Abstract
Background and Objectives
Identifying key factors for a successful transition from once-monthly paliperidone palmitate (PP1M) to three-monthly paliperidone palmitate (PP3M) is crucial for improving treatment outcomes, enhancing patient adherence, and reducing relapse risk in patients with schizophrenia. Providing region-specific insights for evidence-based clinical decisions can aid clinicians in optimizing transition strategies for Chinese patients with schizophrenia. Therefore, the objective of this post hoc analysis of a double-blind parallel-group multicenter phase 3 study (NCT01515423) was to identify factors related to the disease stabilization that may allow for a successful transition from PP1M to PP3M in the treatment of Chinese patients with schizophrenia.
Methods
Adults (18–70 years) diagnosed with schizophrenia using the Diagnostic and Statistical Manual of Mental Disorders, fourth edition text revision, for over 1 year and with a baseline Positive and Negative Syndrome Scale (PANSS) total score between 70 and 120 were entered into an open-label (OL) phase receiving PP1M for 17 weeks. After the 17-week OL phase, patients who met the criteria necessary for stabilization were randomized (1:1) to PP1M (fixed-dose, 50, 75, 100, or 150 mg eq.) or PP3M (fixed-dose, 175, 263, 350, or 525 mg eq.) in a 48-week double-blind phase. Stabilization was defined as a PANSS total score < 70, PANSS item (P1, P2, P3, P6, P7, G8, G14) scores ≤ 4, and a reduction in Clinical Global Impression Severity (CGI-S) score of ≥ 1 from OL baseline. This post hoc analysis evaluated changes and trends in symptom severity using PANSS, changes in mental states using CGI-S, and changes in personal and social functioning using Personal and Social Performance (PSP) scores from baseline to the endpoint of the OL phase in patients who either met or did not meet the stabilization criteria (stabilized versus non-stabilized group). Comparison of changes and trends in the clinical scores between the stabilized group and non-stabilized group were conducted using linear mixed model and Mann–Kendall trend analysis, respectively. Univariate and multivariate logistic regression analyses were conducted to explore factors associated with stabilization status for transition.
Results
Of 296 patients enrolled, 210 achieved disease stabilization (106 patients and 104 patients were randomized to PP1M and PP3M, respectively). Significant downward trends in the PANSS and CGI-S scores were detected in the stabilized patients (n = 210, ZPANSS = −2.21, p = 0.028; ZCGI-S = −2.21, p = 0.028) but not in the non-stabilized patients (n = 86). No significant trends in the PSP scores were observed in either group. The factors significantly associated with disease stabilization were the CGI-S score at baseline [odds ratio (OR) = 0.22, 95% confidence interval (CI): 0.09, 0.5), reduction of the PANSS score at week 13 (OR = 1.11, 95% CI: 1.06, 1.17), and reduction of CGI-S score at week 13 (OR = 2.27, 95% CI: 1.03, 5.02).
Conclusion
A lower CGI-S total score at baseline and greater reductions in PANSS and CGI-S scores at week 13 were associated with patients achieving disease stabilization, that may allow for a successful transition. Evidence from this study indicates that better disease condition at baseline, early functional improvement and symptomatic relief were the key factors associated with disease stabilization. The findings may guide clinicians to identify suitable patients for transition from PP1M to PP3M and further optimize the use of PP3M in China.
Clinical Trials Registration
EudraCT number: 2011-004889-15 and ClinicalTrials.gov (identifier: NCT01515423) for the original double-blind randomized study.
Similar content being viewed by others
Patients with schizophrenia showed improvements in Positive and Negative Syndrome Scale (PANSS) scores, Clinical Global Impression Severity (CGI-S) scores, and Personal and Social Functioning (PSP) scores during the 17-week open-label phase with once-monthly paliperidone palmitate (PP1M) treatment. |
Patients who had a lower CGI-S score at baseline were more likely to achieve disease stabilization upon transitioning to PP3M. |
Significant reductions in PANSS and CGI-S scores at week 13 were also predictors of successful transition and disease stabilization in patients switching to PP3M. |
1 Introduction
1.1 Schizophrenia
Schizophrenia is a chronic, recurrent, highly disabling psychotic disorder [1]. Only 10–15% of patients may have significant improvements in clinical and social domains, and up to 80% of patients may experience a relapse within 5 years of the initial diagnosis [2, 3]. Relapse means more than just the return of symptoms as it has been reported to induce brain structural changes that can potentially result in disease progression and treatment resistance [4]. Treatment discontinuation is the most common cause of relapse in schizophrenia [5]. Patients who discontinue their medication are five times more likely to relapse than those who continue [6]. Compromised adherence has been associated with poor prognosis, such as worsening of symptoms and functional outcome and an increased risk of suicidal ideation and behavior [7,8,9,10,11]. Moreover, the healthcare burden associated with a diagnosis of schizophrenia is substantial due to the relapses and stress experienced by caregivers, which consistently result in an increase in re-hospitalization, length of stay, and cost [12, 13]. These challenges foster the need for employing newly designed long-acting injectable (LAI) antipsychotics that are especially tailored for patients who have a history of non-adherence to oral antipsychotic treatment (OAT).
1.2 LAI Antipsychotics
The 2020 American Psychiatric Association Clinical Practice Guideline for the treatment of schizophrenia recommended that patients with a history of poor or uncertain adherence should receive treatment with LAI antipsychotics [14]. Overall, results in a global population have demonstrated that LAIs are indeed more effective than or at least comparable to OAT in improving clinical outcomes across different study designs [15, 16]. Once-monthly paliperidone palmitate (PP1M) formulation is a common LAI formulation and was launched in China in 2012 [17]. Past studies have shown that treatment with PP1M significantly improves medication adherence and effectively reduces the risk of recurrence while also promoting social functioning [18,19,20]. As a result, three-monthly paliperidone palmitate (PP3M) was subsequently developed to optimize the formulation and lengthen the injection interval [21]. Compared with PP1M, PP3M is noninferior in safety and efficacy and has the extra advantage of increasing adherence because of the reduction in dosing frequency [22,23,24]. Expert consensus in China has recommended that LAI antipsychotics should be a treatment choice for all patients with schizophrenia and that patients should be switched to the longer formulation if they reach stabilization, i.e., Positive and Negative Syndrome Scale (PANSS) total score ≤ 70 and selected PANSS item (P1, P2, P3, P6, P7, G8, G14) scores ≤ 4 [25]. PP3M is therefore a promising treatment option for those patients who are in a stable state following treatment with PP1M or who simply cannot adhere to a dosing schedule of monthly injections [26].
1.3 Considerations for Transition from PP1M to PP3M
Timing of the therapeutic transition from PP1M to PP3M needs to be carefully considered. Premature transition may result in an ineffective or intolerable dose, resulting in an increased risk of relapse or adverse event [26, 27]. The treatment duration with PP1M must be at least 4 months to ensure patients’ symptoms are stable before switching to PP3M [26]. Several factors may potentially impact treatment outcomes, including disease-related factors (baseline state, duration of illness, and symptom reduction, etc.), medication-related factors (previous antipsychotic drug use, medication dose and adherence, etc.) and other sociodemographic factors (age, sex, and race, etc.) [27,28,29]. Clinical guidelines for the dosage of LAI antipsychotics that should be employed for transition remain unclear. Even though there are several studies evaluating LAI transition strategies, the Chinese population has rarely been included in the process of developing an effective strategy for transitioning patients with schizophrenia from PP1M to PP3M [27, 30]. Most practicing clinicians in China rely primarily on their clinical experience to decide whether to switch a patient from PP1M to PP3M [18]. Exploring the factors associated with a successful transition could provide a useful reference for clinicians and help them identify patients who would benefit optimally for undergoing treatment transition.
To provide a more specific understanding of the clinical characteristics associated with disease stability in patients receiving PP1M, we conducted a post hoc analysis focusing on a Chinese patient subgroup from a double-blind placebo-controlled relapse–prevention study that evaluated the efficacy and safety of PP3M compared with PP1M in patients with schizophrenia (NCT01515423) [23]. Only patients treated with PP1M who fulfilled protocol-defined criteria of stabilization based on the Positive and Negative Syndrome Scale (PANSS) in the open-label phase were randomized to either PP3M or PP1M in the double-blind phase. Therefore, meeting the criteria for randomization in the double-blind phase served as an indicator of stabilization and successful transition from PP1M to PP3M. We compared the characteristics of Chinese patients who met stabilization criteria (randomized) with those who did not (non-randomized) and explored the factors associated with successful transition.
2 Methods
2.1 Patients
The full methodology of the original study has been previously published [23]. Distinctively, this post hoc analysis was exclusively conducted in the subgroup of Chinese patients. In this study, adult patients from China (male and female aged from 18 to 70 years) diagnosed with schizophrenia (based on the Diagnostic and Statistical Manual of Mental Disorders, fourth edition, text revision DSM-IV-TR criteria) for ≥ 1 year, and whose PANSS scores were between 70 and 120, were included as eligible subjects. Exclusion criteria included an active DSM-IV-TR diagnosis other than schizophrenia, significant risk of suicidal behavior, history of substance dependence within 6 months before screening, involuntary admission to a psychiatric hospital at screening, history of neuroleptic malignant syndrome, tardive dyskinesia, any unstable or significant medical or neurological illness, morbid obesity [body mass index (BMI) > 40 kg/m2], or other systemic diseases, cognitive impairment, risk factors for prolonged QT interval, torsade de pointes, or sudden death. Patients with a history of medication intolerability or lack of response to risperidone or paliperidone were also excluded from this trial.
The study protocol of the trial was reviewed by an independent Ethics Committee or Institutional Review Board at each Chinese site (listed in Online Resource Table S1). The trial was conducted in compliance with the Declaration of Helsinki consistent with Good Clinical Practices and applicable regulatory requirements. Written informed consent was obtained from all patients before enrollment.
2.2 Study Design
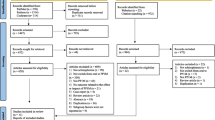
The original study consisted of four phases: a screening phase (up to 3 weeks), an open-label phase (17 weeks with flexible doses of PP1M), a double-blind phase (48 weeks with fixed doses), and a follow-up phase (Fig. 1). A washout period and oral tolerability testing was performed during the screening phase. After results from the screening labs and electrocardiograms (ECGs) were available, patients entered the open-label phase, where all patients received PP1M for 17 weeks [day 1: 150 mg eq. (deltoid; day 8: 100 mg eq. (deltoid); weeks 5 and 9: flexibly dosed 50, 75, 100, or 150 mg eq. (deltoid or gluteal); week 13: same dose of PP1M as at week 9). Then, patients with predefined disease stabilization criteria entered the double-blind phase and were further randomized to receive a fixed dose regimen into the PP3M group (175, 263, 350, or 525 mg eq.), which was 3.5 times the last dose of PP1M, and was administered every 3 months; the months in between patients received placebo to maintain the blinding conditions, while the PP1M group (50, 75, 100, or 150 mg eq.) followed with the 48-week follow-up period.
Clinical stabilization was defined as a PANSS score < 70 and scores of ≤ 4 for the PANSS items P1 (delusions), P2 (conceptual disorganization), P3 (hallucinatory behavior), P6 (suspiciousness /persecution), P7 (hostility), G8 (uncooperativeness), and G14 (poor impulse control) at last observation carried forward (LOCF).
To investigate the factors associated with disease stabilization that may allow for a successful transition from PP1M to PP3M, the present study focused on data reported in the open-label phase where patients were given 17-week PP1M. By week 17, patients who met the predefined disease stabilization criteria were considered to be in the stabilized group eligible for randomization, while those who did not meet the stabilization criteria were included in the non-stabilized group and did not go into the randomization process.
2.3 Study Outcomes
The purpose of this study was to identify factors that influenced disease stabilization to allow for transition from PP1M to PP3M.
Disease stabilization and remission of neuropsychiatric symptoms were evaluated by the 30-item PANSS scale, which consists of three sub-scales (including 7 items assessing positive symptoms, 7 items assessing negative symptoms, and 16 general psychopathology items) [31]. It was a seven-point scale rated from one (symptoms absent) to seven (extreme). A total scale score was calculated ranging from 30 to 210. Higher scores represent more severe symptoms. The PANSS score was measured at baseline and at weeks 1, 5, 9, 13, and 17.
The Personal and Social Performance (PSP) was used to assess psychosocial functions, which were incorporated into four domains (socially useful activities, personal and social relationships, self-care, and disturbing and aggressive behavior) [32]. The PSP provided a single, overall rating score ranging from 1 to 100, where higher scores represent better personal and social functioning. The PSP score was measured at baseline and at weeks 1, 9, and 17.
The one-item Clinical Global Impression Severity scale (CGI-S) was used to assess illness severity. It is a seven-point scale rated from one (normal, not at all ill) to seven (among the most extremely ill) [33]. Higher scores represent more severe illness. The CGI-S score was measured at baseline and at weeks 5, 9, 13, and 17.
Regarding the exploration of factors influencing the stabilization status before transition, the following potential factors were analyzed:
-
(1)
Demographics—age and sex;
-
(2)
Treatment related factors—types of antipsychotic medications before enrollment, administration of clozapine before enrollment, and the dose of the last single injection before randomization;
-
(3)
Clinical scores—the total and reduction score of PANSS, PSP, and CGI-S at baseline and at each time point.
2.4 Statistical Analysis
This post hoc analysis included patients who enrolled in the open-label phase and made a comparison between those who met disease stabilization criteria and those who did not. Demographic and baseline clinical characteristics between groups were summarized and compared using t-test or chi-squared test. The percentage of patients with stabilization over time was calculated and compared between groups using Fisher’s exact test.
A linear mixed model was fitted to evaluate the change in the clinical score, including PANSS total score, CGI-S score, and PSP total scores during the open-label phase after adjustments for baseline characteristics. Mann–Kendall trend test [34] was used to evaluate the trend in the change of the clinical score within each group during the open-label phase.
Univariate and multivariate logistic models were fitted to evaluate the influencing factors for successful transition. Initially, a univariate analysis was applied and those independent variables with a p < 0.1 were considered as candidate variables in the multivariate logistic model (Online Resource Table S2). A multivariate logistic regression analysis was then performed to determine the variables that were significantly associated with disease stabilization. A variance inflation factor (VIF) < 5 was used as a threshold to avoid multicollinearity issues.
3 Results
3.1 Demographic and Baseline Characteristics
Overall, 296 patients from China were included in this post hoc analysis. The proportion of males (43.6%) was lower than that of females (56.4%). The mean [standard deviation (SD)] age was 32.2 (10.6) years. Patients’ mean (SD) PANSS score, CGI-S score, and PSP total score at baseline were 87.1 (12.0), 4.8 (0.7), and 51.5 (13.4), respectively.
Of the included patients at week 17 of the open-label phase, 210 (71%) met the predefined stabilization criteria and 86 patients did not meet the criteria. At baseline of the open-label phase, patients who met the stabilization criteria had significantly lower PANSS and CGI-S scores but a higher PSP score than non-stabilized patients (Table 1).
3.2 Assessment of Clinical Outcomes
During the week 17, open-label, flexible-dose phase when patients received PP1M, PANSS, CGI-S, and PSP scores improved in both the stabilized and non-stabilized group (Fig. 2a–c). For all visits during the open-label phase, PANSS scores of stabilized patients were significantly lower than those of the non-stabilized patients at all time points, from baseline to week 17. At baseline, the least squares (LS) mean of the PANSS total score was 90.05 [95% confidence interval (CI): 87.42, 92.68] in the non-stabilized group and 85.93 (95% CI: 84.33, 87.53) in the stabilized group. After four-monthly doses of PP1M, at week 17, the least squares (LS) mean of the PANSS total score decreased from 85.93 to 56.61 in the stabilized group and from 90.05 to 83.66 in the non-stabilized group. The LS mean difference in the PANSS score between the two groups was significant with a score difference of −4.11 (95% CI: −7.18, −1.05, p < 0.05) at baseline and −27.05 (95% CI: −31.46, −22.65, p < 0.001) at week 17.
Least squares mean a PANSS total scores, b PSP scores, and c CGI-S scores during open-label PP1M and PP3M treatment in randomized and non-randomized patients. CGI-S Clinical Global Impression-Severity, LS least square, PANSS Positive and Negative Syndrome Scale, PP1M once-monthly paliperidone palmitate, PP3M three-monthly paliperidone palmitate, PSP Personal and Social Performance
Compared with the non-stabilized group, the CGI-S scores in the stabilized group were significantly lower at each follow-up visit from baseline to week 17. CGI-S scores decreased from 4.66 at baseline to 3.01 in the stabilized group and from 4.97 to 4.58 in the non-stabilized group at week 17. The mean differences in the CGI-S score between the two groups changed from −0.30 (95% CI: −0.48, −0.13, p = 0.001) at baseline to −1.58 (95% CI: −1.87, −1.28, p < 0.001) at week 17.
The PSP total scores of the stabilized patients were all significantly higher than those of the non-stabilized patients (p < 0.05). The PSP total scores increased from 52.74 to 67.92 in the stabilized patients and from 48.57 to 49.85 in the non-stabilized patients. The mean difference in the PSP total score was significant between the two groups with a value of 4.17 (95% CI: 0.93, 7.41, p < 0.05) at baseline and 18.07 (95% CI: 14.77, 21.38, p < 0.001) at week 17.
Trend analyses in the stabilized and non-stabilized patients revealed a significant downward trend in the PANSS and CGI-S scores in the stabilized patients (ZPANSS = −2.21, p = 0.028; ZCGI-S = −2.21, p = 0.028) and no significant trends in the non-stabilized patients (ZPANSS = 0.00, p = 1.000; ZCGI-S = −1.23, p = 0.221). There was no significant trend in the PSP score in either group of patients (Zstabilized = 1.04, p = 0.296; Znon-stabilized = 1.04, p = 0.296.
During the OL phase, the percentage of patients who met the stabilization criteria was constantly higher in the group eligible for randomization by week 17 (from 17.6% at week 1 to 78.6% at week 13) compared with that of the group ineligible for randomization (from 12.0% at week 1 to 18.2% at week 13, Fig. 3).
3.3 Factors Associated with Stabilization Allowing for Successful Transition
The total PANSS, CGI-S, total PSP scores at baseline, the dose of the last single injection before randomization, the reduction in the PANSS score at weeks 1, 5, and 13, and the CGI-S score at weeks 5 and 13, all demonstrated p < 0.1 in the univariate analysis and were included in the multiple regression analysis.
Multivariable logistic models of included factors from the univariate analysis demonstrated that the CGI-S score at baseline [odds ratio (OR) = 0.22, 95% CI: 0.09, 0.51, p < 0.001], reduction in the PANSS score at week 13 (OR = 1.11, 95% CI: 1.06, 1.17, p < 0.001), and reduction in the CGI-S score at week 13 (OR = 2.27, 95% CI: 1.03, 5.02, p = 0.043) were significantly associated with a successful transition from PP1M to PP3M after adjustment (Table 2).
4 Discussion
This post hoc analysis of an open-label clinical trial explored potential factors associated with disease stabilization that may allow for a successful transition from PP1M to PP3M among Chinese patients with schizophrenia, including preexisting conditions, demographics, and clinical scores.
4.1 Disease Stabilization and Better Disease Condition
Our analysis demonstrated that achieving disease stabilization was associated with a better initial disease condition at baseline and with symptomatic and functional improvement during the open-label phase, especially in the early stage. Specifically, patients in the stabilized group exhibited better disease management, as evidenced by notable improvement in PANSS, CGI-S, and PSP scores, compared with the non-stabilized group. The proportion of patients achieving disease stabilization was significantly higher at all follow-up visits after week 1 in the group defined as stabilized by week 17 than that in the group defined as non-stabilized by that time. In addition, greater improvement in the PANSS and CGI-S scores was observed in the stabilized group during the open-label phase and showed a steeper trend in improvement when compared with the non-stabilized group, especially within the first 4 weeks. Our findings are consistent with previous studies that showed lower psychopathology or illness severity scores at baseline and early improvement in symptoms or functioning were predictive of symptomatic remission [35, 36]. Results from previous studies highlight the significance of identifying early improvement and trends in symptoms and functions in patients treated with PP1M, so as to achieve subsequent treatment conversion. However, demographic and treatment-related factors were not found to be associated with disease stabilization. These findings may indicate that follow-up is of equal importance for patients, irrespective of prior antipsychotic medication use [37].
4.2 Factors Related to Disease Stabilization
Results from the multivariate analysis revealed that a lower CGI-S score at baseline and greater reduction in PANSS and CGI-S scores at week 13 were significantly associated with disease stabilization. Similar results were observed in another study conducted in Chinese patients with schizophrenia, which concluded that symptom and functional improvements were commonly associated with favorable outcomes [38]. However, while the significant effect revealed in this study was a reduction in PANSS at week 5, we found the cut-off to be week 13. A reduction in the PANSS at week 13 further emphasizes the cumulative effect of improvement and the importance of continued follow-up.
In our study, CGI-S had the greatest effect when compared with PANSS. A one-point reduction in the CGI-S total score was associated with in a 2.2-fold increase in the likelihood of achieving stabilization. This was consistent with another similar study conducted in a global population, which examined the predictors of achieving remission in patients with schizophrenia treated with PP3M [39]. These results indicated it was feasible for psychiatrists to apply either the CGI-S or the PANSS rating instruments to identify whether a patient was suitable for switching from PP1M to PP3M therapy. However, it is more time-consuming for psychiatrists to conduct a comprehensive evaluation using the PANSS scale, which is longer and more tedious to conduct. The CGI-S score is preferred due to its simplicity [40]. A study of four large clinical trials of antipsychotic medications also support the validity of extrapolating from the CGI-S to the PANSS [41].
4.3 Implications for Clinical Practice
Treatment with PP3M represents a different paradigm in the management of schizophrenia [23]. LAIs not only ensure that patients with adherence challenges receive their medication, but also effectively addresses hidden non-adherence and alleviates patients from the daily dose regimen [42]. Given the high relapse rate associated with mental illness, long-period maintenance medication is a fundamental principle to follow during the treatment period. Typically, drug maintenance therapy lasts for 3–5 years, and many patients may even require medication for the rest of their lives [43]. Compared with PP1M, PP3M is noninferior in safety and efficacy, with a significant advantage in treatment adherence reported in the real world [22,23,24]. Furthermore, a subgroup analysis has also suggested that PP3M is efficacious in East Asian populations with no new safety issues when compared with the global population [44].
As PP3M has a longer half-life than PP1M and premature transition to PP3M may cause unsatisfactory outcomes [22], it is crucial to ensure dose effectiveness and tolerability at steady plasma concentration. It has been recommended that patients with schizophrenia should be treated with PP1M and respond well to PP1M for at least 4 months—with the same last two consecutive doses considered proper—before transitioning to PP3M from PP1M [27]. Those patients who had early and clinically meaningful improvements in disease symptoms and severity while establishing a stable PP1M dose were more likely to achieve remission following the transition to PP3M [39]. Although PP1M has been approved in China for 10 years, LAIs remain underutilized in clinical practice, with the lowest usage rate of LAIs (0.66%) among 15 Asian countries and regions, far below the average (17.9%) [45].
4.4 Limitations
This study has several limitations that may affect the extrapolation of the results. Our findings are based on a post hoc analysis, which was not designed to fit our original study purpose. Thus, unmeasured confounding bias may have been introduced due to the retrospective grouping of patients and the selection of the influential factors associated with stabilization. In addition, some patients did not enter the double-blind phase because of consent withdrawal or lack of efficacy instead of non-stabilization. Other aspects in the selection of optimal candidates for transition to PP3M should be considered when determining patients’ suitability for transition from PP1M to PP3M, including patient preferences, willingness, drug efficacy, and tolerability. Despite these limitations, our results provide further information for clinicians to use in deciding which patients with schizophrenia can be switched to PP3M from PP1M.
5 Conclusion
Better disease condition at baseline and symptomatic and early functional improvement were the main characteristics associated with achieving disease stabilization. A lower CGI-S total score at baseline and greater reductions in PANSS and CGI-S scores at week 13 were also associated with patients achieving disease stabilization. Findings from this post hoc analysis may guide clinicians in China to identify suitable patients for transition from treatment with PP1M to PP3M and may further promote the use of PP3M in clinical practice.
References
Haller CS, Padmanabhan JL, Lizano P, Torous J, Keshavan M. Recent advances in understanding schizophrenia. F1000prime Reports. 2014. https://doi.org/10.1270/P6-57.
Harrow M, Jobe T, Faull R. Does treatment of schizophrenia with antipsychotic medications eliminate or reduce psychosis? A 20-year multi-follow-up study. Psychol Med. 2014;44(14):3007–16. https://doi.org/10.1017/S0033291714000610.
Jääskeläinen E, Juola P, Hirvonen N, McGrath JJ, Saha S, Isohanni M, et al. A systematic review and meta-analysis of recovery in schizophrenia. Schizophr Bull. 2013;39(6):1296–306. https://doi.org/10.1093/schbul/sbs130.
Emsley R, Chiliza B, Asmal L. The evidence for illness progression after relapse in schizophrenia. Schizophr Res. 2013;148(1–3):117–21. https://doi.org/10.1016/j.schres.2013.05.016.
Lindenmayer J-P, Liu-Seifert H, Kulkarni PM, Kinon BJ, Stauffer V, Edwards SE, et al. Medication nonadherence and treatment outcome in patients with schizophrenia or schizoaffective disorder with suboptimal prior response. J Clin Psychiatry. 2009;70(7):990. https://doi.org/10.4088/JCP.08m04221.
Robinson D, Woerner MG, Alvir JMJ, Bilder R, Goldman R, Geisler S, et al. Predictors of relapse following response from a first episode of schizophrenia or schizoaffective disorder. Arch Gen Psychiatry. 1999;56(3):241–7. https://doi.org/10.1001/archpsyc.56.3.241.
Porcelli S, Bianchini O, De Girolamo G, Aguglia E, Crea L, Serretti A. Clinical factors related to schizophrenia relapse. Int J Psychiatry Clin Pract. 2016;20(2):54–69. https://doi.org/10.3109/13651501.2016.1149195.
Wang C-Y, Xiang Y-T, Cai Z-J, Weng Y-Z, Bo Q-J, Zhao J-P, et al. Risperidone maintenance treatment in schizophrenia: a randomized, controlled trial. Am J Psychiatry. 2010;167(6):676–85. https://doi.org/10.1176/appi.ajp.2009.09030358.
Zygmunt A, Olfson M, Boyer CA, Mechanic D. Interventions to improve medication adherence in schizophrenia. Am J Psychiatry. 2002;159(10):1653–64. https://doi.org/10.1176/appi.ajp.159.10.1653.
Long Y, Tong X, Awad M, Xi S, Yu Y. Violence, runaway, and suicide attempts among people living with schizophrenia in China: prevalence and correlates. PeerJ. 2022;10: e13033. https://doi.org/10.7717/peerj.13033.
Pecenak J. Relapse prevention in schizophrenia: evidence from longterm, randomized, double-blind clinical trials. Neuro Endocrinol Lett. 2007;28(suppl 1):49–70.
Sun SX, Liu GG, Christensen DB, Fu AZ. Review and analysis of hospitalization costs associated with antipsychotic nonadherence in the treatment of schizophrenia in the United States. Curr Med Res Opin. 2007;23(10):2305–12. https://doi.org/10.1185/030079907X226050.
Dilla T, Ciudad A, Alvarez M. Systematic review of the economic aspects of nonadherence to antipsychotic medication in patients with schizophrenia. Patient Prefer Adherence. 2013;7:275. https://doi.org/10.2147/PPA.S41609.
Keepers GA, Fochtmann LJ, Anzia JM, Benjamin S, Lyness JM, Mojtabai R, et al. The American Psychiatric Association practice guideline for the treatment of patients with schizophrenia. Am J Psychiatry. 2020;177(9):868–72. https://doi.org/10.1176/appi.ajp.2020.177901.
Okoli CT, Kappi A, Wang T, Makowski A, Cooley AT. The effect of long-acting injectable antipsychotic medications compared with oral antipsychotic medications among people with schizophrenia: a systematic review and meta-analysis. Int J Ment Health Nurs. 2022;31(3):469–535. https://doi.org/10.1111/inm.12964.
Kishimoto T, Hagi K, Kurokawa S, Kane JM, Correll CU. Long-acting injectable versus oral antipsychotics for the maintenance treatment of schizophrenia: a systematic review and comparative meta-analysis of randomised, cohort, and pre-post studies. Lancet Psychiatry. 2021;8(5):387–404. https://doi.org/10.1016/s2215-0366(21)00039-0.
2012 China Drug Evaluation report: Center for Drug Evaluation, NMPA; 2013. Available from: https://www.cde.org.cn/main/news/viewInfoCommon/a28681d04382f4546668447fdaba90dd.
Magnusson MO, Samtani MN, Plan EL, Jonsson EN, Rossenu S, Vermeulen A, et al. Dosing and switching strategies for paliperidone palmitate 3-month formulation in patients with schizophrenia based on population pharmacokinetic modeling and simulation, and clinical trial data. CNS Drugs. 2017;31(4):273–88. https://doi.org/10.1007/s40263-017-0416-1.
Si T, Zhang K, Tang J, Fang M, Li K, Zhuo J, et al. Efficacy and safety of flexibly dosed paliperidone palmitate in Chinese patients with acute schizophrenia: an open-label, single-arm, prospective, interventional study. Neuropsychiatr Dis Treat. 2015;11:1483. https://doi.org/10.2147/NDT.S81760.
Ohnishi T, Kobayashi H, Yamaoka T, Toma T, Imai K, Wakamatsu A, et al. The effects of paliperidone palmitate 1 month on the employment status and social functioning of patients with schizophrenia. Innov Clin Neurosci. 2020;17(1–3):36.
Wallman P, Clark I, Taylor D. Discontinuation and relapse with paliperidone palmitate three-monthly for maintenance of schizophrenia: two year follow-up of use in clinical practice. J Psychopharmacol. 2021;35(9):1091–8. https://doi.org/10.1177/02698811211009794.
Berwaerts J, Liu Y, Gopal S, Nuamah I, Xu H, Savitz A, et al. Efficacy and safety of the 3-month formulation of paliperidone palmitate vs placebo for relapse prevention of schizophrenia: a randomized clinical trial. JAMA Psychiat. 2015;72(8):830–9. https://doi.org/10.1001/jamapsychiatry.2015.0241.
Savitz AJ, Xu H, Gopal S, Nuamah I, Ravenstijn P, Janik A, et al. Efficacy and safety of paliperidone palmitate 3-month formulation for patients with schizophrenia: a randomized, multicenter, double-blind, noninferiority study. Int J Neuropsychopharmacol. 2016;19(7):pyw018. https://doi.org/10.1093/ijnp/pyw018.
Li G, Keenan A, Daskiran M, Mathews M, Nuamah I, Orman C, et al. Relapse and Treatment adherence in patients with schizophrenia switching from paliperidone palmitate once-monthly to three-monthly formulation: a retrospective health claims database analysis. Patient Prefer Adherence. 2021;15:2239–48. https://doi.org/10.2147/ppa.S322880.
Yang K-C, Liao Y-T, Yang Y-K, Lin S-K, Liang C-S, Bai Y-M. Evidence-based expert consensus regarding long-acting injectable antipsychotics for schizophrenia from the Taiwanese Society of Biological Psychiatry and Neuropsychopharmacology (TSBPN). CNS Drugs. 2021;35(8):893–905. https://doi.org/10.1007/s40263-021-00838-5.
Gopal S, Vermeulen A, Nandy P, Ravenstijn P, Nuamah I, Buron Vidal JA, et al. Practical guidance for dosing and switching from paliperidone palmitate 1 monthly to 3 monthly formulation in schizophrenia. Curr Med Res Opin. 2015;31(11):2043–54. https://doi.org/10.1185/03007995.2015.1085849.
O’Donnell A, Rao S, Turkoz I, Gopal S, Kim E. Defining, “adequately treated”: a post hoc analysis examining characteristics of patients with schizophrenia successfully transitioned from once-monthly paliperidone palmitate to once-every-3-months paliperidone palmitate. Neuropsychiatr Dis Treat. 2021;17:1. https://doi.org/10.2147/NDT.S278298.
Si TM, Long Zhang Y, Feng Y, Zhuo JM, Cai S, Zhang L. Association between symptom control and functional improvement in patients with acute schizophrenia: a post hoc analysis of an open-label, single-arm, multi-center study of paliperidone-extended release formulation. Psychiatry Res. 2019;274:301–5. https://doi.org/10.1016/j.psychres.2019.02.036.
Ohnishi T, Wakamatsu A, Kobayashi H. Early improvement of psychiatric symptoms with long-acting injectable antipsychotic predicts subsequent social functional remission in patients with schizophrenia. Neuropsychiatr Dis Treat. 2021;17:1095. https://doi.org/10.2147/NDT.S294503.
Turkoz I, Daskiran M, Starr HL, Najarian D, Lopena O, Obando C, et al. Comparing relapse rates in real-world patients with schizophrenia who were adequately versus not adequately treated with paliperidone palmitate once-monthly injections before transitioning to once-every-3-months injections. Neuropsychiatric Dis Treat. 2022;18:1927–37. https://doi.org/10.2147/ndt.S373725.
Kay SR, Fiszbein A, Opler LA. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull. 1987;13(2):261–76. https://doi.org/10.1093/schbul/13.2.261.
Nasrallah H, Morosini P, Gagnon DD. Reliability, validity and ability to detect change of the personal and social performance scale in patients with stable schizophrenia. Psychiatry Res. 2008;161(2):213–24. https://doi.org/10.1016/j.psychres.2007.11.012.
Berk M, Ng F, Dodd S, Callaly T, Campbell S, Bernardo M, et al. The validity of the CGI severity and improvement scales as measures of clinical effectiveness suitable for routine clinical use. J Eval Clin Pract. 2008;14(6):979–83. https://doi.org/10.1111/j.1365-2753.2007.00921.x.
Mann HB. Nonparametric tests against trend. Econometrica. 1945. https://doi.org/10.2307/1907187.
Lambert M, Schimmelmann BG, Naber D, Schacht A, Karow A, Wagner T, et al. Prediction of remission as a combination of symptomatic and functional remission and adequate subjective well-being in 2960 patients with schizophrenia. J Clin Psychiatry. 2006;67(11):1690–7. https://doi.org/10.4088/jcp.v67n1104.
Lambert M, De Marinis T, Pfeil J, Naber D, Schreiner A. Establishing remission and good clinical functioning in schizophrenia: predictors of best outcome with long-term risperidone long-acting injectable treatment. Eur Psychiatry. 2010;25(4):220–9. https://doi.org/10.1016/j.eurpsy.2009.09.001.
Lambert M, Karow A, Leucht S, Schimmelmann BG, Naber D. Remission in schizophrenia: validity, frequency, predictors, and patients’ perspective 5 years later. Dialogues Clin Neurosci. 2022. https://doi.org/10.3188/DCNS.2010.12.3/mlambert.
Li N, Feng Y, Lu H, Cai SL, Zhuo J, Si T, et al. Factors related to improvement of symptoms, function, and caregiver burden in Chinese patients with schizophrenia after switching to paliperidone palmitate once-monthly from oral antipsychotics. Neuropsychiatr Dis Treat. 2018;14:825. https://doi.org/10.2147/NDT.S158353.
Nash AI, Turkoz I, Savitz AJ, Mathews M, Kim E. Predictors of achieving remission in schizophrenia patients treated with paliperidone palmitate 3-month formulation. Neuropsychiatr Dis Treat. 2019;15:731. https://doi.org/10.2147/NDT.S194264.
Busner J, Targum SD. The clinical global impressions scale: applying a research tool in clinical practice. Psychiatry (Edgmont). 2007;4(7):28.
Levine SZ, Rabinowitz J, Engel R, Etschel E, Leucht S. Extrapolation between measures of symptom severity and change: an examination of the PANSS and CGI. Schizophr Res. 2008;98(1–3):318–22. https://doi.org/10.1016/j.schres.2007.09.006.
Kane JM, Garcia-Ribera C. Clinical guideline recommendations for antipsychotic long-acting injections. Br J Psychiatry. 2009;195(S52):s63–7. https://doi.org/10.1192/bjp.195.52.s63.
Wang X, Zhang W, Ma N, Guan L, Law SF, Yu X, et al. Adherence to antipsychotic medication by community-based patients with schizophrenia in China: a cross-sectional study. Psychiatric Services (Washington, DC). 2016;67(4):431–7. https://doi.org/10.1176/appi.ps.201500056.
Savitz AJ, Xu H, Gopal S, Nuamah I, Ravenstijn P, Hough D, et al. Efficacy and safety of paliperidone palmitate three-monthly formulation in East Asian patients with schizophrenia: subgroup analysis of a global, randomized, double-blind, Phase III, noninferiority study. Neuropsychiatr Dis Treat. 2017;13:2193. https://doi.org/10.2147/NDT.S134287.
Tang CT, Chua EC, Chew QH, He YL, Si TM, Chiu HFK, et al. Patterns of long acting injectable antipsychotic use and associated clinical factors in schizophrenia among 15 Asian countries and region. Asia Pac Psychiatry. 2020;12(4): e12393. https://doi.org/10.1111/appy.12393.
Acknowledgements
Mr. Zhanghan Lin and Dr. Minye Dong from Happy Life Tech, Ltd provided consultation on statistical analysis and manuscript outline draft.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by Xi’an Janssen Pharmaceutical Ltd. Xi’an Janssen Pharmaceutical Ltd. participated in the study design, data extraction, analysis, and interpretation of the analytical results, and the writing of the manuscript, and was involved in the decision and approval to submit the manuscript for publication.
Conflict of Interest
Gang Wang reports receiving grants outside of the submitted work from Xi'an Janssen, Eli Lily, GlaxoSmithKline, Dainippon Sumitomo Pharma, Otsuka Pharmaceutical, and ShiJiaZhuang No 4 Pharm. Shandong Luye, Chiatai Tianqing, and Jiangsu Hansoh. Xin Li, Chong Ye, Wangyi Zhang, and Miaomiao Jia are employees of Xi’an Janssen Pharmaceutical Ltd.
Availability of Data and Material
The data sharing policy of Janssen Pharmaceutical Companies of Johnson & Johnson is available at https://www.janssen.com/clinical-trials/transparency. As noted on this site, requests for access to the study data can be submitted through Yale Open Data Access [YODA] Project site at http://yoda.yale.edu.
Ethical Approval
This study was approved by Ethics Committee or Institutional Review Board of the following institutions (see Online Resource 1 for detailed information): Beijing Anding Hospital, Capital Medical University; Peking University Six Hospital; Shanghai Mental Health Center; Guangzhou Brain Hospital; The Third Affiliated Hospital of Sun Yat-sen University; The First Affiliated Hospital of Kunshan Medical University; Renmin Hospital of Wuhan University; Beijing Huilongguan Hospital; The Second People’s Hospital, Hunan Province; Xi’an Mental Health Center; The First Affiliated of Fourth Military Medical University; Tianjin Anding Hospital; The Second Affiliated Hospital of Zhejiang University School of Medicine; Zhejiang University School of Medicine; The Institution of the Mental Health Center of Hebei Province; Guangdong Provincial People’s Hospital.
Consent to Participate
Written informed consent was obtained from all patients before enrollment.
Consent for Publication
Not applicable.
Code Availability
Not applicable.
Author’s Contributions
All authors contributed substantially to the study design, analysis, and interpretation of the data and final approval of the manuscript. X.L. and G.W. designed the study. C.Y. performed the statistical analysis. X.L. and C.Y. wrote the first draft of the manuscript. G.W., W.Z., and M.J. reviewed and edited the manuscript. All authors agree to be accountable for the work.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Li, X., Ye, C., Zhang, W. et al. Factors Associated with Symptom Stabilization that Allow for Successful Transition from Once-Monthly Paliperidone Palmitate to Three-Monthly Paliperidone Palmitate: A Post Hoc Analysis Examined Clinical Characteristics in Chinese Patients with Schizophrenia. CNS Drugs 38, 55–65 (2024). https://doi.org/10.1007/s40263-023-01056-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40263-023-01056-x