Abstract
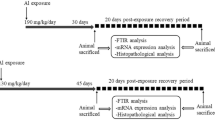
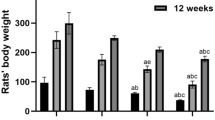
Aluminum is a potent neurotoxin, responsible for memory impairment and cognitive dysfunction. The neurotoxic effect of aluminum on cognitive impairment is well documented, however, exposure to aluminum in a time-dependent manner and post-exposure self-recovery still needs to be elaborated. This research aimed to (1) study the time-dependent effect of aluminum exposure by administering a total dose of 5850 mg/kg of Al over two different time periods: 30 and 45 days (130 and 195 mg/kg of AlCl3 respectively), and (2) study 20 days post-exposure self-recovery effect in both aluminum-exposed groups by giving distilled water. Cognitive abilities were investigated through Morris water maze test and hole board test and compared in both exposure and recovery groups. Oxidative stress markers and neurotransmitter levels were measured for both exposure and recovery groups. To understand the mechanism of aluminum exposure and recovery, immunohistochemical analysis of synaptophysin (Syp) and glial fibrillary acidic protein (GFAP) was performed. Results showed cognitive dysfunction, oxidative stress-induced damage, reduced neurotransmitter levels, decreased immunoreactivity of Syp, and increased GFAP. However, these parameters showed a larger improvement in the recovery group where rats were given aluminum for 30 days period in comparison to recovery group followed by 45 days of aluminum exposure. These results suggest that restoration of cognitive ability is affected by the duration of aluminum exposure. The study findings provide us with insight into the adverse effects of aluminum exposure and can be utilized to guide future preventive and therapeutic strategies against aluminum neurotoxicity.








Similar content being viewed by others
Abbreviations
- Al:
-
Aluminum
- CAT:
-
Catalase
- SOD:
-
Superoxide dismutase
- GPx:
-
Glutathione peroxidase
- MDA:
-
Malondialdehyde
- LPO:
-
Lipid peroxidation
- NA:
-
Noradrenaline
- DA:
-
Dopamine
- DOPAC:
-
Dihydroxyphenyl acetic acid
- 5-HT:
-
5-Hydroxytryptamine
- 5-HIAA:
-
5-Hydroxyindoleacetic acid
- DG:
-
Dentate gyrus
- Syp:
-
Synaptophysin
- GFAP:
-
Glial fibrillary protein
- HBT:
-
Hole board test
- TQ:
-
Target quadrant
- MWM:
-
Morris water maze test
- RME:
-
Reference memory error
- WME:
-
Working memory error
References
Abd el-Rady NM, Ahmed A, Abdel-Rady MM, Ismail OI (2021) Glucagon-like peptide-1 analog improves neuronal and behavioral impairment and promotes neuroprotection in a rat model of aluminum-induced dementia. Physiol Rep 8(24):e14651
Abdelmeguid NE, Khalil MIM, Elhabet R, Sultan AS, Salam SA (2021) Combination of docosahexaenoic acid and Ginko biloba extract improves cognitive function and hippocampal tissue damages in a mouse model of Alzheimer’s disease. J Chem Neuroanat 116:101995
Abdelzaher LA, Hussein OA, Ashry IEM (2021) The novel potential therapeutic utility of montelukast in alleviating autistic behavior induced by early postnatal administration of thimerosal in mice. Cell Mol Neurobiol 41:129–150
Abu-Taweel GM, Al-Mutary MG (2021a) Pomegranate juice rescues developmental, neurobehavioral and biochemical disorders in aluminum chloride-treated male mice. J Trace Elem Med Biol 63:126655
Abu-Taweel GM, Al-Mutary MG (2021b) Pomegranate juice reverses AlCl3-induced neurotoxicity and improves learning and memory in female mice. Environ Res 199:111270
Ahmed GAR, Khalil SKH, El Hotaby W et al (2020) ATR-IR and EPR spectroscopy for following the membrane restoration of isolated cortical synaptosomes in aluminium-induced Alzheimer’s disease—like rat model. Chem Phys Lipids 231:104931
Amari NO, Djebli N, Le Mai Huong TTH, Ha NDL, Bich VT (2020) Neuroprotective effect of 1,3-β-glucan-curcumin mixing (Bioglucur) on Alzheimer disease induced in mice by aluminium toxicity. Indian J Pharm Educ Res 54(4):1089–1097
Cabezas IL, Batista AH, Rol GP (2014) The role of glial cells in Alzheimer disease: potential therapeutic implications. Neurología (English Edition) 29(5):305–309
Chen X, Zhang M, Ahmed M, Surapaneni KM, Veeraraghavan VP, Arulselvan P (2021) Neuroprotective effects of ononin against the aluminium chloride-induced Alzheimer’s disease in rats. Saudi J Biol Sci 28(8):4232–4239
Cirovic A, Cirovic A, Orisakwe OE, Lima RR (2023) Local and systemic hypoxia as inductors of increased aluminum and iron brain accumulation promoting the onset of Alzheimer’s Disease. Biol Trace Elem Res. https://doi.org/10.1007/s12011-023-03599-y
Commissaris RL, Cordon JJ, Sprague S, Keiser J, Mayor GH, Rech RH (1982) Behavioral changes in rats after chronic aluminum and parathyroid hormone administration. Neurobehav Toxicol Teratol 4(3):403–410
Crowe AR, Yue W (2019) Semi-quantitative determination of protein expression using immunohistochemistry staining and analysis: an integrated protocol. Bio Protoc 9(24):e3465
Cullen CL, Burne THJ, Lavidis NA, Moritz KM (2013) Low dose prenatal ethanol exposure induces anxiety-like behaviour and alters dendritic morphology in the basolateral amygdala of rat offspring. PLoS ONE 8(1):e54924
Disdier C, Chalansonnet M, Gagnaire F et al (2017) Brain inflammation, blood brain barrier dysfunction and neuronal synaptophysin decrease after inhalation exposure to titanium dioxide nano-aerosol in aging rats. Sci Rep 7(1):1–13
Douichene S, Hammadi K, Djebli N (2016) Neuroprotective effect of Hypericum thymopsis against chronic exposure to aluminum chloride and Alzheimer’s disease. J Pharm Pharmacol 3(3):20–28
Ekong MB, Ekpo MM, Akpanyung EO, Nwaokonko DU (2017) Neuroprotective effect of Moringa oleifera leaf extract on aluminium-induced temporal cortical degeneration. Metab Brain Dis 32:1437–1447
Elizabeth MA, Samson P, Itohan OR (2020) Histomorphological evaluations on the frontal cortex extrapyramidal cell layer following administration of N-Acetyl cysteine in aluminum induced neurodegeneration rat model. Metab Brain Dis 35:829–839
El-Shetry ES, Mohamed AA-R, Khater SI et al (2021) Synergistically enhanced apoptotic and oxidative DNA damaging pathways in the rat brain with lead and/or aluminum metals toxicity: expression pattern of genes OGG1 and P53. J Trace Elem Med Biol 68:126860
Eltokhi A, Kurpiers B, Pitzer C (2020) Behavioral tests assessing neuropsychiatric phenotypes in adolescent mice reveal strain- and sex-specific effects. Sci Rep 10(1):11263. https://doi.org/10.1038/s41598-020-67758-0
Erazi H, Sansar W, Ahboucha S, Gamrani H (2010) Aluminum affects glial system and behavior of rats. C R Biol 333(1):23–27
Farhat SM, Mahboob A, Ahmed T (2017a) Cortex-and amygdala-dependent learning and nicotinic acetylcholine receptor gene expression is severely impaired in mice orally treated with AlCl3. Biol Trace Elem Res 179(1):91–101
Farhat SM, Mahboob A, Iqbal G, Ahmed T (2017b) Aluminum-induced cholinergic deficits in different brain parts and its implications on sociability and cognitive functions in mouse. Biol Trace Elem Res 177(1):115–121
Ferreira PC, Piai KdA, Takayanagui AMM, Segura-Muñoz SI (2008) Aluminum as a risk factor for Alzheimer’s disease. Rev Lat Am Enfermagem 16:151–157
Flaten TP (2001) Aluminium as a risk factor in Alzheimer’s disease, with emphasis on drinking water. Brain Res Bull 55(2):187–196
Flohé L, Günzler WA (1984) [12] Assays of glutathione peroxidase. Methods Enzymol 105:114–120
Flores-Montoya MG, Sobin C (2015) Early chronic lead exposure reduces exploratory activity in young C57BL/6J mice. J Appl Toxicol 35(7):759–765
Gilani SJ, Bin-Jumah MN, Al-Abbasi FA et al (2022) Antiamnesic potential of malvidin on aluminum chloride activated by the free radical scavenging property. ACS Omega 7(28):24231–24240
Golub MS, Donald JM, Gershwin ME, Keen CL (1989) Effects of aluminum ingestion on spontaneous motor activity of mice. Neurotoxicol Teratol 11(3):231–235
Gonzalez-Burgos I, Feria-Velasco A (2008) Serotonin/dopamine interaction in memory formation. Prog Brain Res 172:603–623
Greger JL (1993) Aluminum metabolism. Annu Rev Nutr 13(1):43–63
Habib R, Wahdan SA, Gad AM, Azab SS (2019) Infliximab abrogates cadmium-induced testicular damage and spermiotoxicity via enhancement of steroidogenesis and suppression of inflammation and apoptosis mediators. Ecotoxicol Environ Saf 182:109398
Haider S, Liaquat L, Shahzad S et al (2015) A high dose of short term exogenous d-galactose administration in young male rats produces symptoms simulating the natural aging process. Life Sci 124:110–119
Haider S, Liaquat L, Ahmad S et al (2020) Naringenin protects AlCl3/d-galactose induced neurotoxicity in rat model of AD via attenuation of acetylcholinesterase levels and inhibition of oxidative stress. PLoS ONE 15(1):e0227631
Hånell A, Marklund N (2014) Structured evaluation of rodent behavioral tests used in drug discovery research. Front Behav Neurosci 8:252–252. https://doi.org/10.3389/fnbeh.2014.00252
Hauser TU, Eldar E, Purg N, Moutoussis M, Dolan RJ (2019) Distinct roles of dopamine and noradrenaline in incidental memory. J Neurosci 39(39):7715–7721
Iqbal G, Ahmed T (2019) Co-exposure of metals and high fat diet causes aging like neuropathological changes in non-aged mice brain. Brain Res Bull 147:148–158
Jadhav R, Kulkarni YA (2023) Neuroprotective effect of quercetin and memantine against AlCl3-induced neurotoxicity in albino Wistar rats. Molecules 28(1):417
Justin Thenmozhi A, William Raja TR, Manivasagam T, Janakiraman U, Essa MM (2017) Hesperidin ameliorates cognitive dysfunction, oxidative stress and apoptosis against aluminium chloride induced rat model of Alzheimer’s disease. Nutr Neurosci 20(6):360–368
Karim N, Khan I, Abdelhalim A, Abdel-Halim H, Hanrahan JR (2017) Molecular docking and antiamnesic effects of nepitrin isolated from Rosmarinus officinalis on scopolamine-induced memory impairment in mice. Biomed Pharmacother 96:700–709
Kincheski GC, Valentim IS, Clarke JR et al (2017) Chronic sleep restriction promotes brain inflammation and synapse loss, and potentiates memory impairment induced by amyloid-β oligomers in mice. Brain Behav Immun 64:140–151
Krewski D, Yokel RA, Nieboer E et al (2007) Human health risk assessment for aluminium, aluminium oxide, and aluminium hydroxide. J Toxicol Environ Health B 10(S1):1–269
Kuc K, Gregersen B, Gannon K, Dodart JC (2006) Holeboard discrimination learning in mice. Genes Brain Behav 5(4):355–363
Kumar S (2002) Aluminium-induced changes in the rat brain serotonin system. Food Chem Toxicol 40(12):1875–1880
Kumar MR, Reddy GR (2018) Influence of age on arsenic-induced behavioral and cholinergic perturbations: amelioration with zinc and α-tocopherol. Hum Exp Toxicol 37(3):295–308
Laabbar W, Abbaoui A, Elgot A et al (2021) Aluminum induced oxidative stress, astrogliosis and cell death in rat astrocytes, is prevented by curcumin. J Chem Neuroanat 112:101915
Lee HS, Ghetti A, Pinto-Duarte A et al (2014) Astrocytes contribute to gamma oscillations and recognition memory. Proc Natl Acad Sci U S A 111(32):E3343–E3352
Lee J-S, Kim H-G, Lee H-W et al (2015) Hippocampal memory enhancing activity of pine needle extract against scopolamine-induced amnesia in a mouse model. Sci Rep 5(1):9651
Li S, Zhang Q, Ding Y, Wang X, Liu P (2020) Flavonoids ameliorate aluminum chloride-induced learning and memory impairments via suppression of apoptosis and oxidative stress in rats. J Inorg Biochem 212:111252
Liaquat L, Sadir S, Batool Z et al (2019) Acute aluminum chloride toxicity revisited: study on DNA damage and histopathological, biochemical and neurochemical alterations in rat brain. Life Sci 217:202–211
Liu W, Liu J, Gao J, Duan X, Zhang L (2022) Effects of subchronic aluminum exposure on learning, memory, and neurotrophic factors in rats. Neurotox Res 40(6):2046–2060
Ma J, Jiang G, Zheng W, Zhang M (2019) A longitudinal assessment of aluminum contents in foodstuffs and aluminum intake of residents in Tianjin metropolis. Food Sci Nutr 7(3):997–1003
Magalingam KB, Radhakrishnan A, Ping NS, Haleagrahara N (2018) Current concepts of neurodegenerative mechanisms in Alzheimer’s disease. Biomed Res Int 2018:3740461
McIlwain KL, Merriweather MY, Yuva-Paylor LA, Paylor R (2001) The use of behavioral test batteries: effects of training history. Physiol Behav 73(5):705–717
Mehpara Farhat S, Mahboob A, Ahmed T (2019) Oral exposure to aluminum leads to reduced nicotinic acetylcholine receptor gene expression, severe neurodegeneration and impaired hippocampus dependent learning in mice. Drug Chem Toxicol 44(3):310–318. https://doi.org/10.1080/01480545.2019.1587452
Memudu AE, Adanike RP (2022) Alpha lipoic acid reverses scopolamine-induced spatial memory loss and pyramidal cell neurodegeneration in the prefrontal cortex of Wistar rats. IBRO Neurosci Rep 13:1–8
Minigalieva IA, Katsnelson BA, Privalova LI et al (2018) Combined subchronic toxicity of aluminum(III), titanium(IV) and silicon(IV) oxide nanoparticles and its alleviation with a complex of bioprotectors. Int J Mol Sci 19(3):837
Nagasawa K, Ito S, Kakuda T et al (2005) Transport mechanism for aluminum citrate at the blood–brain barrier: kinetic evidence implies involvement of system Xc− in immortalized rat brain endothelial cells. Toxicol Lett 155(2):289–296
Pekny M, Pekna M (2014) Astrocyte reactivity and reactive astrogliosis: costs and benefits. Physiol Rev 94(4):1077–1098
Portero-Tresserra M, Martí-Nicolovius M, Tarrés-Gatius M, Candalija A, Guillazo-Blanch G, Vale-Martínez A (2018) Intra-hippocampal d-cycloserine rescues decreased social memory, spatial learning reversal, and synaptophysin levels in aged rats. Psychopharmacology 235:1463–1477
Qiang W, Cai W, Yang Q et al (2018) Artemisinin B improves learning and memory impairment in AD dementia mice by suppressing neuroinflammation. Neuroscience 395:1–12
Saeed A, Qusti SY, Almarwani RH et al (2021) Effects of aluminum chloride and coenzyme Q10 on the molecular structure of lipids and the morphology of the brain hippocampus cells. RSC Adv 11(48):29925–29933
Saiyed SM, Yokel RA (2005) Aluminium content of some foods and food products in the USA, with aluminium food additives. Food Addit Contam 22(3):234–244
Sanchez-Ramos J, Song S, Sava V et al (2009) Granulocyte colony stimulating factor decreases brain amyloid burden and reverses cognitive impairment in Alzheimer’s mice. Neuroscience 163(1):55–72
Saritha S, Davuljigari CB, Kumar KP, Reddy GR (2019) Effects of combined arsenic and lead exposure on the brain monoaminergic system and behavioral functions in rats: reversal effect of MiADMSA. Toxicol Ind Health 35(2):89–108
Sinha AK (1972) Colorimetric assay of catalase. Anal Biochem 47(2):389–394. https://doi.org/10.1016/0003-2697(72)90132-7
Souza MF, Medeiros KAAL, Lins LCRF et al (2022) Motor, memory, and anxiety-like behavioral impairments associated with brain-derived neurotrophic factor and dopaminergic imbalance after inhalational exposure to deltamethrin. Brain Res Bull 181:55–64
Suryavanshi J, Prakash C, Sharma D (2022) Asiatic acid attenuates aluminium chloride-induced behavioral changes, neuronal loss and astrocyte activation in rats. Metab Brain Dis 37(6):1773–1785
Tampellini D, Capetillo-Zarate E, Dumont M et al (2010) Effects of synaptic modulation on β-amyloid, synaptophysin, and memory performance in Alzheimer’s disease transgenic mice. J Neurosci 30(43):14299–14304
Tomljenovic L (2011) Aluminum and Alzheimer’s disease: after a century of controversy, is there a plausible link? J Alzheimers Dis 23(4):567–598
Üçel Uİ, Can ÖD, Özkay ÜD, Ulupinar E (2020) Antiamnesic effects of tofisopam against scopolamine-induced cognitive impairments in rats. Pharmacol Biochem Behav 190:172858
Verma S, Ranawat P, Nehru B (2020) Studies on the neuromodulatory effects of Ginkgo biloba on alterations in lipid composition and membrane integrity of rat brain following aluminium neurotoxicity. Neurochem Res 45(9):2143–2160
Walton JR (2007) A longitudinal study of rats chronically exposed to aluminum at human dietary levels. Neurosci Lett 412(1):29–33
White DN, Stowell MHB (2021) Room for two: the synaptophysin/synaptobrevin complex. Front Synaptic Neurosci 13:740318
Wilhelmsson U, Pozo-Rodrigalvarez A, Kalm M et al (2019) The role of GFAP and vimentin in learning and memory. Biol Chem 400(9):1147–1156
Wolf A, Bauer B, Abner EL, Ashkenazy-Frolinger T, Hartz AM (2016) A comprehensive behavioral test battery to assess learning and memory in 129S6/Tg2576 mice. PLoS ONE 11(1):e0147733
Wong WWK, Chung SWC, Kwong KP, Yin Ho Y, Xiao Y (2010) Dietary exposure to aluminium of the Hong Kong population. Food Addit Contam 27(4):457–463
World Health Organisation (2004) Guidelines for drinking-water quality, vol 1. World Health Organization, Geneva
World Health Organization (1997) International programme on chemical safety. Environmental Health Criteria, Geneva, p 194
World Health Organization (2003) Aluminium in drinking-water: background document for development of WHO Guidelines for drinking-water quality. World Health Organization, Geneva
Xu L, Long J, Su Z et al (2019) Restored presynaptic synaptophysin and cholinergic inputs contribute to the protective effects of physical running on spatial memory in aged mice. Neurobiol Dis 132:104586
Yan D, Jin C, Cao Y et al (2017) Effects of aluminium on long-term memory in rats and on SIRT 1 mediating the transcription of CREB-dependent gene in hippocampus. Basic Clin Pharmacol Toxicol 121(4):342–352
Yang J-Y, Wang J, Hu Y et al (2023) Paeoniflorin improves cognitive dysfunction, restores glutamate receptors, attenuates gliosis and maintains synaptic plasticity in cadmium-intoxicated mice. Arab J Chem 16(1):104406
Yasui M, Kihira T, Ota K (1992) Calcium, magnesium and aluminum concentrations in Parkinson’s disease. Neurotoxicology 13(3):593–600
Yin S, Ran Q, Yang J, Zhao Y, Li C (2020) Nootropic effect of neferine on aluminium chloride-induced Alzheimer’s disease in experimental models. J Biochem Mol Toxicol 34(2):e22429
Yokel RA (2000) The toxicology of aluminum in the brain: a review. Neurotoxicology 21(5):813–828
Zhang L, Zhao Q, Chen C-H, Qin Q-Z, Zhou Z, Yu Z-P (2014) Synaptophysin and the dopaminergic system in hippocampus are involved in the protective effect of rutin against trimethyltin-induced learning and memory impairment. Nutr Neurosci 17(5):222–229
Zhang J, Huang W, Xu F, Cao Z, Jia F, Li Y (2020) Iron dyshomeostasis participated in rat hippocampus toxicity caused by aluminum chloride. Biol Trace Elem Res 197:580–590
Zhong W, Wang L, Fang S et al (2020) Two novel colorimetric fluorescent probes: Hg2+ and Al3+ in the visual colorimetric recognition environment. RSC Adv 10(6):3048–3059
Acknowledgements
We like to express our sincere gratitude to Saba Mehak Zahoor (MSc Neurosciences), King’s College London, UK, for her valuable assistance in proofreading and English language editing. We like to acknowledge Atta-ur-Rahman School of Applied Biosciences (ASAB), National University of Sciences and Technology (NUST), Pakistan, and Higher Education Commission (HEC) of Pakistan for providing support and facilities for this study. First author is recipient of Indigenous 5000 PhD Scholarship program, HEC. HEC is not involved in study design, data interpretation, writing of manuscript, and decision to submit article for publication.
Funding
Authors declare no finding source.
Author information
Authors and Affiliations
Contributions
Humna Asghar: Data curation, Writing - original draft, Investigation, Methodology, Data interpretation, Writing - review & editing. Alveena Siddiqui, Laraib Batool, Zehra Batool: Methodology and Data interpretation. Touqeer Ahmed: Conceptualization, Investigation, Supervision, Writing - review & editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial or non-financial interests to disclose.
Ethical approval
All experimental procedures were approved by an Internal Review Board. Approval number: IRB-135.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Asghar, H., Siddiqui, A., Batool, L. et al. Post-exposure self-recovery reverses oxidative stress, ameliorates pathology and neurotransmitters imbalance and rescues spatial memory after time-dependent aluminum exposure in rat brain. Biometals (2024). https://doi.org/10.1007/s10534-023-00570-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10534-023-00570-1