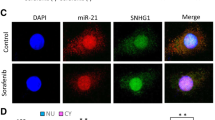
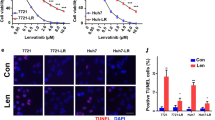
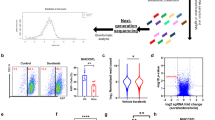
Abstract
Acquired drug resistance is a main reason for limiting the application of sorafenib in HCC treatment. This study aimed to explore the role and mechanisms of a novel long non-coding RNA (lncRNA), lnc-TSI, in sorafenib resistance of HCC. The interaction between lnc-TSI and miR-4726-5p, and miR-4726-5p and KCNMA1 were predicted using bioinformatic tools. Expression of the molecules in the lnc-TSI/miR-4726-5p/KCNMA1 axis in clinical samples and cell lines, as well as the sorafenib resistant HCC cell lines, was determined using qRT-PCR or western blotting. Expressions of lnc-TSI, miR-4726-5p, and KCNMA1 were manipulated in HepG2 and Huh7 cells through plasmid transfection or lentivirus infection. The CCK-8, flow cytometry, and Tunel assays were employed to determine the role of this axis on sorafenib resistance of HCC. A xenograft model was established using sorafenib-resistant HepG2 and Huh7 cells followed by in vivo sorafenib treatments to confirm the in vitro findings. Lnc-TSI and KCNMA1 expressions were significantly downregulated in HCC clinical samples and cell lines, especially in sorafenib resistance ones, while mi-4726-5p presented a reversed expression pattern. Lnc-TSI interacted with miR-4726-5p, and Lnc-TSI acts as a ceRNA via sponging miR-4726-5p in HCC cells. Overexpression of lnc-TSI and KCNMA1 promoted apoptosis and decreased cell viability of sorafenib-treated HCC cells, thus alleviated sorafenib resistance. miR-4726-5p mimic reversed the KCNMA1-mediated sorafenib sensitivity-promoting effect, while additional overexpression of lnc-TSI reversed the effect of miR-4726-5p. In vivo analysis also showed that overexpression of ln-TSI diminished sorafenib resistance in mice inoculated with sorafenib-resistant HCC cells via increasing KCNMA1 expression and decreasing miR-4726-5p expression. The lnc-TSI/miR-4726-5p/KCNMA1 axis plays a critical role in regulating the resistance of HCC to sorafenib, and might serve as a therapeutic target to manage sorafenib resistance of HCC in clinic.






Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Abdelgalil AA, Alkahtani HM, Al-Jenoobi FI (2019) Chapter four - sorafenibProfiles of drug substances, Excipients and related methodology academic press, pp. 239–266
Amin N, McGrath A, Chen Y-PP (2019) Evaluation of deep learning in non-coding RNA classification. Nat Mach Intell 1:246–256. https://doi.org/10.1038/s42256-019-0051-2
Bai Y, Long J, Liu Z, Lin J, Huang H, Wang D, Yang X, Miao F, Mao Y, Sang X et al (2019) Comprehensive analysis of a ceRNA network reveals potential prognostic cytoplasmic lncRNAs involved in HCC progression. J Cell Physiol 234:18837–18848. https://doi.org/10.1002/jcp.28522
Blankenburg M, Elhamamy M, Zhang D, Corbin A, Jin G, Harris J, Knobloch G (2022) Evaluation of the health economic impact of initial diagnostic modality selection in patients suspected of having HCC in China and the USA. J Med Econ. https://doi.org/10.1080/13696998.2022.2110353
Cheng Z, Lei Z, Yang P, Si A, Xiang D, Zhou J, Hüser N (2019) Long non-coding RNA THOR promotes Liver cancer stem cells expansion via β-catenin pathway. Gene 684:95–103. https://doi.org/10.1016/j.gene.2018.10.051
Conte F, Fiscon G, Sibilio P, Licursi V, Paci P (2021) An overview of the computational models dealing with the regulatory ceRNA mechanism and ceRNA deregulation in cancer. In: Poliseno, L. (eds) Pseudogenes. Methods in Molecular Biology, vol 2324. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1503-4_10
Dhanoa JK, Sethi RS, Verma R, Arora JS, Mukhopadhyay CS (2018) Long non-coding RNA: its evolutionary relics and biological implications in mammals: a review. J Anim Sci Technol 60:25. https://doi.org/10.1186/s40781-018-0183-7
Ding B, Lou W, Xu L, Fan W (2018) Non-coding RNA in drug resistance of hepatocellular carcinoma. Biosci Rep. https://doi.org/10.1042/BSR20180915
Fan Y, Xia X, Zhu Y, Diao W, Zhu X, Gao Z, Chen X (2018) Circular RNA expression profile in laryngeal squamous cell carcinoma revealed by microarray. Cell Physiol Biochem 50:342–352
Feng R-M, Zong Y-N, Cao S-M, Xu R-H (2019) Current cancer situation in China: good or bad news from the 2018 global cancer statistics? Cancer Commun 39:22. https://doi.org/10.1186/s40880-019-0368-6
Fornari F, Giovannini C, Piscaglia F, Gramantieri L (2021) Elucidating the molecular basis of sorafenib resistance in HCC: current findings and future directions. J Hepatocell Carcinoma 8:741–757. https://doi.org/10.2147/jhc.s285726
He M, Li Q, Zou R, Shen J, Fang W, Tan G, Zhou Y, Wu X, Xu L, Wei W et al (2019) Sorafenib plus hepatic arterial infusion of oxaliplatin, fluorouracil, and leucovorin vs sorafenib alone for hepatocellular carcinoma with portal vein invasion: a randomized clinical trial. JAMA Oncol 5:953–960. https://doi.org/10.1001/jamaoncol.2019.0250
Hu B, Xiao L, Wang C, Liu C, Zhang Y, Ding B, Gao D, Lu Y, Xu H (2022) Circ_0022382 ameliorated intervertebral disc degeneration by regulating TGF-β3 expression through sponge adsorption of miR-4726-5p. Bone 154:116185
Ji X, Zhang J, Liu L, Lin Z, Pi L, Lin Z, Tian N, Lin X, Chen S, Yu X et al (2019) Association of tagSNPs at lncRNA MALAT-1 with HCC susceptibility in a Southern Chinese population. Sci Rep 9:10895. https://doi.org/10.1038/s41598-019-47165-w
Kong F-H, Ye Q-F, Miao X-Y, Liu X, Huang S-Q, Xiong L, Wen Y, Zhang Z-J (2021) Current status of sorafenib nanoparticle delivery systems in the treatment of hepatocellular carcinoma. Theranostics 11:5464
Lai Y, Feng B, Abudoureyimu M, Zhi Y, Zhou H, Wang T, Chu X, Chen P, Wang R (2019) Non-coding RNAs: emerging regulators of sorafenib resistance in hepatocellular carcinoma. Front Oncol. https://doi.org/10.3389/fonc.2019.01156
Li C, Li R, Zhang W (2018) Progress in non-invasive detection of liver fibrosis. Cancer Biol Med 15:124
Li J, Zhao J, Wang H, Li X, Liu A, Qin Q, Li B (2018) MicroRNA-140-3p enhances the sensitivity of hepatocellular carcinoma cells to sorafenib by targeting pregnenolone X receptor. Onco Targets Ther 11:5885–5894. https://doi.org/10.2147/ott.s179509
Liu K, Gao L, Ma X, Huang J-J, Chen J, Zeng L, Ashby CR, Zou C, Chen Z-S (2020) Long non-coding RNAs regulate drug resistance in cancer. Mol Cancer 19:1–13
Ma G, Liu H, Hua Q, Wang M, Du M, Lin Y, Ge Y, Gong W, Zhao Q, Qiang F et al (2017) KCNMA1 cooperating with PTK2 is a novel tumor suppressor in gastric cancer and is associated with disease outcome. Mol Cancer 16:46. https://doi.org/10.1186/s12943-017-0613-z
Ming-hui C, Xiao-yan H, Hao-jie Y, Yang-fan Z, Zi-cong T (2021) Long non-coding RNA TSI inhibited the metastasis induced by TGF-1 in breast cencer cell MCF-7 and BT474. Lingnan Mod Clin Surg 21:171–176. https://doi.org/10.3969/j.issn.1009-976X.2021.02.008
Negri F, Porta C (2021) Donafenib in Chinese patients with advanced hepatocellular carcinoma (HCC): really a new standard of care, or should we change paradigm for drug development in HCC? Oncol Rev 15:564. https://doi.org/10.4081/oncol.2021.564
Oeggerli M, Tian Y, Ruiz C, Wijker B, Sauter G, Obermann E, Güth U, Zlobec I, Sausbier M, Kunzelmann K et al (2012) Role of KCNMA1 in breast cancer. PLoS ONE 7:e41664. https://doi.org/10.1371/journal.pone.0041664
Ouadid-Ahidouch H, Ahidouch A (2008) K+ channel expression in human breast cancer cells: involvement in cell cycle regulation and carcinogenesis. J Membr Biol 221:1–6
Ouadid-Ahidouch H, Roudbaraki M, Ahidouch A, Delcourt P, Prevarskaya N (2004) Cell-cycle-dependent expression of the large Ca2+-activated K+ channels in breast cancer cells. Biochem Biophys Res Commun 316:244–251
Panni S, Lovering RC, Porras P, Orchard S (2020) Non-coding RNA regulatory networks. biochimica et biophysica acta (BBA). Gene Regul Mech 1863:194417. https://doi.org/10.1016/j.bbagrm.2019.194417
Salmena L, Poliseno L, Tay Y, Kats L, Pandolfi PP (2011) A ceRNA hypothesis: the rosetta stone of a hidden RNA language? Cell 146:353–358
Samuel P, Pink RC, Caley DP, Currie JMS, Brooks SA, Carter DRF (2016) Over-expression of miR-31 or loss of KCNMA1 leads to increased cisplatin resistance in ovarian cancer cells. Tumor Biol 37:2565–2573. https://doi.org/10.1007/s13277-015-4081-z
Seal RL, Chen L-L, Griffiths-Jones S, Lowe TM, Mathews MB, O’Reilly D, Pierce AJ, Stadler PF, Ulitsky I, Wolin SL et al (2020) A guide to naming human non-coding RNA genes. EMBO J 39:e103777. https://doi.org/10.15252/embj.2019103777
Sui Z, Xue H, Jing F, Leng P (2001) Sorafenib plus Capecitabine for Patients with Advanced Hepatocellular Carcinoma. China Pharmacy 12:2001. https://pesquisa.bvsalud.org/portal/resource/pt/wpr-530379
Xia C, Dong X, Li H, Cao M, Sun D, He S, Yang F, Yan X, Zhang S, Li N et al (2022) Cancer statistics in China and United States, 2022: profiles, trends, and determinants. Chin Med J 135:584–590. https://doi.org/10.1097/CM9.0000000000002108
Xia S, Pan Y, Liang Y, Xu J, Cai X (2020) The microenvironmental and metabolic aspects of sorafenib resistance in hepatocellular carcinoma. EBioMedicine 51:102610. https://doi.org/10.1016/j.ebiom.2019.102610
Yadi W, Shurui C, Tong Z, Suxian C, Qing T, Dongning H (2020) Bioinformatic analysis of peripheral blood miRNA of breast cancer patients in relation with anthracycline cardiotoxicity. BMC Cardiovasc Disord 20:1–10
Zeng R, Wang C, Wang W, Wang S (2021) Long non-coding RNA DUXAP9 promotes hepatocellular carcinoma cell stemness via directly interacting with sox9. Environ Toxicol 36:1793–1801. https://doi.org/10.1002/tox.23300
Acknowledgements
We are very grateful for the equipment and technical support provided by The Second Affiliated Hospital of Xi’an Jiaotong University.
Funding
This work was supported by the Basic and Applied Research Promotion Engineering/Medical Research Project of Xi’an Science and Technology Plan (No. 21YXYJ0118), the General Project in the Field of Social Development of Shaanxi Provincial Department of Science and Technology: the research on the mechanism and potential clinical significance of new long chain non coding RNA AC006262.5 in promoting hepatocellular carcinogenesis (NO. 2020SF-065) and the General Project of Key Research and Development Projects of Shaanxi Province: the experimental study of EGCG against liver fibrosis through miR-1207-5p (NO. 2021SF-224).
Author information
Authors and Affiliations
Contributions
FC, JJ, and GZ: designed the experiments; FC and JJ: wrote the manuscript; DL, LD and HL: performed the experiments; YS, YZ, JW and YQ: analyzed the data, GZ: revised the manuscript. All the authors have read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Research involving human and animal participants
All animal experiments were approved by the Ethics Committee of the Second Affiliated Hospital of Xi’an Jiaotong University.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, F., Jiang, J., Liu, D. et al. The lncRNA lnc-TSI antagonizes sorafenib resistance in hepatocellular carcinoma via downregulating miR-4726-5p expression and upregulating KCNMA1 expression. J Mol Histol 55, 83–96 (2024). https://doi.org/10.1007/s10735-023-10173-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-023-10173-2