Abstract
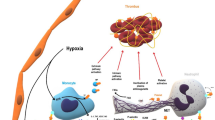
An excessive von Willebrand factor (VWF) secretion, coupled with a moderate to severe deficiency of ADAMTS13 activity, serves as a linking mechanism between inflammation to thrombosis. The former facilitates platelet adhesion to the vessel wall and the latter is required to cleave VWF multimers. As a result, the ultra-large VWF (UL-VWF) multimers released by Weibel–Palade bodies remain uncleaved. In this study, using a computational model based on first principles, we quantitatively show how the uncleaved UL-VWF multimers interact with the blood cells to initiate microthrombosis. We observed that platelets first adhere to unfolded and stretched uncleaved UL-VWF multimers anchored to the microvessel wall. By the end of this initial adhesion phase, the UL-VWF multimers and platelets make a mesh-like trap in which the red blood cells increasingly accumulate to initiate a gradually growing microthrombosis. Although high-shear rate and blood flow velocity are required to activate platelets and unfold the UL-VWFs, during the initial adhesion phase, the blood velocity drastically drops after thrombosis, and as a result, the wall shear stress is elevated near UL-VWF roots, and the pressure drops up to 6 times of the healthy condition. As the time passes, these trends progressively continue until the microthrombosis fully develops and the effective size of the microthrombosis and these flow quantities remain almost constant. Our findings quantitatively demonstrate the potential role of UL-VWF in coagulopathy.







Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author, NM, upon reasonable request.
References
Allford SL, Harrison P, Lawrie AS et al (2000) von Willebrand factor-cleaving protease activity in congenital thrombotic thrombocytopenic purpura: VWF-cleaving protease deficiency and congenital TTP. Br J Haematol 111:1215–1222. https://doi.org/10.1111/j.1365-2141.2000.02503.x
Anvari S, Osei E, Maftoon N (2021) Interactions of platelets with circulating tumor cells contribute to cancer metastasis. Sci Rep 11:15477. https://doi.org/10.1038/s41598-021-94735-y
Arabghahestani M, Poozesh S, Akafuah NK (2019) Advances in computational fluid mechanics in cellular flow manipulation: a review. Appl Sci 9:4041. https://doi.org/10.3390/app9194041
Arnold A, Lenz O, Kesselheim S et al (2013) ESPResSo 3.1: molecular dynamics software for coarse-grained models. In: Griebel M, Schweitzer MA (eds) Meshfree methods for partial differential equations VI. Springer, Berlin Heidelberg, pp 1–23
Baiocchi M, Barsoum S, Khodaei S et al (2021) Effects of choice of medical imaging modalities on a non-invasive diagnostic and monitoring computational framework for patients with complex valvular, vascular, and ventricular diseases who undergo transcatheter aortic valve replacement. Front Bioeng Biotechnol 9:643453. https://doi.org/10.3389/fbioe.2021.643453
Banno F, Kokame K, Okuda T et al (2006) Complete deficiency in ADAMTS13 is prothrombotic, but it alone is not sufficient to cause thrombotic thrombocytopenic purpura. Blood 107:3161–3166. https://doi.org/10.1182/blood-2005-07-2765
Barg A, Ossig R, Goerge T et al (2007) Soluble plasma-derived von Willebrand factor assembles to a haemostatically active filamentous network. Thromb Haemost 97:514–526
Bauer AT, Suckau J, Frank K et al (2015) von Willebrand factor fibers promote cancer-associated platelet aggregation in malignant melanoma of mice and humans. Blood 125:3153–3163. https://doi.org/10.1182/blood-2014-08-595686
Belyaev AV, Kushchenko YK (2023) Biomechanical activation of blood platelets via adhesion to von Willebrand factor studied with mesoscopic simulations. Biomech Model Mechanobiol 22:785–808. https://doi.org/10.1007/s10237-022-01681-3
Béranger N, Benghezal S, Savigny S et al (2019) Loss of von Willebrand factor high-molecular-weight multimers at acute phase is associated with detectable anti-ADAMTS13 IgG and neurological symptoms in acquired thrombotic thrombocytopenic purpura. Thromb Res 181:29–35. https://doi.org/10.1016/j.thromres.2019.07.012
Bernardo A, Ball C, Nolasco L et al (2005) Platelets adhered to endothelial cell-bound ultra-large von Willebrand factor strings support leukocyte tethering and rolling under high shear stress. J Thromb Haemost 3:562–570. https://doi.org/10.1111/j.1538-7836.2005.01122.x
Bhatnagar PL, Gross EP, Krook M (1954) A model for collision processes in gases. I. Small amplitude processes in charged and neutral one-component systems. Phys Rev 94:511–525. https://doi.org/10.1103/PhysRev.94.511
Bianchi V, Robles R, Alberio L et al (2002) Von Willebrand factor–cleaving protease (ADAMTS13) in thrombocytopenic disorders: a severely deficient activity is specific for thrombotic thrombocytopenic purpura. Blood 100:710–713. https://doi.org/10.1182/blood-2002-02-0344
Blasi A, Meijenfeldt FA, Adelmeijer J et al (2020) In vitro hypercoagulability and ongoing in vivo activation of coagulation and fibrinolysis in COVID-19 patients on anticoagulation. J Thromb Haemost 18:2646–2653. https://doi.org/10.1111/jth.15043
Blot E, Decaudin D, Veyradier A et al (2002) Cancer-related thrombotic microangiopathy secondary to Von Willebrand factor-cleaving protease deficiency. Thromb Res 106:127–130. https://doi.org/10.1016/S0049-3848(02)00095-6
Bridges DJ, Bunn J, Van Mourik JA et al (2010) Rapid activation of endothelial cells enables Plasmodium falciparum adhesion to platelet-decorated von Willebrand factor strings. Blood 115:1472–1474. https://doi.org/10.1182/blood-2009-07-235150
Chang JC (2020) Disseminated intravascular coagulation: new identity as endotheliopathy-associated vascular microthrombotic disease based on in vivo hemostasis and endothelial molecular pathogenesis. Thromb J 18:25. https://doi.org/10.1186/s12959-020-00231-0
Chaturvedi S, DeBaun MR (2022) Altered VWF:ADAMTS13 homeostasis is a target for therapeutic intervention in sickle cell disease. Proc Natl Acad Sci 119:e2213079119. https://doi.org/10.1073/pnas.2213079119
Chen S, Doolen GD (1998) Lattice Boltzmann method for fluid flows. Annu Rev Fluid Mech 30:329–364. https://doi.org/10.1146/annurev.fluid.30.1.329
Chiasakul T, Cuker A (2018) Clinical and laboratory diagnosis of TTP: an integrated approach. Hematology 2018:530–538. https://doi.org/10.1182/asheducation-2018.1.530
Cimrak I (2018) Computational blood cell mechanics: road towards models and biomedical applications, 1st edn. CRC Press, Boca Raton
Cortes GA, Moore MJ, El-Nakeep S (2023) Physiology, Von Willebrand Factor. In: StatPearls. StatPearls Publishing, Treasure Island (FL)
Dao M, Lim CT, Suresh S (2003) Mechanics of the human red blood cell deformed by optical tweezers. J Mech Phys Solids 51:2259–2280. https://doi.org/10.1016/j.jmps.2003.09.019
Dembo M, Bell GI (1987) The thermodynamics of cell adhesion. In: Bronner F, Klausner RD, Kempf C, van Renswoude J (eds) Current topics in membranes and transport. Academic Press, Cambridge, pp 71–89
Doevelaar AAN, Bachmann M, Hölzer B et al (2021) von Willebrand factor multimer formation contributes to immunothrombosis in coronavirus disease 2019. Crit Care Med 49:e512–e520. https://doi.org/10.1097/CCM.0000000000004918
Federici AB, Bader R, Pagani S et al (1989) Binding of von Willebrand factor to glycoproteins Ib and IIb/IIIa complex: affinity is related to multimeric size. Br J Haematol 73:93–99. https://doi.org/10.1111/j.1365-2141.1989.tb00226.x
Fedosov DA, Caswell B, Karniadakis GE (2010a) Systematic coarse-graining of spectrin-level red blood cell models. Comput Methods Appl Mech Eng 199:1937–1948. https://doi.org/10.1016/j.cma.2010.02.001
Fedosov DA, Caswell B, Karniadakis GE (2010b) A Multiscale red blood cell model with accurate mechanics, rheology, and dynamics. Biophys J 98:2215–2225. https://doi.org/10.1016/j.bpj.2010.02.002
Goh CY, Patmore S, Smolenski A et al (2021) The role of von Willebrand factor in breast cancer metastasis. Transl Oncol 14:101033. https://doi.org/10.1016/j.tranon.2021.101033
Groot E, Fijnheer R, Sebastian SAE et al (2009) The active conformation of von Willebrand factor in patients with thrombotic thrombocytopenic purpura in remission. J Thromb Haemost 7:962–969. https://doi.org/10.1111/j.1538-7836.2009.03433.x
Guck J, Schinkinger S, Lincoln B et al (2005) Optical deformability as an inherent cell marker for testing malignant transformation and metastatic competence. Biophys J 88:3689–3698. https://doi.org/10.1529/biophysj.104.045476
Hammer DA, Apte SM (1992) Simulation of cell rolling and adhesion on surfaces in shear flow: general results and analysis of selectin-mediated neutrophil adhesion. Biophys J 63:35–57. https://doi.org/10.1016/S0006-3495(92)81577-1
He X, Luo L-S (1997) Lattice Boltzmann model for the incompressible Navier–Stokes equation. J Stat Phys 88:927–944. https://doi.org/10.1023/B:JOSS.0000015179.12689.e4
Henry BM, Benoit SW, Oliveira MHS et al (2021) ADAMTS13 activity to von Willebrand factor antigen ratio predicts acute kidney injury in patients with COVID-19: evidence of SARS-CoV-2 induced secondary thrombotic microangiopathy. Int J Lab Hematol 43:129–136. https://doi.org/10.1111/ijlh.13415
Hollestelle MJ, Donkor C, Mantey EA et al (2006) von Willebrand factor propeptide in malaria: evidence of acute endothelial cell activation. Br J Haematol 133:562–569. https://doi.org/10.1111/j.1365-2141.2006.06067.x
Jehring L (1992) Chapman, S.; Cowling, T. G., The Mathematical Theory of Non-Uniform Gases. 3rd edition. Cambridge etc., Cambridge University Press 1990. XXIV, 422 pp., £ 19.50 P/b. ISBN 0-521-40844-X. ZAMM—J Appl Math Mech Z Für Angew Math Mech 72:610–610. https://doi.org/10.1002/zamm.19920721124
Kalafatis M, Takahashi Y, Girma J, Meyer D (1987) Localization of a collagen-interactive domain of human von Willebrand factor between amino acid residues Gly 911 and Glu 1365. Blood 70:1577–1583. https://doi.org/10.1182/blood.V70.5.1577.1577
Karpman D, Holmberg L, Jirgård L, Lethagen S (1996) Increased platelet retention in familial recurrent thrombotic thrombocytopenic purpura. Kidney Int 49:190–199. https://doi.org/10.1038/ki.1996.26
Keshavarz-Motamed Z (2020) A diagnostic monitoring and predictive tool for patients with complex valvular vascular and ventricular diseases. Sci Rep 10(1):6905. https://doi.org/10.1038/s41598-020-63728-8
Keshavarz Motamed P, Maftoon N (2021) A systematic approach for developing mechanistic models for realistic simulation of cancer cell motion and deformation. Sci Rep 11:21545. https://doi.org/10.1038/s41598-021-00905-3
Keshavarz Motamed P, Abouali H, Poudineh M, Maftoon N (2024) Experimental measurement and numerical modeling of deformation behavior of breast cancer cells passing through constricted microfluidic channels. Microsyst Nanoeng. https://doi.org/10.1038/s41378-023-00644-7
Kichloo A, Dettloff K, Aljadah M et al (2020) COVID-19 and hypercoagulability: a review. Clin Appl Thromb 26:107602962096285. https://doi.org/10.1177/1076029620962853
Larkin D, De Laat B, Jenkins PV et al (2009) Severe Plasmodium falciparum malaria is associated with circulating ultra-large von Willebrand multimers and ADAMTS13 inhibition. PLoS Pathog 5:e1000349. https://doi.org/10.1371/journal.ppat.1000349
Latt J, Malaspinas O, Kontaxakis D et al (2020) Palabos: parallel lattice Boltzmann solver. Comput Math Appl. https://doi.org/10.1016/j.camwa.2020.03.022
Lenarda P, Coclite A, Decuzzi P (2019) Unraveling the vascular fate of deformable circulating tumor cells via a hierarchical computational model. Cell Mol Bioeng 12:543–558. https://doi.org/10.1007/s12195-019-00587-y
Lester WA, Williams MD, Allford SL et al (2002) Successful treatment of congenital thrombotic thrombocytopenic purpura using the intermediate purity factor VIII concentrate BPL 8Y: short report. Br J Haematol 119:176–179. https://doi.org/10.1046/j.1365-2141.2002.03809.x
Liu ZL, Ku DN, Aidun CK (2021) Mechanobiology of shear-induced platelet aggregation leading to occlusive arterial thrombosis: a multiscale in silico analysis. J Biomech 120:110349. https://doi.org/10.1016/j.jbiomech.2021.110349
Liu ZL, Bresette C, Aidun CK, Ku DN (2022) SIPA in 10 milliseconds: VWF tentacles agglomerate and capture platelets under high shear. Blood Adv 6:2453–2465. https://doi.org/10.1182/bloodadvances.2021005692
Löf A, Walker PU, Sedlak SM et al (2019) Multiplexed protein force spectroscopy reveals equilibrium protein folding dynamics and the low-force response of von Willebrand factor. Proc Natl Acad Sci 116:18798–18807. https://doi.org/10.1073/pnas.1901794116
Mancini I, Baronciani L, Artoni A et al (2021) The ADAMTS13-von Willebrand factor axis in COVID-19 patients. J Thromb Haemost 19:513–521. https://doi.org/10.1111/jth.15191
Marco A, Marco P (2021) Von Willebrand factor and ADAMTS13 activity as clinical severity markers in patients with COVID-19. J Thromb Thrombolysis 52:497–503. https://doi.org/10.1007/s11239-021-02457-9
Martín-Rojas RM, Chasco-Ganuza M, Casanova-Prieto S et al (2021) A mild deficiency of ADAMTS13 is associated with severity in COVID-19: comparison of the coagulation profile in critically and noncritically ill patients. Blood Coagul Fibrinolysis 32:458–467. https://doi.org/10.1097/MBC.0000000000001068
Meijenfeldt FA, Havervall S, Adelmeijer J et al (2021) Prothrombotic changes in patients with COVID-19 are associated with disease severity and mortality. Res Pract Thromb Haemost 5:132–141. https://doi.org/10.1002/rth2.12462
Mills JP, Qie L, Dao M et al (2004a) Nonlinear elastic and viscoelastic deformation of the human red blood cell with optical tweezers. Mol Cell Biomech 1(3):169
Mills JP, Qie L, Dao M-T et al (2004b) Nonlinear elastic and viscoelastic deformation of the human red blood cell with optical tweezers. Mech Chem Biosyst MCB. https://doi.org/10.3970/mcb.2004.001.169
Moake JL, Chow TW (1998) Thrombotic thrombocytopenic purpura: understanding a disease no longer rare. Am J Med Sci 316:105–119. https://doi.org/10.1097/00000441-199808000-00006
Moake JL, Rudy CK, Troll JH et al (1982) Unusually large plasma factor VIII: von Willebrand factor multimers in chronic relapsing thrombotic thrombocytopenic purpura. N Engl J Med 307:1432–1435. https://doi.org/10.1056/NEJM198212023072306
Moake JL, Turner NA, Stathopoulos NA et al (1986) Involvement of large plasma von Willebrand factor (vWF) multimers and unusually large vWF forms derived from endothelial cells in shear stress-induced platelet aggregation. J Clin Invest 78:1456–1461. https://doi.org/10.1172/JCI112736
Mohamad AA (2011) Lattice Boltzmann method. Springer, London
Mountrakis L, Lorenz E, Hoekstra AG (2014) Validation of an efficient two-dimensional model for dense suspensions of red blood cells. Int J Mod Phys C 25:1441005. https://doi.org/10.1142/S0129183114410058
Mountrakis L, Lorenz E, Malaspinas O et al (2015) Parallel performance of an IB-LBM suspension simulation framework. J Comput Sci 9:45–50. https://doi.org/10.1016/j.jocs.2015.04.006
Patmore S, Dhami SPS, O’Sullivan JM (2020) Von Willebrand factor and cancer; metastasis and coagulopathies. J Thromb Haemost 18:2444–2456. https://doi.org/10.1111/jth.14976
Peskin CS (2002) The immersed boundary method. Acta Numer 11:479–517. https://doi.org/10.1017/S0962492902000077
Philippe A, Chocron R, Gendron N et al (2021) Circulating Von Willebrand factor and high molecular weight multimers as markers of endothelial injury predict COVID-19 in-hospital mortality. Angiogenesis 24:505–517. https://doi.org/10.1007/s10456-020-09762-6
Rack K, Huck V, Hoore M et al (2017) Margination and stretching of von Willebrand factor in the blood stream enable adhesion. Sci Rep 7:14278. https://doi.org/10.1038/s41598-017-14346-4
Ruggeri ZM (2003) Von Willebrand factor, platelets and endothelial cell interactions. J Thromb Haemost JTH 1:1335–1342. https://doi.org/10.1046/j.1538-7836.2003.00260.x
Ruggeri ZM, Orje JN, Habermann R et al (2006) Activation-independent platelet adhesion and aggregation under elevated shear stress. Blood 108:1903–1910. https://doi.org/10.1182/blood-2006-04-011551
Sadler JE (2017) Pathophysiology of thrombotic thrombocytopenic purpura. Blood 130:1181–1188. https://doi.org/10.1182/blood-2017-04-636431
Savage B, Saldívar E, Ruggeri ZM (1996) Initiation of platelet adhesion by arrest onto fibrinogen or translocation on von Willebrand factor. Cell 84:289–297. https://doi.org/10.1016/S0092-8674(00)80983-6
Savage B, Almus-Jacobs F, Ruggeri ZM (1998) Specific synergy of multiple substrate-receptor interactions in platelet thrombus formation under flow. Cell 94:657–666. https://doi.org/10.1016/S0092-8674(00)81607-4
Scully M, Hunt BJ, Benjamin S et al (2012) Guidelines on the diagnosis and management of thrombotic thrombocytopenic purpura and other thrombotic microangiopathies. Br J Haematol 158:323–335. https://doi.org/10.1111/j.1365-2141.2012.09167.x
Shen J, Faruqi AH, Jiang Y, Maftoon N (2021) Mathematical reconstruction of patient-specific vascular networks based on clinical images and global optimization. IEEE Access 9:20648–20661. https://doi.org/10.1109/ACCESS.2021.3052501
Shi H, Shao B, Gao L et al (2022) Endothelial VWF is critical for the pathogenesis of vaso-occlusive episode in a mouse model of sickle cell disease. Proc Natl Acad Sci 119:e2207592119. https://doi.org/10.1073/pnas.2207592119
Sinkovits G, Réti M, Müller V et al (2022) Associations between the von Willebrand factor—ADAMTS13 axis, complement activation, and COVID-19 severity and mortality. Thromb Haemost 122:240–256. https://doi.org/10.1055/s-0041-1740182
South K, Roberts L, Morris L, et al (2020) Severity-stratified and longitudinal analysis of VWF/ADAMTS13 imbalance, altered fibrin crosslinking and inhibition of fibrinolysis as contributors to COVID-19 coagulopathy. Infectious Diseases (except HIV/AIDS)
Stockschlaeder M, Schneppenheim R, Budde U (2014) Update on von Willebrand factor multimers: focus on high-molecular-weight multimers and their role in hemostasis. Blood Coagul Fibrinolysis 25:206–216. https://doi.org/10.1097/MBC.0000000000000065
Sweeney JM, Barouqa M, Krause GJ et al (2021) Low ADAMTS13 activity correlates with increased mortality in COVID-19 patients. TH Open 05:e89–e103. https://doi.org/10.1055/s-0041-1723784
Tarksalooyeh VWA, Závodszky G, van Rooij BJ, Hoekstra AG (2018) Inflow and outflow boundary conditions for 2D suspension simulations with the immersed boundary lattice Boltzmann method. Comput Fluids 172:312–317. https://doi.org/10.1016/j.compfluid.2018.04.025
Tarksalooyeh VA, Závodszky G, Hoekstra AG (2019) Optimizing parallel performance of the cell based blood flow simulation software HemoCell. In: Rodrigues JMF, Cardoso PJS, Monteiro J et al (eds) Computational science—ICCS 2019. Springer International Publishing, Cham, pp 537–547
Tsyu NG, Belyaev AV (2023) Coarse-grained simulations of von Willebrand factor adsorption to collagen with consequent platelet recruitment. Int J Numer Methods Biomed Eng. https://doi.org/10.1002/cnm.3747
Ward SE, Curley GF, Lavin M et al (2021a) Von Willebrand factor propeptide in severe coronavirus disease 2019 (COVID-19): evidence of acute and sustained endothelial cell activation. Br J Haematol 192:714–719. https://doi.org/10.1111/bjh.17273
Ward SE, Fogarty H, Karampini E et al (2021b) ADAMTS13 regulation of VWF multimer distribution in severe COVID-19. J Thromb Haemost JTH. https://doi.org/10.1111/jth.15409
Xiao X, Zhong H, Zhang G, Deng M (2013) Thrombotic thrombocytopenic purpura as initial and major presentation of multiple myeloma. J Thromb Thrombolysis 36:422–423. https://doi.org/10.1007/s11239-013-0893-0
Yazdani A, Deng Y, Li H et al (2021) Integrating blood cell mechanics, platelet adhesive dynamics and coagulation cascade for modelling thrombus formation in normal and diabetic blood. J R Soc Interface 18:20200834. https://doi.org/10.1098/rsif.2020.0834
Ye T, Zhang X, Li G, Wang S (2020) Biomechanics in thrombus formation from direct cellular simulations. Phys Rev E 102:042410. https://doi.org/10.1103/PhysRevE.102.042410
Závodszky G, Paál G (2013) Validation of a lattice Boltzmann method implementation for a 3D transient fluid flow in an intracranial aneurysm geometry. Int J Heat Fluid Flow 44:276–283. https://doi.org/10.1016/j.ijheatfluidflow.2013.06.008
Závodszky G, van Rooij B, Azizi V, Hoekstra A (2017) Cellular level in-silico modeling of blood rheology with an improved material model for red blood cells. Front Physiol 8:563. https://doi.org/10.3389/fphys.2017.00563
Závodszky G, van Rooij B, Czaja B et al (2019) Red blood cell and platelet diffusivity and margination in the presence of cross-stream gradients in blood flows. Phys Fluids 31:031903. https://doi.org/10.1063/1.5085881
Zavodszky G, van Rooij B, Azizi V et al (2017) Hemocell: a high-performance microscopic cellular library. Procedia Comput Sci 108:159–165. https://doi.org/10.1016/j.procs.2017.05.084
Zheng X, Majerus EM, Sadler JE (2002) ADAMTS13 and TTP. Curr Opin Hematol 9:389–394. https://doi.org/10.1097/00062752-200209000-00001
Zhussupbekov M, Méndez Rojano R, Wu W-T, Antaki JF (2022) von Willebrand factor unfolding mediates platelet deposition in a model of high-shear thrombosis. Biophys J 121:4033–4047. https://doi.org/10.1016/j.bpj.2022.09.040
Acknowledgements
The simulations were performed using infrastructures of Compute Canada (www.computecanada.ca). This study was supported by a grant from the Natural Sciences and Engineering Research Council of Canada.
Author information
Authors and Affiliations
Contributions
N.R. was contributed to writing—original draft, computational analysis, methodology, visualization. P.K.M. was contributed to writing—original draft, computational analysis. N.M. was contributed to writing—original draft, writing—review and editing, conceptualization, supervision, project administration, funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file2 (AVI 12008 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rahmati, N., Keshavarz Motamed, P. & Maftoon, N. Numerical study of ultra-large von Willebrand factor multimers in coagulopathy. Biomech Model Mechanobiol (2024). https://doi.org/10.1007/s10237-023-01803-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10237-023-01803-5