Abstract
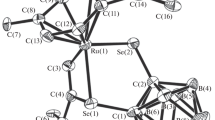
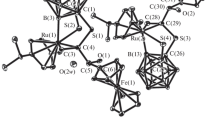
A novel complex (p-cymene)Ru(S2C2B10H9)(PhCH=CCH2OCH(Ph)C≡CH)·CH2Cl2 based on the ortho-carborane-1,2-dithiolate ligand and 1-phenyl-2-propyn-1-ol has been synthesized successfully and characterized by IR, NMR, MS, elemental analysis and single-crystal X-ray diffraction. The complex crystallizes in monoclinic system, space group P21/n with: a = 10.221(4) Å, b = 14.912(6) Å, c = 24.182(10) Å, α = 90.00°, β = 99.096(6)°, γ = 90.00°, C31H40B10Cl2ORuS2, Mr = 772.82, V = 3639(3) Å3, Dc = 1.410 g/cm3, Z = 4, F(000) = 1576, μ(MoKα) = 0.719 mm–1, R = 0.0600 and wR = 0.1561 for 5463 observed reflections (I > 2σ(I)). Structural analysis shows that there are the coordination of C=C bond, the formation of C–S bod, and metal-induced B–H activation with the formation of Ru–B bond.





Similar content being viewed by others
REFERENCES
A. Marfavi, P. Kavianpour, and L. M. Rendina. Carboranes in drug discovery, chemical biology and molecular imaging. Nat. Rev. Chem., 2022, 6(7), 486-504. https://doi.org/10.1038/s41570-022-00400-x
A. F. Armstrong and J. F. Valliant. The bioinorganic and medicinal chemistry of carboranes: From new drug discovery to molecular imaging and therapy. Dalton Trans., 2007, (38), 4240. https://doi.org/10.1039/b709843j
J. F. Valliant, K. J. Guenther, A. S. King, P. Morel, P. Schaffer, O. O. Sogbein, and K. A. Stephenson. The medicinal chemistry of carboranes. Coord. Chem. Rev., 2002, 232(1/2), 173-230. https://doi.org/10.1016/s0010-8545(02)00087-5
Z. Xie. Group 4 metallocenes incorporating constrained-geometry carboranyl ligands. Coord. Chem. Rev., 2006, 250(1/2), 259-272. https://doi.org/10.1016/j.ccr.2005.05.009
Z.-J. Yao and G.-X. Jin. Transition metal complexes based on carboranyl ligands containing N, P, and S donors: Synthesis, reactivity and applications. Coord. Chem. Rev., 2013, 257(17/18), 2522-2535. https://doi.org/10.1016/j.ccr.2013.02.004
X. Meng, F. Wang, and G.-X. Jin. Construction of M–M bonds in late transition metal complexes. Coord. Chem. Rev., 2010, 254(11/12), 1260-1272. https://doi.org/10.1016/j.ccr.2009.07.002
Z. Wang, H. Ye, Y. Li, Y. Li, and H. Yan. Unprecedented boron-functionalized carborane derivatives by facile and selective cobalt-induced B–H activation. J. Am. Chem. Soc., 2013, 135(30), 11289-11298. https://doi.org/10.1021/ja4047075
M. Herberhold, H. Yan, W. Milius, and B. Wrackmeyer. Metal-induced B–H activation: Addition of acetylene, propyne, or 3-methoxypropyne to Rh(Cp*), Ir(Cp*), Ru(p-cymene), and Os(p-cymene) half-sandwich complexes containing a chelating 1,2-dicarba-closo-dodecaborane-1,2-dichalcogenolato ligand. Chem. - Eur. J., 2002, 8(2), 388-395. https://doi.org/10.1002/1521-3765(20020118)8:2<388::aid-chem388>3.0.co;2-u
M. A. Esteruelas, A. M. López, and M. Oliván. Osmium–carbon double bonds: Formation and reactions. Coord. Chem. Rev., 2007, 251(5/6), 795-840. https://doi.org/10.1016/j.ccr.2006.07.008
R. Castarlenas, C. Vovard, C. Fischmeister, P.H. Dixneuf. Allenylidene-to-indenylidene rearrangement in arene–ruthenium complexes: A key step to highly active catalysts for olefin metathesis reactions. J. Am. Chem. Soc., 2006, 128(12), 4079-4089. https://doi.org/10.1021/ja0579762
B. Liu, H. Wang, H. Xie, B. Zeng, J. Chen, J. Tao, T. Bin Wen, Z. Cao, and H. Xia. Osmapyridine and osmapyridinium from a formal [4+2] cycloaddition reaction. Angew. Chem., Int. Ed., 2009, 48(30), 5430-5434. https://doi.org/10.1002/anie.200900998
L. Gong, Y. Lin, T. Bin Wen, and H. Xia. Synthesis of coordinated η2-α,β-unsaturated ketone osmacycles from an osmium-coordinated alkyne alcohol complex. Organometallics, 2009, 28(4), 1101-1111. https://doi.org/10.1021/om800907v
J. R. Hu, W. H. Chen, D. K. Nie, Y. H. Wang, and H. D. Ye. Reactivity of dinuclear ruthenium complex containing two 1,2-dicarba-closo-dodecaborane-1,2-dithiolate ligands toward HC≡CCH(OH)(CH3)2. Russ. J. Coord. Chem., 2016, 42(12), 783-788. https://doi.org/10.1134/s1070328416120034
J. R. Hu and J. H. Wang. Synthesis and structural characterization of the ruthenium complexes based on ortho-carborane-1,2-diselenolate ligand and terminal alkynone. Russ. J. Inorg. Chem., 2022, 67(11), 1739-1744. https://doi.org/10.1134/s0036023622600654
J. R. Hu, J. H. Wang, K. G. Jin, and C. P. Zhu. Synthesis and structural characterization of the ruthenium complex based on 1,2-dicarba-closo-dodecaborane-1,2-dithiolate ligand and FcCH(OH)C≡CH. Russ. J. Coord. Chem., 2020, 46(6), 437-442. https://doi.org/10.1134/s1070328420060019
M. A. Bennett, T.-N. Huang, T. W. Matheson, A. K. Smith, S. Ittel, and W. Nickerson. (η6-Hexamethylbenzene)ruthenium complexes. In: Inorganic Syntheses, Vol. 21 / Ed. J.P. Fackler Jr. Inorganic Syntheses, Inc., 1982, 21, 74-78. https://doi.org/10.1002/9780470132524.ch16
M. Herberhold, H. Yan, W. Milius, and B. Wrackmeyer. Metal-induced B–H activation. Addition of phenylacetylene to Cp*Rh-, Cp*Ir-, (p-cymene)Ru- and (p-cymene)Os halfsandwich complexes containing a chelating 1,2-dicarba-closo-dodecaborane-1,2-dichalcogenolate ligand. J. Organomet. Chem., 2000, 604(2), 170-177. https://doi.org/10.1016/s0022-328x(00)00221-7
SMART (Version 5.0), SAINT (Version 6), SHELXTL (Version 6.1) and SADABS (Version 2.03). Madison, Wisconsin, USA: Bruker AXS Inc., 2000.
J. R. Hu, W. J. Zhang, L. H. Liu, and H. N. Peng. Synthesis, structure, and mechanism of half-sandwich ruthenium complexes based on ortho-carborane-1,2-dithiolate ligand and alkyne alcohol. Russ. J. Coord. Chem., 2014, 40(12), 954-958. https://doi.org/10.1134/s1070328414120057
J. Hu, W. Dong, and J. Wang. Synthesis, structure, and mechanism of the isomers of ruthenium complexes based on HC≡CC(OH)(CH3)2 and ortho-carborane dithiolate ligands. Inorg. Chem. Commun., 2022, 143, 109722. https://doi.org/10.1016/j.inoche.2022.109722
J. R. Hu and J. H. Wang. Reactivity of 16-electron (p-cymene)Ru half-sandwich complex containing a chelating 1,2-dicarba-closo-dodecaborane-1,2-dithiolate ligand with 1-ethynyl-1-cyclohexanol. J. Struct. Chem., 2022, 63(10), 1551-1557. https://doi.org/10.1134/s0022476622100018
J. Hu, S. Sun, K. He, and Y. Wang. Synthesis and characterization of multinuclear ruthenium clusters assembled by terminal alkyne alcohols and ortho-carborane diselenolate ligands. J. Mol. Struct., 2022, 1263, 133198. https://doi.org/10.1016/j.molstruc.2022.133198
D. Wu, Y. Li, L. Han, Y. Li, and H. Yan. Mixed-valent diruthenium half-sandwich complexes containing two chelating 1,2-dicarba-closo-dodecaborane-1,2-dithiolate ligands: Reactivity towards phenylacetylene, 1,4-diethynylbenzene, and ethynylferrocene. Inorg. Chem., 2008, 47(14), 6524-6531. https://doi.org/10.1021/ic800619z
E. Bustelo, M. Jiménez-Tenorio, M. C. Puerta, and P. Valerga. γ-Substituted vinylidene, chroman-2-ylidene, and hexahydrochromen-2-ylidene from ruthenium allenylidene/alkenylcarbyne complexes. Organometallics, 2007, 26(17), 4300-4309. https://doi.org/10.1021/om700431n
K. Y. Ghebreyessus and J. H. Nelson. Reactions of [(η6-Me6C6)Ru(Ph2PCH–CH2)Cl2] with 1-ethynylcycloalkanols leading to methoxy-alkenyl carbenes via vinylvinylidene and/or allenylidene intermediates. Inorg. Chem. Commun., 2003, 6(8), 1044-1047. https://doi.org/10.1016/s1387-7003(03)00177-1
Y. Quan and Z. Xie. Controlled functionalization of o-carborane via transition metal catalyzed B–H activation. Chem. Soc. Rev., 2019, 48(13), 3660-3673. https://doi.org/10.1039/c9cs00169g
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors of this work declare that they have no conflicts of interests.
Additional information
Text © The Author(s), 2023, published in Zhurnal Strukturnoi Khimii, 2023, Vol. 64, No. 12, 119203.https://doi.org/10.26902/JSC_id119203
Publisher’s Note. Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hu, J.R., Zhao, S.Q., Wang, J.H. et al. Synthesis, Structure, and Mechanism of Half-Sandwich Ruthenium Complex Containing OrthoCarborane-1,2-Dithiolate Ligand and 1-Phenyl-2-Propyn-1-ol. J Struct Chem 64, 2343–2350 (2023). https://doi.org/10.1134/S0022476623120053
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476623120053