium sulfate may enhance the chances for an additional mitigation in both surface and subsurface irrigation systems.
Similar content being viewed by others
Introduction
The expansion of sustainable irrigation requires maintaining or improving crop yields from these systems while minimizing their environmental impacts. For instance, in semiarid areas, irrigated crops are associated with higher nitrous oxide (N2O) emissions (a potent greenhouse gas) than rainfed crops (Cayuela et al. 2017). This is usually explained by high N fertilization rates and optimal conditions (warm soil temperatures and sufficient soil moisture) for microbial processes, leading to N2O emissions from agricultural soils (Aguilera et al. 2013; Butterbach-Bahl et al. 2013; Lassaletta et al. 2021). Meteorological (mainly temperature) and management (e.g., irrigation frequency) conditions usually lead to drying-rewetting episodes—particularly in semi-arid regions—and the co-occurrence of both nitrification and denitrification processes that increase the chances for significant pulses of N2O (Barrat et al. 2021; Guardia et al. 2017a).
The opportunities for N2O mitigation in irrigated crops are related to the improved management of N fertilization and irrigation. The recent meta-analysis of Kuang et al. (2021) reported a significant reduction in N2O emissions using drip irrigation instead of furrow irrigation (average, 32%) and sprinkler irrigation (average, 46%). The N2O mitigation potential of drip irrigation/fertigation compared with other irrigation systems has also been observed under Mediterranean conditions (Cayuela et al. 2017; Guardia et al. 2017b). Fertigation offers the opportunity for a better placement of N (i.e., through the application of soluble N forms close to the plant roots) and split N supply. Drip irrigation keeps part of the soil dry and involves a high irrigation frequency, thus limiting N2O fluxes and the intensity of drying-rewetting episodes in the dry and wet areas, respectively (Barrat et al. 2021). The environmental, agronomical, and economic effectiveness of drip fertigation could be further enhanced by replacing surface drip irrigation with subsurface drip irrigation. Subsurface irrigation applies water below the soil surface by microirrigation emitters, leading less runoff and evaporative losses and more water available to the soil and crop (Lamm 2016). Subsurface irrigation, when properly installed and managed, improves environmental water quality and reduces water consumption and footprint without decreasing yields (Lamm et al. 2012; Plauborg et al. 2022; Singh et al. 2022; Wang et al. 2022a). Regarding N2O, subsurface fertigation maintains high soil moisture and N availability at deeper layers, thus leading to higher N2O concentrations in subsurface layers. Under these conditions, subsurface fertigation is expected to decrease the chances for the diffusion and release of N2O from the soil while promoting N2O consumption (i.e., complete denitrification) across the soil profile (Butterbach-Bahl et al. 2013; Kallenbach et al. 2010), while changing the distribution of soil mineral N. From a N2O mitigation perspective, subsurface drip systems have been successfully compared with sprinkler and furrow irrigation systems (e.g., Ardenti et al. 2022; Bronson et al. 2018; Kallenbach et al. 2010) and with surface drip irrigation under controlled conditions (Wang et al. 2022b; Ye et al. 2018). To our knowledge, however, only Maris et al. (2015) have measured N2O emissions for both drip-irrigation systems (surface versus subsurface) under field conditions. Moreover, in the study of Maris et al. (2015) conducted in an olive orchard, subsurface fertigation was used with almost half of the water dose of the surface drip system. Therefore, it is impossible to know whether the effect of subsurface irrigation on N2O emissions was due to the different irrigation rates or by the system itself and the different water and N application depths.
Besides the effect of water application depth on N2O emissions (surface versus subsurface), the combination of different drip-irrigation practices and different N sources is also an important issue. The substitution of NH4+–N with nitrate (NO3−–N) synthetic fertilizers in semiarid areas with basic pH, poor C-content soils and dominated by nitrification can mitigate N2O emissions (Guardia et al. 2017a; Nishimura et al. 2021). This practice could be particularly effective for subsurface irrigation due to the limited nitrification and the conversion of N2O to dinitrogen (N2) during diffusion to the soil-atmosphere interface. Regarding the N2O released from nitrification (mainly in the topsoil during drying or in the subsurface aerobic macropores), nitrifier denitrification and nitrification coupled with denitrification, the use of nitrification inhibitors such as 3,4-dimethylpyrazole phosphate (DMPP) may be the most effective N2O mitigation strategy (Feng et al. 2016; Gilsanz et al. 2016; Maris et al. 2015; Vilarrasa-Nogué et al. 2020). The DMPP can inhibit nitrifying populations and enhance the abundance of genes involved in the reduction of N2O to N2 (Torralbo et al. 2017). However, to date, no information has been published for drip-fertigated maize that addressed the effect of the form of mineral N (i.e., NH4+–N– and NO3−–N-based) fertilizers and the use of DMPP (compared with a conventional fertilizer without inhibitors) on N2O emissions.
In addition to the effect of burial depth and N source management, it is still necessary to improve our understanding at a mechanistic level, i.e., the effect of these treatments on nitrification and denitrification processes. Together with soil moisture and soil mineral N, the abundances of key genes involved in nitrification and denitrification may shed light on this hitherto unexplored effect of drip burial depth on N2O fluxes. The oxidation of ammonium is catalyzed by enzymes encoded by the amoA gene from the ammonia-oxidizing bacteria (AOB) and archaea (AOA) involved in nitrification (Rotthauwe et al. 1997; Tourna et al. 2008), whereas the nitrite reductase genes nirK and nirS are involved in denitrification (Rinaldo and Cutruzzolà 2007), as well as the N2O reductase genes nosZI and nosZII (Bueno et al. 2012; Hallin et al. 2018). Lower topsoil moisture in subsurface drip irrigation may decrease the abundance of these genes in the upper layer (but not in the subsurface), while combining it with NO3−–N-based fertilizers or nitrification inhibitors may have a detrimental effect on nitrifying communities. Ye et al. (2018) did not find differences in the abundance of nitrifiers between surface and subsurface drip fertigation in a greenhouse experiment, while denitrifiers were not quantified. There is, therefore, a knowledge gap in the understanding of the effect of drip irrigation systems and N sources from a biochemical viewpoint, under field conditions.
In this context, a new experiment was set up in 2020 during two consecutive maize growing seasons studying the effects of surface and subsurface drip irrigation and different N sources on N2O emissions and on the abundances of key genes involved in nitrification and denitrification. We proposed three hypotheses. The first hypothesis is that, compared with surface drip fertigation, subsurface fertigation would decrease N2O emissions by decreasing the abundance of nitrifying and denitrifying genes in the topsoil while promoting N2O consumption (nosZ activity) in subsurface layers. The second hypothesis is that the use of a NO3−–N-based fertilizer and the DMPP inhibitor would mitigate N2O emissions in comparison with a NH4+–N source without inhibitors, due to a detrimental effect on nitrifying communities and a change in the abundances of N2O-producing and -consuming denitrifiers. The third hypothesis we proposed is that the N2O mitigation potential of NO3−–N-based fertilizer and DMPP would be enhanced under subsurface irrigation due to the decreased chances of diffusion of denitrification-derived N2O to the soil-atmosphere interface.
Materials and methods
Experimental site
The experiment was conducted from June, 2020, to November, 2021, at La Chimenea Field Station (IMIDRA), located in Madrid (Spain, 40°03′1.31″N, 3°31′45.07″W 550 m a.s.l.). The topsoil (0–20 cm) was a silty loam (17.6% clay, 55.1% silt, 27.3% sand) Typic xerofluvent (Soil Survey Staff 2014) and had the following physico-chemical properties (mean ± standard error): bulk density, 1.29 ± 0.06 g cm−3; EC (soil:water 1:5), 0.16 ± 0.03 dS m−1; pH (soil:water 1:2.5), 8.60 ± 0.06; and total organic matter, 11.78 ± 0.67 g kg−1. The last 10-year average mean temperatures and yearly rainfall were 14.0 °C and 350 mm, respectively, as reported by SIAR (http://eportal.mapama.gob.es/websiar/Inicio.aspx), recorded at the meteorological station located at La Chimenea. In the 5 years prior to the experiment, the field was maintained as bare fallow.
Experimental design and management
The experimental design consisted of a three-times replicated split plot, with a drip irrigation system (laid over the soil surface, SD, or buried at a depth of 30 cm, BD) as the main factor and N fertilizer (control, ammonium sulfate, AS, ammonium sulfate + DMPP, AS + DMPP, and calcium nitrate, CN) as the split factor. Both the main and split factors were a completely randomized setup. Each subplot (2.5 m × 20 m) covered four maize rows. The subsurface drip-fertigation system was installed at the end of May, 2020 (and revised in May, 2021). The soil was subsequently tilled using a disk harrow, a cultivator, and a rotovator pass, and then seeded with maize (Zea mays L. RAGT Mexican FAO 500, nine plants m−2) on June 19, 2020, and July 2, 2021. After maize seeding, surface drip irrigation lines were set up in the corresponding subplots. One dripper line (with 3.5-L h−1 drippers placed 0.33 m apart) was set up in each maize row for both irrigation systems. A portable sprinkler irrigation system was used to provide enough soil moisture for preseeding, soil preparation, and to promote maize nascence since the soil is prone to crusting, and then these temporary sprinklers were uninstalled after maize emergence. All N fertilizers were applied at a rate of 210 kg N ha−1 and evenly split into two/three fertigation events between V4 and flowering. In the AS + DMPP subplot, 105 kg N ha−1 was applied in two fertigation events on July 27 and August 10, 2020, while in 2021, the doses were split into three fertigation events (70 kg N ha−1 in each). The other fertilizers (CN and AS) were split (70 kg N ha−1 in each application) into three fertigation events (July 27, August 10, and August 17 in 2020; July 26, August 9, and August 23 in 2021) in both campaigns. The N fertilizers were previously dissolved in 0.4 m3 water and then applied for 50 min (with an additional 0.05 m3 of water applied during 10 min between treatments) using the field station fertigation system (valves, pumping system, filters, and tanks). Control subplots received the same amount of water as that applied to the N-fertilized subplots. All subplots (including the control) received phosphorus (P) and potassium (K) fertilizers at rates of 61 kg P ha−1 and 116 kg K ha−1 in 2020 (0–14–14), and 31 kg P ha−1 (as superphosphate) and 166 kg K ha−1 (as potassium sulfate) in 2021. The P and K fertilizers were applied over the soil surface during preseeding preparation and then incorporated into the topsoil. Irrigation was applied twice per week (usually every Monday and Thursday) from maize emergence to mid-September, providing a total of approximately 700 mm in each campaign for both the surface and subsurface drip-irrigated main plots. We decided to apply a similar gross water dose in both irrigation systems, contrary to previous studies (e.g., Maris et al. 2015), in order to be able to discriminate among the effect of water dose and the effect of the irrigation system and the different moisture distributions on N2O emissions and microbial populations. For each week, the water dose was calculated using the reference evapotranspiration data from the field station and the crop coefficient (Kc) of maize at each crop development stage. Decano® herbicide consisting of sulcotrione ([2-(2-chloro-4-mesylbenzoyl)cyclohexane-1,3-dione]) at 30% w:v was applied at 2 L product ha−1 over the entire experimental surface once each maize season, due to the emergence of Cyperus rotundus L. and other annual weeds. Both sulcotrione-related molecules (e.g., mesotrione, applied exceeding field rates) and other herbicides (e.g., atrazine, glyphosate) have temporary effects on N-cycling microbial communities and N transformation rates (Crouzet et al. 2016; Zhang et al. 2018).
Sampling and analysis of N2O emissions
Gas samples were taken from the beginning of June to the end of December in 2020 and from the beginning of June to harvest (November) in 2021. After fertigation events, the gas sampling frequency was increased to two to three times per week during the first month. Afterward, the sampling frequency decreased progressively but covered all rainfall/irrigation events that could lead to N2O peaks after soil rewetting (Barrat et al. 2021). Nitrous oxide fluxes were measured using closed opaque chambers (one chamber per plot; volume 22 L, diameter 35 cm, height 23 cm), which were hermetically closed for 1 h by stainless steel rings inserted (at the beginning of the study) into the soil to a depth of 10 cm to minimize the lateral diffusion of gases. The chambers were placed close to the maize row/drip line and covered the whole moisture gradient from close to the drip line to the central point between maize rows (Fig. S1). Gas samples were taken at the same time of day (10:00–12:00 am) at 0, 30, and 60 min, and put into 20-mL vials. The concentrations of N2O fluxes were quantified with a gas chromatograph equipped with an electron-capture detector (ECD). More details about N2O sampling and analyses can be found in Guardia et al. (2017a).
Soil mineral N and moisture
Three soil cores (0–10 cm) per plot were taken on the same dates as gas sampling to determine the moisture content (expressed as water-filled pore space, WFPS), which was calculated using the soil bulk density (measured by the core method), soil porosity, and gravimetric water content (determined after drying at 105 °C). Additional soil samples were taken in the 0- to 10-cm layer on approximately half of the gas sampling dates, while three (2020) and five (2021) samples were also taken from the 0–20 cm and 20–40 cm layers to determine the mineral N concentrations, EC (soil:water 1:5), and gene abundances in both soil layers. Soil mineral N (i.e., NH4+–N and NO3––N) were extracted using potassium chloride (soil: 1 M KCl, 1:6.25) and then measured using flow injection analysis as reported by Montoya et al. (2022).
Extraction of DNA and quantification of 16S rRNA and N-cycling genes
Gene abundances of the 0–20 cm and 20–40 cm soil layers were determined immediately after the second fertigation event (August 11, 2020, and August 17, 2021), immediately after the third fertigation event (August 18, 2020, and August 31, 2021), and at harvest (November 11, 2020, and November 4, 2021). At each sampling event and depth, composite samples were obtained by mixing three soil cores. Soil microbial DNA was extracted using a commercial kit (Qiagen) and quantified by a Qubit fluorimeter (Invitrogen, Carlsbad, CA, USA). The amoA gene of AOB and AOA (Rotthauwe et al. 1997; Tourna et al. 2008), the nitrite reductase genes nirK (Hallin and Lindgren 1999) and nirS (Throbäck et al. 2004), and the N2O reductase genes nosZI (Henry et al. 2006) and nosZII (Jones et al. 2013) were estimated by quantitative PCR (qPCR) using target primers. In addition, the 16S rRNA gene was quantified as a proxy for the total bacterial communities using taxa-specific primers (Parada et al. 2016; Quince et al. 2011). All quantifications were performed in duplicate on two independent runs, and the potential inhibition of the PCR was also evaluated (no inhibition was observed by any of the DNA extracts), as reported by Montoya et al. (2022).
Calculations and statistical analyses
Cumulative gas emissions for the experimental period were calculated as the sum of the data obtained by multiplying the average flux of two successive determinations for the period between sampling times and adding that amount to the previous cumulative total. Emission factors (EFs) were estimated as the ratio of cumulative N2O emissions to the total amount of applied N (after subtracting those of the corresponding control in each irrigation system). The mitigation percentages, when given, were calculated from the EFs.
Analyses of variance (two-way ANOVA with irrigation and fertilizer as fixed factors) were performed for almost all variables of the experiment (except for climatic variables) using Statgraphics 18-X64. We analyzed soil variables for each soil sampling event, as well as cumulative N2O emissions and the average values of soil parameters (mineral N, gene abundances) throughout the experimental period. The normal distribution of the data and variance homogeneity was previously assessed by the Shapiro–Wilk test and Levene’s tests, respectively, and log-transformed before analysis when necessary. Means were separated by the LSD test at P < 0.05. Simple linear regression analyses were performed to determine the relationships between daily values of N2O, WFPS, air temperature, and mineral N. In addition, cumulative N2O emissions, abundances of N cycling genes, and soil mineral N content were related using principal component analysis (PCA). The analysis was carried out separately for each year considering the different soil depths and sampling dates, and was carried out in R using the package factoextra (Kassambara and Mundt 2020; R Core Team 2021).
Results
Environmental conditions
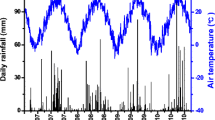
The mean air temperatures from June to mid-November (harvest) were 20.2 °C and 20.3 °C in 2020 and 2021, respectively. The cumulative rainfall from June to mid-November (cropping period) was 139.3 mm and 202.0 mm (71 mm during September) in 2020 and 2021, respectively. The main rainfall events during the critical growth stages of maize occurred on August 11, 2020 (34 mm, Fig. 1a) and September 1, 2021 (46 mm, Fig. 1b). The WFPS of the SD topsoil (0–10 cm) ranged from 25.0 to 62.4% in 2020 (average value, 49.3%; Fig. 1c) and from 24.4 to 63.4% in 2021 (average value, 49.4%; Fig. 1d). In the case of BD plots, the values ranged from 12.9 to 53.9% in 2020 (average value, 37.0%; Fig. 1c) and from 27.1 to 54.8% in 2021 (average value, 43.1%; Fig. 1d).
Soil mineral N
Ammonium concentrations in the topsoil were higher (P < 0.05) in SD (Fig. S2a, b) than in BD (Fig. S2c, d), but these differences were attenuated in the deeper layer (Fig. S2e–h). The highest topsoil concentrations in both years were observed in the AS + DMPP treatment under SD irrigation. In the 20–40 cm depth layer, NH4+ concentrations were generally below 10 mg kg soil−1 in both SD (Fig. S2e, f) and BD (Fig. S2g, h) treatments, with the highest concentrations observed again for the AS + DMPP treatment under SD irrigation in the first and second campaigns (in which the highest values were observed in the last sampling at harvest; Fig. S2f, h). In the 20–40 cm layer, no significant differences in the average concentrations were observed between irrigation or fertilizer treatments.
Topsoil NO3− concentrations were usually higher in BD (Fig. S3c, d) than in SD (Fig. S3a, b) in both years and in 2020 compared with 2021. Values in the topsoil fluctuated greatly, and no significant differences between fertilizer treatments (including control) were observed in the average concentrations, which were higher (by 51–72%, P < 0.05) in BD than in SD. Regarding the 20–40 cm depth, the concentrations were highest on August 11 (after the second fertigation event) in the first campaign (Fig. S3e, g) and on July 30 (after the first fertigation event) in the second campaign (Fig. S3f, h), decreasing to below 10 mg N kg soil−1 (in the last sampling event) in both irrigation systems and years. In this layer, differences in average values between irrigation systems or fertilizer treatments were nonsignificant (P > 0.05) and did not follow a consistent pattern for the different sampling dates.
N2O fluxes
In 2020, the highest N2O emission peak occurred on August 12 (Fig. 2a) after the substantial rainfall of the day before (the “Environmental conditions” section, Fig. 1a) and 2 days after the second fertigation event (Fig. 2a, c). This peak was observed in both irrigation systems and in all fertilizer treatments, including control and AS + DMPP (Fig. 2a, c). During the rest of the cropping seasons, emissions were generally lower than 1 mg N m−2 d−1. As a result, N2O EFs ranged from 0.01 (CN) to 0.07% (AS). No significant differences between irrigation systems were observed, although the magnitude of the August pulse and the cumulative losses were lower in the subsurface drip-irrigated plots (Fig. 2a, c, Table 1).
Daily N2O emissions during the 2020 (A, C) and 2021 (B, D) campaigns in surface (A, B pale colors) and subsurface (C, D dark colors) drip-fertigated plots for the different fertilizer treatments (control; ammonium sulfate, AS; AS with the nitrification inhibitor 3,4-dimethylpyrazole phosphate, AS + DMPP; and calcium nitrate, CN). Vertical lines indicate standard errors (n = 3). Blue arrows denote the dates for N fertilizer applications through drip fertigation
During 2021, the highest N2O fluxes occurred after fertigation events in both irrigation systems, reaching 1.8 mg N m−2 d−1 in SD plots with the AS treatment (Fig. 2b). In BD plots, the highest peaks were also observed in the AS treatment (Fig. 2d). Increases in N2O fluxes were observed at the beginning of the irrigation period (i.e., mid-June) but were lower than those observed after fertigation events. Cumulative emissions significantly decreased in the DMPP-amended subplots in comparison with the AS-only treatment, with the CN treatment had intermediate values (Table 1), with no significant interaction between irrigation systems and fertilizer treatments. Nitrous oxide EFs ranged from 0.00 (AS + DMPP) to 0.14% (AS). Regarding irrigation systems, the BD irrigation decreased the cumulative N2O fluxes in comparison with SD, but differences were not statistically significant (P > 0.05). Daily N2O emissions were positively correlated with soil WFPS (r = 0.37) and air temperature (r = 0.55).
Total abundance of bacterial community and functional genes
In 2020, the greatest abundances of the amoA gene were observed in the unfertilized control in both irrigation systems during the fertigation period (i.e., August 11 and August 18), but not at the end of the cropping period (November 11) (Figs. 3b–d and S4). In addition, DMPP decreased the abundances with respect to the AS-only treatment, particularly on August 18 for BD in the 20–40 cm layer (Fig. S4). Nonsignificant differences were generally observed between irrigation systems. In 2020, the abundances of 16S rRNA and nitrifiers were significantly higher at the end of the maize cropping period than during the fertigation period (except for control subplots) (Figs. 3a and S4). In 2021, BD had higher abundances of AOB and AOA genes in the 20–40 cm layer (Table 2) than SD, while no significant differences were obtained in the topsoil layer or for 16S RNA. Some “depth × treatment” interactions were observed for nitrifiers (Tables S2 and S3). For 16S rRNA, differences between treatments (P < 0.05) were only observed on August 31 (Fig. 4a, b). Buried drip fertigation increased the copy numbers of nitrifying genes in comparison with SD at both depths during the fertigation period (August, Fig. 4c, f). Regarding the fertilizer effect, the AS + DMPP treatment often resulted in lower abundances of AOB in the 0–20 cm layer and SD than the AS-only treatment on August 31. By contrast, the inhibitor had the opposite effect on AOB abundances at the end of the cropping period (November 11) and in both irrigation systems.
Principal component analysis showing the differences in gene abundances (16S RNA, nitrifying and denitrifying genes) in the first campaign (2020) between sampling dates (A) and between fertilization treatments (control; ammonium sulfate, AS; AS with the nitrification inhibitor 3,4-dimethylpyrazole phosphate, AS + DMPP; and calcium nitrate, CN) at each sampling date (August 11 (B); August 18 (C): and November 11 (D)). Colored ellipses denote confidence regions with a confidence level of 95%
Copy numbers of 16S rRNA (A, B), the amoA gene from AOB (C, D), and AOA (E, F) after the second (August 17) and third fertilization (August 31) events and at harvest (November 4) for the fertigation system (surface, SD; and subsurface, BD) combined with different N fertilizer treatments (control; ammonium sulfate, AS; ammonium sulfate with the nitrification inhibitor 3,4-dimethylpyrazole phosphate, AS + DMPP; and calcium nitrate, CN) in the 2021 campaign. The scale of the Y-axes has been adapted in each case to improve the visualization of the data. Vertical lines indicate the standard errors of the mean. Different uppercase letters indicate significant differences between irrigation systems within N fertilizer treatments. Different lowercase letters denote significant differences between different N fertilizer treatments within each irrigation system. In addition, the significant differences between depths and the interaction effect of “depth × treatment” are shown in Tables S1, S2, and S3
In the 2020 campaign, the effect of the irrigation system was negligible, but the effect of fertilization was clearly observed on both sampling dates in August, with a significant decrease in the abundances of denitrifiers in fertilized subplots in comparison with the control, which was observed for both surface and subsurface drip (Figs. 3b–d and S5). In addition, the use of DMPP resulted in lower abundances of denitrifiers in the AS + DMPP than in the AS subplots on August 18 for BD irrigation, especially in the 20–40 cm layer (P < 0.05; Fig. S5b, d, f). On November 11 (i.e., after harvest), the abundances of denitrifying genes were higher than those during August (Figs. 3a and S5), except for the control plots, and no differences were observed between fertilizer or irrigation treatments. During this first campaign, the abundances of 16S RNA and genes involved in nitrification and denitrification were higher in the 0–20 cm layer than in the 20–40 cm layer (Figs. S5 and S6).
In 2021, both nitrite (nirK and nirS) and N2O (nosZI and nosZII) reducer genes generally showed higher abundances (P < 0.05) in BD than in SD during August (Table 2). On August 31, the effect of the nitrification inhibitor resulted in lower abundances of nirK and nirS in AS + DMPP than in the control and AS subplots for BD and at both depths. On November 4, AS + DMPP had higher abundances of both nir genes, which were significant for SD.
Regarding the genes encoding N2O reductase (Fig. 5e–h), no differences between irrigation systems were found on November 4. However, greater abundances of nosZI were observed in BD with respect to SD in August for the control and CN treatments and at 20–40 cm depth (P < 0.05). On August 31, the highest abundances of nosZI were generally observed in the control and AS treatments, while the lowest number of copies of this gene occurred in the AS + DMPP treatment (BD), although no significant differences between fertilizers were found in the topsoil. On November 4 (end of the growing season), the abundances of both nosZ genes were lower in the AS treatment than in the AS + DMPP and the control treatments (particularly in SD, P < 0.05), but differences were only observed in the 20–40 cm soil layer for both irrigation systems.
Copy numbers of nirK (A, B), nirS (C, D), nosZI (E, F), and nosZII (G, H) genes after the second (August 17) and third (August 31) fertigation events and at harvest (November 4) for the fertigation system (surface, SD; and subsurface, BD) combined with different N fertilizer treatments (control; ammonium sulfate, AS; ammonium sulfate with the nitrification inhibitor 3,4-dimethylpyrazole phosphate, AS + DMPP; and calcium nitrate, CN) in the 2021 campaign. The scale of the Y-axes has been adapted in each case to improve the visualization of the data. Vertical lines indicate the standard errors of the mean. Different uppercase letters indicate significant differences between irrigation systems within N fertilizer treatments. Different lowercase letters denote significant differences between different N fertilizer treatments within each irrigation system. In addition, the significant differences between depths and the interaction effect of “depth × treatment” are shown in Tables S4, S5, S6, and S7
Discussion
This study is the first to compare surface and subsurface irrigation in an arable crop under field conditions, using the same amount of water in both systems but combined with different N sources. It also improves our understanding of the main mechanisms that drive these fluxes. The subsurface drip fertigation changed the topsoil distribution of mineral N and the abundances of certain denitrifiers in the 20–40 cm soil layer (Fig. 6). However, only the choice of the N source (calcium nitrate or the use of an inhibitor) was important to mitigate N2O (P < 0.05). Contrasting patterns in gene abundances were observed between the first campaign (i.e., a detrimental effect of all N fertilizers on all genes) and the second campaign (i.e., a significant and transitory effect of DMPP and a stimulatory effect of subsurface drip in the 20–40 cm layer).
Drip burial depth affected microbial communities but not N2O emissions
Contrary to our first hypothesis, subsurface drip did not decrease N2O fluxes compared with subsurface drip, as previously found by Ye et al. (2018) in a greenhouse experiment. In an experiment conducted in an olive orchard, Maris et al. (2015) observed a reduction in N2O emissions with subsurface drip (in comparison with surface drip), but the effect could also be associated with the different amounts of water applied in both drip-fertigation systems (i.e., 422 mm and 221 mm in surface and subsurface drip, respectively). As expected, drip burial depth changed the distribution of soil moisture as well as NH4+ and NO3− ions in the topsoil. Ammonium concentrations were higher in SD while nitrate concentrations were higher in BD (Figs. S2 and S3), but these transitory differences did not have any significant effect on cumulative N2O fluxes. Nitrous oxide EFs were in agreement with those reported for drip-fertigated plots (Flynn et al. 2022; Guardia et al. 2017a; Yangjin et al. 2021; Yu et al. 2022) and were lower than the EFs reported for maize and irrigated Mediterranean croplands (Cayuela et al. 2017). The low N2O emissions in drip fertigation is the result of the water distribution and the improved N supply, both spatially (close to plant roots and dissolved) and temporally (split applications during the critical demand moments) (Tian et al. 2020). In addition, drip fertigation (both surface and subsurface) also leads to a distribution of mineral N that favors low N2O emissions, with high concentrations of NH4+ in the high-moisture bulb area close to the drippers and NO3− transported with water to drier areas (with conditions more favorable for nitrification rather than for denitrification) (Guardia et al. 2017a). The low N2O emissions in drip-fertigated maize were probably the reason why significant differences between surface and subsurface irrigation were not observed. However, our results suggest that further mitigation of greenhouse gas emissions could be achieved by applying different gross irrigation rates. This is mainly because of the positive effect of reducing CO2 equivalent emissions from irrigation (particularly upstream emissions derived from irrigation water consumption) that contribute to the C footprint of irrigated semi-arid croplands (Aguilera et al. 2019; Koushki et al. 2023). Besides, subsurface irrigation may result in additional benefits from water footprint mitigation, enhanced water use efficiency, and climate change adaptation (Lamm 2016).
It could be hypothesized that nitrification would have been favored more in SD than in BD because of the enhanced NH4+ availability in the topsoil. This was indeed observed (P < 0.05, Fig. S2a, c), but probably the higher—and more favorable for denitrification—topsoil moisture in SD than in BD (Fig. 1c, d) limited the relevance of N2O released from nitrification and offset this effect (Pilegaard 2013). The significant increment of nosZ abundances in subsurface versus surface drip (20–40 cm, Table 2) is a likely indicator that complete denitrification (i.e., N2O reduction to N2) may be more favored in BD than in SD (as we initially hypothesized). This effect may have been offset by the higher abundances of nitrifying (i.e., amoA from AOB and AOA) and denitrifying (i.e., nirK and nirS) genes (P < 0.05) in the subsurface layer (Table 2). These higher microbial abundances in BD with respect to SD in the 20–40 cm layer could be indicative of enhanced microbial activity in the subsurface layer. They could also indicate a pivotal role of the limited diffusion of N2O from deeper layers (since these higher abundances were not associated with greater N2O fluxes). The lack of differences between irrigation systems in the topsoil layer (Table 2) is in agreement with that reported by Ye et al. (2018), who found no differences between SD and BD in the abundance of AOB and AOA (0–20 cm layer). The present study provides a more comprehensive view on the effect of both drip burial depths from a mechanistic perspective since it quantifies the abundances of nitrifiers and denitrifiers at two different soil depths. In 2021, gene abundances were not correlated with soil mineral N (Fig. S7), and the relationships between the copy numbers of genes and N2O emissions were weak at both depths (Fig. S8). Cumulative N2O emissions were related to mineral N concentrations in August (Fig. S9), revealing a positive relationship with NH4+, which may highlight the importance of nitrification in calcareous soils in Mediterranean areas (Aguilera et al. 2013).
N2O emissions and microbial communities as influenced by fertilization management
As we hypothesized, the effect of the nitrification inhibitor DMPP resulted in a statistically significant (P < 0.05) decrease in N2O emissions in 2021 (with respect to AS-only), which was observed in both irrigation systems (Table 2). The efficacy of DMPP on N2O mitigation has been demonstrated in different soil and climatic conditions, including dry rainfed, humid rainfed, and irrigated croplands (Fan et al. 2022; Gilsanz et al. 2016; Tufail et al. 2022). It has also been demonstrated when applied through drip fertigation (Maris et al. 2015) and when added to AS (Huérfano et al. 2016). In our study, the mitigation efficacy of DMPP was 34% and 99% in 2020 and 2021, respectively. This result indicates that maybe splitting the fertilizer into three applications was more advisable from an environmental viewpoint than splitting it into two applications. However, these results could also perhaps be explained by the critical rainfall events. In 2020, DMPP reduced N2O fluxes (compared with AS-only) in subsurface irrigation (Fig. 2c) but not during the rewetting peak of surface-fertigated plots (Fig. 2a). This could have been due to the relevance of denitrification in this rewetting pulse (Friedl et al. 2022) or the adsorption of DMPP by surface-reactive particles (Marsden et al. 2016). This rainfall event in August, 2020, therefore, affected the co-location of DMPP and NH4+ and gave rise to the highest N2O emission peak as a result of the reactivation of soil microbial populations and of the anaerobic conditions favoring denitrification (Barrat et al. 2021; Leitner et al. 2017). In 2021, a similar rainfall event occurred, although it was at the beginning of September, leading to a much lower N2O peak than that of the previous year. This was possibly because of the lower soil N availability (after the most intense stages of crop uptake) or the different intensities of the rewetting event (Barrat et al. 2021), which depends strongly on the soil previous dryness conditions.
The abundances of AOB, AOA, nitrite reducers, and N2O reducers could also explain the differences in DMPP efficacy between years. In 2020, the largest effect was observed for N fertilization (in comparison with the control) but not between N sources (Fig. 3c). In 2021, the nitrification inhibitor DMPP generally reduced the abundances of nitrifying genes during the fertigation period (Table 2), particularly on August 31 (Fig. S5b) and in the deeper layer (Table 2). This effect, which was also observed in the first campaign during fertigation (Table S8) and in the subsurface drip plots, was also found by Nair et al. (2021), Lan et al. (2022), and Lei et al. (2022), who also observed a significant inhibition of gross nitrification rates with DMPP. On August 31, the abundance of denitrifiers (nirK and nirS) was also reduced in the plots that received DMPP. This could be mainly due to the indirect effect of a reduced substrate for denitrification (Barrena et al. 2017). However, it could also be a direct effect of the inhibitor, similar to that found by Guardia et al. (2018), who observed a reduction in denitrification using 15N labeling for DMPSA (2-(3,4-dimethyl-1H-pyrazol-1-yl)-succinic acid), which is a pyrazole-based inhibitor like DMPP with a similar inhibition mechanism; Corrochano-Monsalve et al. 2021). In contrast to the findings of Torralbo et al. (2017), we also found a reduction in the abundances of N2O reducers (nosZ communities) with the use of DMPP, as observed for AOB and nir genes. It should be highlighted that the effect of DMPP on nitrifiers and denitrifiers seems to be transitory. This is demonstrated by the consistent increase in abundances of these communities in AS + DMPP compared with AS once the fertigation period ended (i.e., at harvest, Figs. 5 and S7c). At the second soil sampling event (August 31), the abundances were consistent with the effect of DMPP on N2O fluxes (Figs. 4 and 5). This is also supported by Fig. S10 (c, d), in which cluster 3 grouped observations with low N2O emissions and low gene abundances in the topsoil but high gene abundances in the deeper layer. The majority (80%) of the observations of this cluster belonged to the AS + DMPP fertilizer treatment.
The AS fertilizer gave higher cumulative emissions than CN (in agreement with our second hypothesis) in 2020 and in both irrigation systems. The higher emissions in the NH4+–N-based fertilizer versus the NO3−–N-based fertilizer are consistent with previous findings (e.g., Abalos et al. 2014) in semiarid areas where nitrification (itself or coupled with denitrification) is an important process leading to N2O emissions, even in irrigated crops (Guardia et al. 2018). Our results highlight the potential of the right N source (NO3−–N versus NH4+–N fertilizers) to mitigate N2O, although this strategy is not recommended in areas with a humid climate and C-rich soils (Rahman et al. 2021; Velthof and Rietra 2018).
In 2020, N2O emissions were only positively correlated with soil NH4+ concentrations (consistent with the lower cumulative emissions from CN) during the fertigation period (Fig. S10a, b). Generally, the abundances of nitrifying and denitrifying genes in AS (which provides a substrate for nitrifiers and indirectly for denitrifiers) were greater than those in CN plots (Table 2, Figs. 4 and 5), which is consistent with the comparison of cumulative N2O emissions (Table 1). In 2020, the ratio between bacterial and archaeal amoA and all denitrifying genes was higher for control plots (0.75 on average) than for the N-fertilized plots (0.27 on average; data not shown) during the fertigation period. In 2021, this ratio was higher in the DMPP and NC treatments (0.48 on average) than in the control and AS treatments (0.39 and 0.41, respectively; data not shown). The high ratios in low-emitting treatments in both years may suggest that the relative contribution of denitrification to N2O fluxes might not be ignored.
Under the conditions of this study and during the first campaign (2020), the abundances of key genes involved in nitrification and denitrification were not trustworthy predictors of the differences in N2O emissions between treatments, as indicated by the lack of correlation between N2O fluxes and all target genes (Fig. S10c, d). This finding is supported by the study of Zhang et al. (2022), which showed that the abundances of key denitrifying and nitrifying genes were poor predictors of N2O emissions after N addition, in comparison with other abiotic factors. However, nitrification genes were strongly positively correlated with denitrification genes (Figs. 3, S6, S7, and S8), as previously found by Nair et al. (2021).
The present results show contrasting patterns of gene abundances between 2020 and 2021. The first campaign was at the end of a transition from several years of rainfed fallow to intensive drip fertigation. In this campaign there was no effect of the irrigation system but a highly consistent pattern between genes, suggesting an unexpected and strong inhibitory effect of N fertilization (regardless of the N source) on the abundances of all microbial communities (AOB and AOA nitrifiers, nitrite reducers, and N2O reducers) (Fig. 3). This effect was attenuated in 2021, but still observed for, e.g., 16S RNA, nirK, and nosZI at 20–40 cm (Table 2). Additionally, the abundances of all key N genes were higher at harvest (November 4) than during the fertigation period, contradicting what has generally been reported: that N addition stimulates the abundances of genes involved in nitrification and denitrification (Ouyang et al. 2018; You et al. 2022). However, in a pomelo orchard, Zhao et al. (2022) observed a detrimental effect of synthetic N fertilization on the abundance of AOA but not for the abundances of denitrifying genes, as also shown in the recent meta-analysis of Dong et al. (2022). Yang et al. (2022) also reported a negative relationship between N fertilization and microbial diversity.
The potential detrimental effect of N fertilizer on the abundances of nitrifying and denitrifying communities could be explained by two hypotheses. First, the application of fertilizers could have temporally increased soil salinity, thus causing stress on the microbial populations. Even though Sun et al. (2022) found an intense nitrite oxidation activity under saline-sodic soil conditions, other authors have reported that small changes in soil salinity due to N application can modify N2O emission patterns (Wei et al. 2022) and microbial communities (Yao et al. 2022). However, we did not observe significant differences in the EC values of the control and N-fertilized subplots (data not shown). The second hypothesis is related to the different responses of microbial populations to management practices depending on the previous history of the plot, i.e., a “home-field-advantage” effect (Ayres et al. 2006; Perez et al. 2013; Rúa et al. 2016). Our plots were managed as unfertilized fallow for several years, so it could be argued that the control plots were those reflecting similar conditions to the fallow.
Conclusions
This 2-year study under field conditions confirmed that subsurface irrigation was not a significant N2O mitigation strategy in comparison with surface drip fertigation. This result is in spite of various differences observed in topsoil concentrations of mineral N and in the abundances of some genes (including nosZ), particularly in the subsurface layer. However, compared with AS-only, the use of either calcium nitrate or ammonium sulfate with DMPP split into three applications has the potential to further decrease N2O emissions from low-emitting, drip-fertigated crops. The uses of calcium nitrate or ammonium sulfate with DMPP are, therefore, advisable mitigation strategies for these conditions. We recommend that the following four issues should be addressed in future studies. The first is the possible detrimental effect of N fertilization on the abundances of N cycle genes (including nitrite oxidizing bacteria), particularly after significant changes in cropping intensity or a change from rainfed to irrigated agriculture. The second is the relationships of N2O with the distribution of nitrifiers and denitrifiers and mineral N across the soil profile in surface and subsurface drip-fertigated crops. The third issue that we recommend for further study is the possible enhancement of complete denitrification in subsurface drip in comparison with surface drip (i.e., denitrification potential, N2 measurements). The fourth issue is the overall potential of subsurface irrigation for climate change mitigation (including upstream and downstream emissions) and adaptation (including yields, phenology, and N use efficiency). Regarding this last point, the use of an alternative experimental design with different irrigation doses in surface and subsurface irrigation could also be of interest as an approach that is closer to real conditions.
References
Abalos D, Sanchez-Martin L, Garcia-Torres L, Van Groenigen JW, Vallejo A (2014) Management of irrigation frequency and nitrogen fertilization to mitigate GHG and NO emissions from drip-fertigated crops. Sci Total Environ 490:880–888. https://doi.org/10.1016/j.scitotenv.2014.05.065
Aguilera E, Guzmán GI, De Molina MG, Soto D, Infante-Amate J (2019) From animals to machines. The impact of mechanization on the carbon footprint of traction in Spanish agriculture: 1900–2014. J Clean Prod 221:295–305. https://doi.org/10.1016/j.jclepro.2019.02.247
Aguilera E, Lassaletta L, Sanz-Cobena A, Garnier J, Vallejo A (2013) The potential of organic fertilizers and water management to reduce N2O emissions in Mediterranean climate cropping systems. A review. Agric Ecosyst Environ 164:32–52. https://doi.org/10.1016/j.agee.2012.09.006
Ardenti F, Abalos D, Capra F, Lommi M, Maris SC, Perego A, Bertora C, Tabaglio V, Fiorini A (2022) Matching crop row and dripline distance in subsurface drip irrigation increases yield and mitigates N2O emissions. Field Crops Res 289:108732. https://doi.org/10.1016/j.fcr.2022.108732
Ayres E, Dromph KM, Bardgett RD (2006) Do plant species encourage soil biota that specialise in the rapid decomposition of their litter? Soil Biol Biochem 38:183–186. https://doi.org/10.1016/j.soilbio.2005.04.018
Barrat HA, Evans J, Chadwick DR, Clark IM, Le Cocq K, Cardenas LM (2021) The impact of drought and rewetting on N2O emissions from soil in temperate and Mediterranean climates. Eur J Soil Sci 72:2504–2516. https://doi.org/10.1111/ejss.13015
Barrena I, Menéndez S, Correa-Galeote D, Vega-Mas I, Bedmar EJ, González-Murua C, Estavillo JM (2017) Soil water content modulates the effect of the nitrification inhibitor 3,4-dimethylpyrazole phosphate (DMPP) on nitrifying and denitrifying bacteria. Geoderma 303:1–8. https://doi.org/10.1016/j.geoderma.2017.04.022
Bronson KF, Hunsaker DJ, Williams CF, Thorp KR, Rockholt SM, Del Grosso SJ, Venterea RT, Barnes EM (2018) Nitrogen management affects nitrous oxide emissions under varying cotton irrigation systems in the Desert Southwest, USA. J Environ Qual 47:70–78. https://doi.org/10.2134/jeq2017.10.0389
Bueno E, Mesa S, Bedmar EJ, Richardson DJ, Delgado MJ (2012) Bacterial adaptation of respiration from oxic to microoxic and anoxic conditions: redox control. Antioxid Redox Sign 16:819–852. https://doi.org/10.1089/ars.2011.4051
Butterbach-Bahl K, Baggs EM, Dannenmann M, Kiese R, Zechmeister-Boltenstern S (2013) Nitrous oxide emissions from soils: how well do we understand the processes and their controls? Philos T R Soc B 368:20130122. https://doi.org/10.1098/rstb.2013.0122
Cayuela ML, Aguilera E, Sanz-Cobena A, Adams DC, Abalos D, Barton L, Ryals R, Silver WL, Alfaro MA, Pappa VA, Smith P, Garnier J, Billen G, Bouwman L, Bondeau A, Lassaletta L (2017) Direct nitrous oxide emissions in Mediterranean climate cropping systems: emission factors based on a meta-analysis of available measurement data. Agric Ecosyst Environ 238:25–35. https://doi.org/10.1016/j.agee.2016.10.006
Corrochano-Monsalve M, González-Murua C, Bozal-Leorri A, Lezama L, Artetxe B (2021) Mechanism of action of nitrification inhibitors based on dimethylpyrazole: a matter of chelation. Sci Total Environ 752:141885. https://doi.org/10.1016/j.scitotenv.2020.141885
Crouzet O, Poly F, Bonnemoy F, Bru D, Batisson I, Bohatier J, Bohatier H, Mallet C (2016) Functional and structural responses of soil N-cycling microbial communities to the herbicide mesotrione: a dose-effect microcosm approach. Environ Sci Pollut R 23:4207–4217. https://doi.org/10.1007/s11356-015-4797-8
Dong J, Zhang J, Liu Y, Jing H (2022) How climate and soil properties affect the abundances of nitrogen-cycling genes in nitrogen-treated ecosystems: a meta-analysis. Plant Soil 477:389–404. https://doi.org/10.1007/s11104-022-05420-6
Fan D, He W, Smith WN, Drury CF, Jiang R, Grant BB, Shi Y, Song D, Chen Y, Wang X, He P, Zou G (2022) Global evaluation of inhibitor impacts on ammonia and nitrous oxide emissions from agricultural soils: a meta-analysis. Glob Change Biol 28:5121–5141. https://doi.org/10.1111/gcb.16294
Feng J, Li F, Deng A, Feng X, Fang F, Zhang W (2016) Integrated assessment of the impact of enhanced-efficiency nitrogen fertilizer on N2O emission and crop yield. Agric Ecosyst Environ 231:218–228. https://doi.org/10.1016/j.agee.2016.06.038
Flynn NE, Stewart CE, Comas LH, Del Grosso SJ, Schnarr C, Schipanski M, von Fischer JC, Stuchiner ER, Fonte SJ (2022) Deficit irrigation impacts on greenhouse gas emissions under drip-fertigated maize in the Great Plains of Colorado. J Environ Qual 51:877–889. https://doi.org/10.1002/jeq2.20353
Friedl J, Deltedesco E, Keiblinger KM, Gorfer M, De Rosa D, Scheer C, Grace PR, Rowlings DW (2022) Amplitude and frequency of wetting and drying cycles drive N2 and N2O emissions from a subtropical pasture. Biol Fert Soils 58:593–605. https://doi.org/10.1007/s00374-022-01646-9
Gilsanz C, Báez D, Misselbrook TH, Dhanoa MS, Cárdenas LM (2016) Development of emission factors and efficiency of two nitrification inhibitors, DCD and DMPP. Agric Ecosyst Environ 216:1–8. https://doi.org/10.1016/j.agee.2015.09.030
Guardia G, Cangani MT, Andreu G, Sanz-Cobena A, García-Marco S, Álvarez JM, Recio-Huetos J, Vallejo A (2017a) Effect of inhibitors and fertigation strategies on GHG emissions, NO fluxes and yield in irrigated maize. Field Crop Res 204:135–145. https://doi.org/10.1016/j.fcr.2017.01.009
Guardia G, Cangani MT, Sanz-Cobena A, Junior JL, Vallejo A (2017b) Management of pig manure to mitigate NO and yield-scaled N2O emissions in an irrigated Mediterranean crop. Agric Ecosyst Environ 238:55–66. https://doi.org/10.1016/j.agee.2016.09.022
Guardia G, Vallejo A, Cardenas LM, Dixon ER, García-Marco S (2018) Fate of 15N-labelled ammonium nitrate with or without the new nitrification inhibitor DMPSA in an irrigated maize crop. Soil Biol Biochem 116:193–202. https://doi.org/10.1016/j.soilbio.2017.10.013
Hallin S, Lindgren P-E (1999) PCR detection of genes encoding nitrite reductase in denitrifying bacteria. Appl Environ Microbiol 65:1652–1657. https://doi.org/10.1128/AEM.65.4.1652-1657.1999
Hallin S, Philippot L, Löffler FE, Sanford RA, Jones CM (2018) Genomics and ecology of novel N2O-reducing microorganisms. Trends Microbiol 26:43–55. https://doi.org/10.1016/j.tim.2017.07.003
Henry S, Bru D, Stres B, Hallet S, Philippot L (2006) Quantitative detection of the nosZ gene, encoding nitrous oxide reductase, and comparison of the abundances of 16S rRNA, narG, nirK, and nosZ genes in soils. Appl Environ Microbiol 72:5181–5189. https://doi.org/10.1128/AEM.00231-06
Huérfano X, Fuertes-Mendizábal T, Fernández-Diez K, Estavillo JM, González-Murua C, Menéndez S (2016) The new nitrification inhibitor 3, 4-dimethylpyrazole succinic (DMPSA) as an alternative to DMPP for reducing N2O emissions from wheat crops under humid Mediterranean conditions. Eur J Agron 80:78–87. https://doi.org/10.1016/j.eja.2016.07.001
Kallenbach CM, Rolston DE, Horwath WR (2010) Cover cropping affects soil N2O and CO2 emissions differently depending on type of irrigation. Agric Ecosyst Environ 137:251–260. https://doi.org/10.1016/j.agee.2010.02.010
Kassambara A, Mundt F (2020) factoextra: extract and visualize the results of multivariate data analyses. R package version 1:7 Available at: https://CRAN.R-project.org/package=factoextra
Koushki R, Warren J, Krzmarzick MJ (2023) Carbon footprint of agricultural groundwater pumping with energy demand and supply management analysis. Irrigation Sci 1-10. https://doi.org/10.1007/s00271-023-00885-4
Kuang W, Gao X, Tenuta M, Zeng F (2021) A global meta-analysis of nitrous oxide emission from drip-irrigated cropping system. Glob Change Biol 27:3244–3256. https://doi.org/10.1111/gcb.15636
Jones CM, Graf DR, Bru D, Philippot L, Hallin S (2013) The unaccounted yet abundant nitrous oxide-reducing microbial community: a potential nitrous oxide sink. ISME J 7:417–426. https://doi.org/10.1038/ismej.2012.125
Lamm FR (2016) Cotton, tomato, corn, and onion production with subsurface drip irrigation: A review. T. ASABE 59:263–278. https://doi.org/10.13031/trans.59.11231
Lamm FR, Bordovsky JP, Schwankl LJ, Grabow GL, Enciso-Medina J, Peters RT, Colaizzi PD, Trooien TP, Porter DO (2012) Subsurface drip irrigation: status of the technology in 2010. T ASABE 55:483–491. https://doi.org/10.13031/2013.41387
Lan T, Li M, He X, Deng O, Zhou W, Luo L, Chen G, Yuan S, Ling J, Zeng M, Gao X (2022) Effects of synthetic nitrification inhibitor (3, 4-dimethylpyrazole phosphate; DMPP) and biological nitrification inhibitor (methyl 3-(4-hydroxyphenyl) propionate; MHPP) on the gross N nitrification rate and ammonia oxidizers in two contrasting soils. Biol Fert Soils 58:333–344. https://doi.org/10.1007/s00374-022-01628-x
Lassaletta L, Sanz-Cobena A, Aguilera E, Quemada M, Billen G, Bondeau A, Cayuela ML, Cramer W, Eekhout JPC, Garnier J, Grizzetti B, Intrigiolo DS, Ruiz Ramos M, Romero E, Vallejo A, Gimeno BS (2021) Nitrogen dynamics in cropping systems under Mediterranean climate: a systemic analysis. Environ Res Lett 16:073002. https://doi.org/10.1088/1748-9326/ac002c
Lei J, Fan Q, Yu J, Ma Y, Yin J, Liu R (2022) A meta-analysis to examine whether nitrification inhibitors work through selectively inhibiting ammonia-oxidizing bacteria. Front Microbiol 13:962146. https://doi.org/10.3389/fmicb.2022.962146
Leitner S, Homyak PM, Blankinship JC, Eberwein J, Jenerette GD, Zechmeister-Boltenstern S, Schimel JP (2017) Linking NO and N2O emission pulses with the mobilization of mineral and organic N upon rewetting dry soils. Soil Biol Biochem 115:461–466. https://doi.org/10.1016/j.soilbio.2017.09.005
Maris SC, Teira-Esmatges MR, Arbonés A, Rufat J (2015) Effect of irrigation, nitrogen application, and a nitrification inhibitor on nitrous oxide, carbon dioxide and methane emissions from an olive (Olea europaea L.) orchard. Sci Total Environ 538:966–978. https://doi.org/10.1016/j.scitotenv.2015.08.040
Marsden KA, Marín-Martínez AJ, Vallejo A, Hill PW, Jones DL, Chadwick DR (2016) The mobility of nitrification inhibitors under simulated ruminant urine deposition and rainfall: a comparison between DCD and DMPP. Biol Fert Soils 52:491–503. https://doi.org/10.1007/s00374-016-1092-x
Montoya M, Juhanson J, Hallin S, García-Gutiérrez S, García-Marco S, Vallejo A, Recio J, Guardia G (2022) Nitrous oxide emissions and microbial communities during the transition to conservation agriculture using N-enhanced efficiency fertilisers in a semiarid climate. Soil Biol Biochem 170:108687. https://doi.org/10.1016/j.soilbio.2022.108687
Nair D, Abalos D, Philippot L, Bru D, Mateo-Marín N, Petersen SO (2021) Soil and temperature effects on nitrification and denitrification modified N2O mitigation by 3, 4-dimethylpyrazole phosphate. Soil Biol Biochem 157:108224. https://doi.org/10.1016/j.soilbio.2021.108224
Nishimura S, Sugito T, Nagatake A, Oka N (2021) Nitrous oxide emission reduced by coated nitrate fertilizer in a cool-temperate region. Nutr Cycl Agroecosys 119:275–289. https://doi.org/10.1007/s10705-020-10116-3
Ouyang Y, Evans SE, Friesen ML, Tiemann LK (2018) Effect of nitrogen fertilization on the abundance of nitrogen cycling genes in agricultural soils: a meta-analysis of field studies. Soil Biol Biochem 127:71–78. https://doi.org/10.1016/j.soilbio.2018.08.024
Parada AE, Needham DM, Fuhrman JA (2016) Every base matters: assessing small subunit rRNA primers for marine microbiomes with mock communities, time series and global field samples. Environ Microbiol 18:1403–1414. https://doi.org/10.1111/1462-2920.13023
Perez G, Aubert M, Decaëns T, Trap J, Chauvat M (2013) Home-field advantage: a matter of interaction between litter biochemistry and decomposer biota. Soil Biol Biochem 67:245–254. https://doi.org/10.1016/j.soilbio.2013.09.004
Pilegaard K (2013) Processes regulating nitric oxide emissions from soils. Philos T R Soc 368:20130126. https://doi.org/10.1098/rstb.2013.0126
Plauborg F, Motarjemi SK, Nagy D, Zhou Z (2022) Analysing potato response to subsurface drip irrigation and nitrogen fertigation regimes in a temperate environment using the Daisy model. Field Crop Res 276:108367. https://doi.org/10.1016/j.fcr.2021.108367
Quince C, Lanzen A, Davenport RJ, Turnbaugh PJ (2011) Removing noise from pyrosequenced amplicons. BMC Bioinformatics 12:38. https://doi.org/10.1186/1471-2105-12-38
R Core Team (2021) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria Available at: https://www.R-project.org/
Rahman N, Richards KG, Harty MA, Watson CJ, Carolan R, Krol D, Lanigan GJ, Forrestal PJ (2021) Differing effects of increasing calcium ammonium nitrate, urea and urea+ NBPT fertiliser rates on nitrous oxide emission factors at six temperate grassland sites in Ireland. Agric Ecosyst Environ 313:107382. https://doi.org/10.1016/j.agee.2021.107382
Rinaldo S, Cutruzzolà F (2007) Chapter 3 - Nitrite reductases in denitrification. In: Bothe H, Ferguson SJ, Newton WE (eds) Biology of the Nitrogen Cycle. Elsevier, Amsterdam, pp 37–55. https://doi.org/10.1016/B978-044452857-
Rotthauwe J-H, Witzel K-P, Liesack W (1997) The ammonia monooxygenase structural gene amoa as a functional marker: molecular fine-scale analysis of natural ammonia-oxidizing populations. Appl Environ Microbiol 63:4704–4712. https://doi.org/10.1128/aem.63.12.4704-4712.1997
Rúa MA, Antoninka A, Antunes PM, Chaudhary VB, Gehring C, Lamit LJ, Piculell BJ, Bever JD, Zabinski C, Meadow JF, Lajeunesse MJ, Milligan BG, Karst J, Hoeksema JD (2016) Home-field advantage? Evidence of local adaptation among plants, soil, and arbuscular mycorrhizal fungi through meta-analysis. BMC Evol Biol 16:122. https://doi.org/10.1186/s12862-016-0698-9
Singh K, Mishra SK, Singh M, Singh K, Brar AS (2022) Water footprint assessment of surface and subsurface drip fertigated cotton-wheat cropping system–a case study under semi-arid environments of Indian punjab. J Clean Prod 365:132735. https://doi.org/10.1016/j.jclepro.2022.132735
Soil Survey Staff (2014) Keys to soil taxonomy. Natural Resources Conservation Service, Washington
Sun X, Zhao J, Bei Q, Xia W, Zhou X, Zhao B, Zhang J, Jia Z (2022) Niche specialization and ecophysiological adaptation strategies of salt-tolerant nitrite oxidizers in soil. Biol Fert Soils 58:815–825. https://doi.org/10.1007/s00374-022-01663-8
Throbäck IN, Enwall K, Jarvis Å, Hallin S (2004) Reassessing PCR primers targeting nirS, nirK and nosZ genes for community surveys of denitrifying bacteria with DGGE. FEMS Microbiol Ecol 49:401–417. https://doi.org/10.1016/j.femsec.2004.04.011
Tian D, Zhang Y, Mu Y, Liu J, He K (2020) Effect of N fertilizer types on N2O and NO emissions under drip fertigation from an agricultural field in the North China Plain. Sci Total Environ 715:136903. https://doi.org/10.1016/j.scitotenv.2020.136903
Torralbo F, Menéndez S, Barrena I, Estavillo JM, Marino D, González-Murua C (2017) Dimethyl pyrazol-based nitrification inhibitors effect on nitrifying and denitrifying bacteria to mitigate N2O emission. Sci Rep 7:1–11. https://doi.org/10.1038/s41598-017-14225-y
Tourna M, Freitag TE, Nicol GW, Prosser JI (2008) Growth, activity and temperature responses of ammonia-oxidizing archaea and bacteria in soil microcosms. Environ Microbiol 10:1357–1364. https://doi.org/10.1111/j.1462-2920.2007.01563.x
Tufail MA, Naeem A, Arif MS, Farooq TH, Shahzad SM, Dar AA, Albasher G, Shakoor A (2022) Unraveling the efficacy of nitrification inhibitors (DCD and DMPP) in reducing nitrogen gases emissions across agroecosystems: a three-decade global data synthesis (1993–2021). Fuel 324:124725. https://doi.org/10.1016/j.fuel.2022.124725
Velthof GL, Rietra RPJJ (2018) Nitrous oxide emission from agricultural soils. Wageningen Environ Res Rep 2921:40–47
Vilarrasa-Nogué M, Teira-Esmatges MR, Pascual M, Villar JM, Rufat J (2020) Effect of N dose, fertilisation duration and application of a nitrification inhibitor on GHG emissions from a peach orchard. Sci Total Environ 699:134042. https://doi.org/10.1016/j.scitotenv.2019.134042
Wang H, Wang N, Quan H, Zhang F, Fan J, Feng H, Cheng M, Liao Z, Wang X, Xiang Y (2022a) Yield and water productivity of crops, vegetables and fruits under subsurface drip irrigation: a global meta-analysis. Agr Water Manage 269:107645. https://doi.org/10.1016/j.agwat.2022.107645
Wang J, Yao Z, Han J, Niu W, Li Y (2022b) Different pipe burial depths associated with subsurface drip irrigation significantly affected soil gas emissions. Ann Appl Biol 180:294–305. https://doi.org/10.1111/aab.12736
Wei Q, Li X, Xu J, Dai H, Li B, Xu J, Wei Q, Wang K (2022) Responses of soil N2O and CO2 emissions and their global warming potentials to irrigation water salinity. Atmosphere 13:1777. https://doi.org/10.3390/atmos13111777
Yang Y, Chen X, Liu L, Li T, Dou Y, Qiao J, Wang Y, An S, Chang SX (2022) Nitrogen fertilization weakens the linkage between soil carbon and microbial diversity: a global meta-analysis. Glob Change Biol 28:6446–6461. https://doi.org/10.1111/gcb.16361
Yangjin D, Wu X, Bai H, Gu J (2021) A meta-analysis of management practices for simultaneously mitigating N2O and NO emissions from agricultural soils. Soil Till Res 213:105142. https://doi.org/10.1016/j.still.2021.105142
Yao R, Li H, Yang J, Zhu W, Yin C, Wang X, Xie W, Zhang X (2022) Combined application of biochar and N fertilizer shifted nitrification rate and amoA gene abundance of ammonia-oxidizing microorganisms in salt-affected anthropogenic-alluvial soil. Appl Soil Ecol 171:104348. https://doi.org/10.1016/j.apsoil.2021.104348
Ye XH, Han B, Li W, Zhang XC, Zhang YL, Lin XG, Zou HT (2018) Effects of different irrigation methods on nitrous oxide emissions and ammonia oxidizers microorganisms in greenhouse tomato fields. Agr Water Manage 203:115–123. https://doi.org/10.1016/j.agwat.2018.03.012
You L, Ros GH, Chen Y, Yang X, Cui Z, Liu X, Jiang R, Zhang F, de Vries W (2022) Global meta-analysis of terrestrial nitrous oxide emissions and associated functional genes under nitrogen addition. Soil Biol Biochem 165:108523. https://doi.org/10.1016/j.soilbio.2021.108523
Yu Y, Jiao Y, Yang W, Song C, Zhang J, Liu Y (2022) Mechanisms underlying nitrous oxide emissions and nitrogen leaching from potato fields under drip irrigation and furrow irrigation. Agr Water Manage 260:107270. https://doi.org/10.1016/j.agwat.2021.107270
Zhang M, Wang W, Tang L, Heenan M, Xu Z (2018) Effects of nitrification inhibitor and herbicides on nitrification, nitrite and nitrate consumptions and nitrous oxide emission in an Australian sugarcane soil. Biol Fert Soils 54:697–706. https://doi.org/10.1007/s00374-018-1293-6
Zhang Y, Zhang F, Abalos D, Luo Y, Hui D, Hungate BA, García-Palacios P, Kuzyakov Y, Olesen JE, Jørgensen U, Chen J (2022) Stimulation of ammonia oxidizer and denitrifier abundances by nitrogen loading: poor predictability for increased soil N2O emission. Glob Change Biol 28:2158. https://doi.org/10.1111/gcb.16042
Zhao L, Li Q, Qian X, Chen H, Wang F, Yi Z (2022) Effects of the combined application of biochar-based fertilizer and urea on N2O emissions, nitrifier, and denitrifier communities in the acidic soil of pomelo orchards. J Soil Sediment 22:1–18. https://doi.org/10.1007/s11368-022-03273-1
Acknowledgements
Special thanks are extended to the assistants working with us at La Chimenea FIELD Station and (IMIDRA), particularly to Roberto Saiz and Alejandro Benito. We also thank the technicians at the Department of Chemistry and Food Technology of ETSIAAB (particularly Alba Monistrol and Rocío Rodríguez) for their collaboration in sampling and analyses. We are grateful to Drs. Eduardo Aguilera, Sonia García-Marco, and Alberto Sanz-Cobena for their technical support. We also acknowledge the doctors and technicians at the Department of Forest Mycology and Plant Pathology of the Swedish University of Agricultural Sciences (particularly Jaanis Juhanson) for their collaboration in the quantification of N-cycling genes. Special thanks are given to Dr. Mark Theobald from CIEMAT who revised the English in the re-submitted version. This work was done within the framework of the Moncloa Campus of International Excellence (UCM-UPM).
Funding
This research was funded by the RTI2018-096267-B-I00 project funded by the Ministerio de Ciencia, Innovación y Competitividad (MCIN)/Agencia Estatal de Investigación (AEI)/10.13039/501100011033/Fondo Europeo de Desarrollo Regional (FEDER) “Una manera de hacer Europa,” the Comunidad de Madrid (Spain), and Structural Funds 2014–2020 (ERDF and ESF) (AGRISOST-CM S2018/BAA-4330 project). Funding for this research was also provided by EuroChemAgro GmbH. S. García-Gutiérrez is the recipient of the PRE2019-087594 FPI grant funded by MCIN/AEI/10.13039/501100011033 and Fondo Social Europeo (FSE) “El FSE invierte en tu futuro.” M. Montoya is the recipient of the Margarita Salas grant of the Ministerio de Universidades and Universidad Politécnica de Madrid (RD 289/2021) supported by the European Union Next-GenerationEU.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 3287 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Guardia, G., García-Gutiérrez, S., Vallejo, A. et al. Nitrous oxide emissions and N-cycling gene abundances in a drip-fertigated (surface versus subsurface) maize crop with different N sources. Biol Fertil Soils 60, 375–391 (2024). https://doi.org/10.1007/s00374-023-01791-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-023-01791-9