Abstract
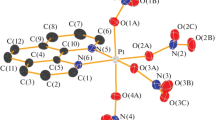
Two novel platinum(IV) nitrate complexes, Cs2[Pt(NO3)6] (1) and Cs2[Pt2(μ-OH)2(NO3)8] (2), are prepared by adding cesium nitrate to a solution of platinum(IV) hydroxide in nitric acid. It is established that the main reaction product is the Cs2[Pt(NO3)6] mononuclear complex, while Cs2[Pt2(μ-OH)2(NO3)8] is formed only after the removal of the first portion of the precipitate and subsequent long-term storage of the mother liquor. The structures of the salts are studied by XRD. Crystal data for Cs2[Pt(NO3)6] (1): a = 9.7346(3) Å, b = 11.5072(3) Å, c = 13.6813(2) Å, space group Pbcn, Z = 4. Crystal data for Cs2[Pt2(μ-OH)2(NO3)8] (2): a = 7.0889(2) Å, b = 14.2216(6) Å, c = 10.7931(4) Å, β = 91.776(1)°, space group P21/n, Z = 2. It is shown that NO3 ligands in (1) occupy atypical positions. The geometry of the complex anion in (2) is similar to the one described for (Me4N)2[Pt2(μ-OH)2(NO3)8]. Powdered samples of (1) and those of a mixture of (1) and (2) are characterized by powder XRD and IR spectroscopy. The obtained compounds are shown to be stable upon long-term storage in air.






Similar content being viewed by others
REFERENCES
D. Dou, D.-J. Liu, W. B. Williamson, K. C. Kharas, and H. J. Robota. Structure and chemical properties of Pt nitrate and application in three-way automotive emission catalysts. Appl. Catal., B, 2001, 30(1/2), 11-24. https://doi.org/10.1016/s0926-3373(00)00223-x
D. Vasilchenko, S. Tkachev, I. Baidina, and S. Korenev. Speciation of platinum(IV) in nitric acid solutions. Inorg. Chem., 2013, 52(18), 10532-10541. https://doi.org/10.1021/ic401499j
T. Suzuki, U. Otsubo, T. Ogata, H. Shiwaku, T. Kobayashi, T. Yaita, M. Matsuoka, N. Murayama, and H. Narita. Speciation and separation of platinum(IV) polynuclear complexes in concentrated nitric acid solutions. Dalton Trans., 2021, 50(33), 11390-11397. https://doi.org/10.1039/d1dt01392k
D. Vasilchenko, P. Topchiyan, S. Berdyugin, E. Filatov, S. Tkachev, I. Baidina, V. Komarov, E. Slavinskaya, A. Stadnichenko, and E. Gerasimov. Tetraalkylammonium salts of platinum nitrato complexes: Isolation, structure, and relevance to the preparation of PtOx/CeO2 catalysts for low-temperature CO oxidation. Inorg. Chem., 2019, 58(9), 6075-6087. https://doi.org/10.1021/acs.inorgchem.9b00370
D. B. Vasilchenko, P. A. Topchiyan, S. V. Tkachev, I. A. Baidina, I. V. Korolkov, E. Y. Filatov, and D. G. Sheven. Structure and properties of (C4N2H12)[Pt(NO3)6] salt. J. Struct. Chem., 2019, 60(8), 1327-1334. https://doi.org/10.1134/s0022476619080122
P. A. Topchiyan, I. A. Baidina, I. V. Korolkov, S. V. Tkachev, and D. B. Vasilchenko. Platinum(IV) nitrato complexes with 1,10-phenanthroline. Russ. J. Inorg. Chem., 2020, 65(10), 1552-1557. https://doi.org/10.1134/s0036023620100216
M. S. Wickleder, F. Gerlach, S. Gagelmann, J. Bruns, M. Fenske, and K. Al-Shamery. Thermolabile noble metal precursors: (NO)[Au(NO3)4], (NO)2[Pd(NO3)4], and (NO)2[Pt(NO3)6]. Angew. Chem., Int. Ed., 2012, 51(9), 2199-2203. https://doi.org/10.1002/anie.201106107
D. Vasilchenko, P. Topchiyan, I. Baidina, I. Korolkov, E. Filatov, V. Zvereva, P. Plyusnin, E. Slavinskaya, and E. Gerasimov. Double complex salts containing [Pt(NO3)6]2- anion and Rh(III) complex cations: Synthesis, structure and utilisation for preparing (Rh–Pt)/CeO2 catalysts. J. Mol. Struct., 2020, 1211, 128108. https://doi.org/10.1016/j.molstruc.2020.128108
D. Vasilchenko, S. Berdugin, S. Tkachev, I. Baidina, G. Romanenko, O. Gerasko, and S. Korenev. Polynuclear hydroxido-bridged complexes of platinum(IV) with terminal nitrato ligands. Inorg. Chem., 2015, 54(10), 4644-4651. https://doi.org/10.1021/ic502896s
P. A. Topchiyan, D. B. Vasilchenko, I. A. Baidina, and I. V. Korolkov. Adducts of platinum(IV) nitrate complexes with 15-crown-5 ether. J. Struct. Chem., 2020, 61(9), 1422-1431. https://doi.org/10.1134/s0022476620090097
A. B. Venediktov, S. V. Korenev, D. B. Vasil′chenko, A. V. Zadesenets, E. Y. Filatov, S. N. Mamonov, L. V. Ivanova, N. G. Prudnikova, and E. Y. Semitut. On preparation of platinum(IV) nitrate solutions from hexahydroxoplatinates(IV). Russ. J. Appl. Chem., 2012, 85(7), 995-1002. https://doi.org/10.1134/s1070427212070014
SADABS. Madison, Wisconsin, USA: Bruker AXS, 2001.
G. M. Sheldrick. SHELXT - Integrated space-group and crystal-structure determination. Acta Crystallogr., Sect. A: Found. Adv., 2015, 71(1), 3-8. https://doi.org/10.1107/s2053273314026370
G. M. Sheldrick. Crystal structure refinement with SHELXL. Acta Crystallogr., Sect. C: Struct. Chem., 2015, 71(1), 3-8. https://doi.org/10.1107/s2053229614024218
P. Topchiyan, A. Komarovskikh, I. Yushina, V. Komarov, D. Vasilchenko, and S. Korenev. Photoinduced transformation of (Bu4N)2[Pt(NO3)6] complex in the solid state. J. Photochem. Photobiol., A, 2022, 427, 113788. https://doi.org/10.1016/j.jphotochem.2022.113788
Funding
The synthesis and characterization of platinum compounds was funded by the Russian Science Foundation (project No. 21-73-10038). The crystal structure determination was funded by the Ministry of Science and Higher Education of the Russian Federation, project No. 121031700315-2.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors of this work declare that they have no conflicts of interests.
Additional information
Russian Text © The Author(s), 2023, published in Zhurnal Strukturnoi Khimii, 2023, Vol. 64, No. 12, 119510.https://doi.org/10.26902/JSC_id119510
Publisher’s Note. Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Topchiyan, P.A., Kuratieva, N.V. & Vasilchenko, D.B. Nitrato Complexes of Platinum(IV) with Cesium Cations. J Struct Chem 64, 2378–2385 (2023). https://doi.org/10.1134/S0022476623120090
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476623120090