Abstract
Background
Rotator cuff injury (RCI) is a common shoulder injury, which is difficult to be completely repaired by surgery. Hence, new strategies are needed to promote the healing of tendon-bone.
Objective
We aimed to investigate the effect of human umbilical cord mesenchymal stem cells (hUC-MSCs) overexpressing RUNX1 on the tendon-bone healing after RCI, and to further explore its mechanism.
Methods
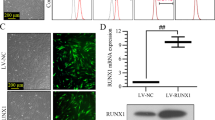
Lentiviral vector was used to mediate the overexpression of RUNX1. RUNX1-overexpressed UCB-MSCs (referred to as MSC-RUNX1) were co-cultured with osteoclasts, and TRAP staining was performed to observe the formation of osteoclasts. Then MSC-RUNX1 was cultured in osteogenic differentiation medium, Alizarin red staining was conducted to detect osteogenic differentiation. The expression of markers of osteogenesis and osteoclast was detected by RT-qPCR. EA. hy926 cells were co-cultured with MSC-RUNX1. Transwell assay was used to detect the migration, and the expression of angiogenesis related-genes VEGF and TGF-β was detected by RT-qPCR. The rat rotator cuff reconstruction model was established and MSCs were injected at the tendon-bone junction. Biomechanical test and micro-CT scanning were performed, and HE, Masson and Alcian Blue staining were used for histological evaluation of tendon-bone healing. TUNEL and PCNA immunofluorescence (IF) staining were performed to evaluate apoptosis and proliferation at the tendon-bone healing site. The levels of TNF-α, IL-6 and IL-8 in serum were detected by ELISA. The expression of CD31 and Endomucin that related to angiogenesis was detected by IF. Safranin O-fast and TRAP/CD40L immunohistochemical staining were used to assess the levels of osteoclasts and osteoblasts at the tendon-bone healing site.
Results
hUC-MSCs overexpressing RUNX1 inhibited osteoclast formation and promoted osteogenic differentiation. MSC-RUNX1 could promote the migration and tube formation of EA. hy926 cells, and up-regulate the levels of VEGF and TGF-β. Model mice treated with MSC-RUNX1 partially restored the biomechanical indexes. Treatment of MSC-RUNX1 obviously increased the bone density, accompanied by the formation of new bone. In vivo experiments showed that MSC-RUNX1 treatment could promote tendon-bone healing and inhibit inflammatory response in rats. MSC-RUNX1 treatment also promoted angiogenesis at the tendon-bone healing site, while inhibiting osteoclast formation and promoting osteogenic differentiation.
Conclusion
hUC-MSCs overexpressing RUNX1 can inhibit the formation of osteoclasts and differentiation of osteoblasts, promote angiogenesis and inhibit inflammation, thereby promoting tendon-bone healing after RCI.







Similar content being viewed by others
Data availability
The dataset used and/or analyzed in this study is available from the corresponding author on reasonable request.
References
Chen HS, Yau YC, Ko PT, Yen BL, Ho CT, Hung SC (2022) Mesenchymal stem cells from a hypoxic culture can improve rotator cuff tear repair. Cell Transplant 31:9636897221089632
Chen Z, Jin M, He H, Dong J, Li J, Nie J et al (2023) Mesenchymal stem cells and macrophages and their interactions in tendon-bone healing. J Orthopaedic Trans 39:63–73
Dang A, Davies M (2018) Rotator cuff disease: treatment options and considerations. Sports Med Arthrosc Rev 26(3):129–133
Dulak J, Szade K, Szade A, Nowak W, Józkowicz A (2015) Adult stem cells: hopes and hypes of regenerative medicine. Acta Biochim Pol 62(3):329–337
Fitzpatrick LA, Atinga A, White L, Henry PDG, Probyn L (2022) Rotator cuff injury and repair. Seminars in Musculoskeletal Radiology 26(5):585–596
Fu X, Liu G, Halim A, Ju Y, Luo Q, Song AG. Mesenchymal Stem Cell Migration and Tissue Repair. Cells. 2019;8(8).
Galipeau J, Sensébé L (2018) Mesenchymal stromal cells: clinical challenges and therapeutic opportunities. Cell Stem Cell 22(6):824–833
Han L, Fang WL, Jin B, Xu SC, Zheng X, Hu YG (2019) Enhancement of tendon-bone healing after rotator cuff injuries using combined therapy with mesenchymal stem cells and platelet rich plasma. Eur Rev Med Pharmacol Sci 23(20):9075–9084
Huang Y, He B, Wang L, Yuan B, Shu H, Zhang F et al (2020) Bone marrow mesenchymal stem cell-derived exosomes promote rotator cuff tendon-bone healing by promoting angiogenesis and regulating M1 macrophages in rats. Stem Cell Res Ther 11(1):496
Kang K, Geng Q, Cui L, Wu L, Zhang L, Li T et al (2022) Upregulation of Runt related transcription factor 1 (RUNX1) contributes to tendon-bone healing after anterior cruciate ligament reconstruction using bone mesenchymal stem cells. J Orthop Surg Res 17(1):266
Li B, Leung JCK, Chan LYY, Yiu WH, Tang SCW (2020) A global perspective on the crosstalk between saturated fatty acids and Toll-like receptor 4 in the etiology of inflammation and insulin resistance. Prog Lipid Res 77:101020
Lin KM, Wang D, Dines JS (2018) Injection therapies for rotator cuff disease. Orthop Clin North Am 49(2):231–239
Longo UG, Rizzello G, Berton A, Maltese L, Fumo C, Khan WS et al (2013) Biological strategies to enhance rotator cuff healing. Curr Stem Cell Res Ther 8(6):464–470
Longo UG, Carnevale A, Piergentili I, Berton A, Candela V, Schena E et al (2021) Retear rates after rotator cuff surgery: a systematic review and meta-analysis. BMC Musculoskelet Disord 22(1):749
Luo Y, Zhang Y, Miao G, Zhang Y, Liu Y, Huang Y (2019) Runx1 regulates osteogenic differentiation of BMSCs by inhibiting adipogenesis through Wnt/β-catenin pathway. Arch Oral Biol 97:176–184
Malekpour K, Hazrati A, Zahar M, Markov A, Zekiy AO, Navashenaq JG et al (2022) The potential use of mesenchymal stem cells and their derived exosomes for orthopedic diseases treatment. Stem Cell Reviews and Reports 18(3):933–951
Maruyama M, Rhee C, Utsunomiya T, Zhang N, Ueno M, Yao Z et al (2020) Modulation of the inflammatory response and bone healing. Front Endocrinol 11:386
Mazini L, Rochette L, Admou B, Amal S, Malka G. Hopes and Limits of Adipose-Derived Stem Cells (ADSCs) and Mesenchymal Stem Cells (MSCs) in Wound Healing. International journal of molecular sciences. 2020;21(4).
Omatsu Y, Aiba S, Maeta T, Higaki K, Aoki K, Watanabe H et al (2022) Runx1 and Runx2 inhibit fibrotic conversion of cellular niches for hematopoietic stem cells. Nat Commun 13(1):2654
Paglia DN, Diaz-Hernandez ME, Roberts JL, Kalinowski J, Lorenzo J, Drissi H (2020) Deletion of Runx1 in osteoclasts impairs murine fracture healing through progressive woven bone loss and delayed cartilage remodeling. J Orthopaedic Res 38(5):1007–1015
Samsonraj RM, Raghunath M, Nurcombe V, Hui JH, van Wijnen AJ, Cool SM (2017) Concise review: multifaceted characterization of human mesenchymal stem cells for use in regenerative medicine. Stem Cells Transl Med 6(12):2173–2185
Song F, Jiang D, Wang T, Wang Y, Chen F, Xu G et al (2017) Mechanical loading improves tendon-bone healing in a rabbit anterior cruciate ligament reconstruction model by promoting proliferation and matrix formation of mesenchymal stem cells and tendon cells. Cell Physiol Biochem 41(3):875–889
Speidel D, Wellbrock J, Abas M (2017) RUNX1 upregulation by cytotoxic drugs promotes apoptosis. Can Res 77(24):6818–6824
Tang J, Xie J, Chen W, Tang C, Wu J, Wang Y et al (2020) Runt-related transcription factor 1 is required for murine osteoblast differentiation and bone formation. J Biol Chem 295(33):11669–11681
Uccelli A, Moretta L, Pistoia V (2008) Mesenchymal stem cells in health and disease. Nat Rev Immunol 8(9):726–736
Um S, Ha J, Choi SJ, Oh W, Jin HJ (2020) Prospects for the therapeutic development of umbilical cord blood-derived mesenchymal stem cells. World J Stem Cells 12(12):1511–1528
White JL, Walker NJ, Hu JC, Borjesson DL, Athanasiou KA (2018) A Comparison of bone marrow and cord blood mesenchymal stem cells for cartilage self-assembly. Tissue Eng Part A 24(15–16):1262–1272
Xiao L, Peng Z, Zhu A, Xue R, Lu R, Mi J et al (2020) Inhibition of RUNX1 promotes cisplatin-induced apoptosis in ovarian cancer cells. Biochem Pharmacol 180:114116
Xu S, Ge J, Zhang Z, Zhou W (2018) miR-141 inhibits prostatic cancer cell proliferation and migration, and induces cell apoptosis via targeting of RUNX1. Oncol Rep 39(3):1454–1460
Xu Y, Zhang WX, Wang LN, Ming YQ, Li YL, Ni GX (2021) Stem cell therapies in tendon-bone healing. World J Stem Cells 13(7):753–775
Zhou C, Cui Y, Yang Y, Guo D, Zhang D, Fan Y et al (2021) Runx1 protects against the pathological progression of osteoarthritis. Bone Research 9(1):50
Zou J, Yang W, Cui W, Li C, Ma C, Ji X et al (2023) Therapeutic potential and mechanisms of mesenchymal stem cell-derived exosomes as bioactive materials in tendon-bone healing. Journal of Nanobiotechnology 21(1):14
Zuo R, Liu J, Zhang Y, Zhang H, Li J, Wu J et al (2022) In situ regeneration of bone-to-tendon structures: Comparisons between costal-cartilage derived stem cells and BMSCs in the rat model. Acta Biomater 145:62–76
Funding
No applicable.
Author information
Authors and Affiliations
Contributions
Conceived and designed the study: DG, JW. Performed the literature search and data extraction: JY, DL. Analyzed the data: PZ, HS. Drafted the manuscript: DG, JW.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval and consent participate
The scheme was approved by the Laboratory Animal Ethics Committee of Yangzhou university (No: 202303138).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guo, D., Yang, J., Liu, D. et al. Human umbilical cord mesenchymal stem cells overexpressing RUNX1 promote tendon-bone healing by inhibiting osteolysis, enhancing osteogenesis and promoting angiogenesis. Genes Genom 46, 461–473 (2024). https://doi.org/10.1007/s13258-023-01478-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-023-01478-3