Abstract
Introduction
Pediatric prucalopride studies for treatment of gastrointestinal (GI) disorders have reported mixed results. We aimed to assess the safety and effectiveness of prucalopride in functional constipation (FC) with and without upper GI symptoms.
Methods
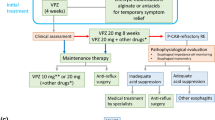
Retrospective data on patients with FC receiving combined prucalopride and conventional therapy was compared with those receiving conventional therapy alone within 12 months. Thirty patients on combined therapy and those on conventional therapy were each matched on the basis of age, gender, race, and presence of fecal soiling. Response (complete, partial, or no resolution) was compared. Similarly, response to concurrent functional upper GI symptoms (postprandial pain, bloating, weight loss, vomiting, early satiety, or nausea) and dysphagia, as well as adverse effects, were evaluated in the combined group.
Results
Mean age of 57 cases was 14.7 ± 4.9 years and 68% were female. Comorbidities included functional upper GI (UGI) symptoms (84%), dysphagia (12%), mood disorders (49%), and hypermobility spectrum disorder (37%). Unmatched cases reported 63% improvement to FC; response did not differ between the matched cohorts (70% versus 76.6%, p = 0.84). Cases showed a 56% improvement in functional UGI symptoms and 100% in dysphagia. Adverse effects were reported in 30%, abdominal cramps being most common. Four (7%) patients with a known mood disorder reported worsened mood, of which two endorsed suicidal ideation.
Conclusion
Prucalopride efficaciously treated concurrent UGI symptoms and dysphagia in constipated pediatric patients and was overall well tolerated. Preexisting mood disorders seemed to worsen in a small subset of cases.

Similar content being viewed by others
References
Mugie SM, Benninga MA, Di Lorenzo C. Epidemiology of constipation in children and adults: a systematic review. Best Pract Res Clin Gastroenterol. 2011;25:3–18. https://doi.org/10.1016/j.bpg.2010.12.010.
Robin SG, Keller C, Zwiener R, Hyman PE, Nurko S, Saps M, et al. Prevalence of pediatric functional gastrointestinal disorders utilizing the Rome IV criteria. J Pediatr. 2018;195:134–9. https://doi.org/10.1016/j.jpeds.2017.12.012.
Tabbers MM, DiLorenzo C, Berger MY, Faure C, Langendam MW, Nurko S, et al. Evaluation and treatment of functional constipation in infants and children: evidence-based recommendations from ESPGHAN and NASPGHAN. J Pediatr Gastroenterol Nutr. 2014;58:258–74. https://doi.org/10.1097/MPG.0000000000000266.
van Mill MJ, Koppen IJN, Benninga MA. Controversies in the management of functional constipation in children. Curr Gastroenterol Rep. 2019;21:23. https://doi.org/10.1007/s11894-019-0690-9.
Bongers MEJ, van Wijk MP, Reitsma JB, Benninga MA. Long-term prognosis for childhood constipation: clinical outcomes in adulthood. Pediatrics. 2010;126:e156–62. https://doi.org/10.1542/peds.2009-1009.
Baran M, Cagan Appak Y, Karakoyun M, Yalcinkaya S, Eliacik K, Dundar BN. The overlap of gastroesophageal reflux disease and functional constipation in children: the efficacy of constipation treatment. Eur J Gastroenterol Hepatol. 2017;29:1264–8. https://doi.org/10.1097/MEG.0000000000000979.
Hirsch S, Nurko S, Mitchell P, Rosen R. Prucalopride for treatment of upper gastrointestinal symptoms in children. Paediatr Drugs. 2022;24:73–81. https://doi.org/10.1007/s40272-021-00489-5.
Winter HS, Di Lorenzo C, Benninga MA, Gilger MA, Kearns GL, Hyman PE, et al. Oral prucalopride in children with functional constipation. J Pediatr Gastroenterol Nutr. 2013;57:197–203. https://doi.org/10.1097/MPG.0b013e318292f9ea.
Singh R, Zogg H, Ghoshal UC, Ro S. Current treatment options and therapeutic insights for gastrointestinal dysmotility and functional gastrointestinal disorders. Front Pharmacol. 2022;13: 808195. https://doi.org/10.3389/fphar.2022.808195.
Camilleri M, Atieh J. New Developments in prokinetic therapy for gastric motility disorders. Front Pharmacol. 2021;12: 711500. https://doi.org/10.3389/fphar.2021.711500.
Mutalib M, Kammermeier J, Vora R, Borrelli O. Prucalopride in intestinal pseudo obstruction, paediatric experience and systematic review. Acta Gastro Enterol Belg. 2021;84:429–34. https://doi.org/10.51821/84.3.002.
Drug Approval Package: Motegrity (prucalopride) n.d. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2018/210166Orig1s000TOC.cfm. Accessed 21 Feb 2022.
Camilleri M, Van Outryve MJ, Beyens G, Kerstens R, Robinson P, Vandeplassche L. Clinical trial: the efficacy of open-label prucalopride treatment in patients with chronic constipation—follow-up of patients from the pivotal studies. Aliment Pharmacol Ther. 2010;32:1113–23. https://doi.org/10.1111/j.1365-2036.2010.04455.x.
US Food and Drug Administration. FDA Talk Paper 00-14: Janssen Pharmaceutic stops marketing cisapride in the US. 2000. www.fda.gov. Accessed on 21 Feb 2022.
Tameez Ud Din A, Khan AH, Bajwa H, Maqsood MH, Malik MN. Clinical efficacy and safety profile of prucalopride in chronic idiopathic constipation. Cureus. 2019;11:e4382. https://doi.org/10.7759/cureus.4382.
Carbone F, Van den Houte K, Clevers E, Andrews CN, Papathanasopoulos A, Holvoet L, et al. Prucalopride in gastroparesis: a randomized placebo-controlled crossover study. Am J Gastroenterol. 2019;114:1265–74. https://doi.org/10.14309/ajg.0000000000000304.
Mugie SM, Korczowski B, Bodi P, Green A, Kerstens R, Ausma J, et al. Prucalopride is no more effective than placebo for children with functional constipation. Gastroenterology. 2014;147:1285-1295.e1. https://doi.org/10.1053/j.gastro.2014.09.005.
Ho D, Imai K, King G, Stuart EA. MatchIt: nonparametric preprocessing for parametric causal inference. J Stat Softw. 2011;42:1–28. https://doi.org/10.18637/jss.v042.i08.
van Ginkel R, Reitsma JB, Büller HA, van Wijk MP, Taminiau JAJM, Benninga MA. Childhood constipation: longitudinal follow-up beyond puberty. Gastroenterology. 2003;125:357–63. https://doi.org/10.1016/S0016-5085(03)00888-6.
Gershon MD. Review article: serotonin receptors and transporters—roles in normal and abnormal gastrointestinal motility. Aliment Pharmacol Ther. 2004;20:3–14. https://doi.org/10.1111/j.1365-2036.2004.02180.x.
Sikander A, Rana SV, Prasad KK. Role of serotonin in gastrointestinal motility and irritable bowel syndrome. Clin Chim Acta. 2009;403:47–55. https://doi.org/10.1016/j.cca.2009.01.028.
Tack J, Camilleri M, Chang L, Chey WD, Galligan JJ, Lacy BE, et al. Systematic review: cardiovascular safety profile of 5-HT4 agonists developed for gastrointestinal disorders. Aliment Pharmacol Ther. 2012;35:745–67. https://doi.org/10.1111/j.1365-2036.2012.05011.x.
van Schaick E, Benninga MA, Levine A, Magnusson M, Troy S. Development of a population pharmacokinetic model of prucalopride in children with functional constipation. Pharmacol Res Perspect. 2016;4: e00236. https://doi.org/10.1002/prp2.236.
Acknowledgements
We would like to thank Jennifer Hardy for data parsing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by pediatric gastroenterology divisional funds.
Conflict of Interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Ethics Approval
This retrospective chart review study involving human participants was done in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The Human Investigation Committee (IRB) of the Cincinnati Children’s Hospital approved this study.
Informed Consent
Not applicable.
Consent for Publication
Not applicable.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author Contribution Statement
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Lesley Knowlton, Erick Madis, and Alejandro Velez Lopez supervised by Dr. Neha R Santucci. Dr. Rashmi Sahay provided statistical expertise, Sarah Stiehl provided pharmaceutical expertise, while Drs. Kaul, El-Chammas and Santucci provided expertise on functional constipation and other motility disorders. The first draft of the manuscript was written by Alejandro Velez Lopez and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Previous Communications:
As an abstract/poster for the 2021 North American Society for Pediatric Gastroenterology, Hepatology and Nutrition (NASPGHAN) that only included the prucalopride cohort results. As an abstract/poster for NASPGHAN 2022 that included the conventional therapy–prucalopride cohort analysis and the propensity matched data. As an abstract/podium presentation for 2022 Pediatric Colorectal and Pelvic Reconstruction Conference (PCPLC).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Velez, A., Kaul, A., El-Chammas, K.I. et al. Safety and Effectiveness of Prucalopride in Children with Functional Constipation with and without Upper Symptoms. Pediatr Drugs 26, 187–195 (2024). https://doi.org/10.1007/s40272-023-00612-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40272-023-00612-8