Abstract
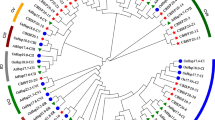
CMT2 and cpSRP43 are key factors in tolerance to heat stress, which have been revealed in Arabidopsis. It is largely unknown whether they play similar roles in heat stress resistance in monocotyledon species. Here we carried out genome-enabled identification of CHROMO domain genes in five monocots, to which both CMT2 and cpSRP43 belong. Results showed that gene copy numbers are similar in monocots to those in Arabidopsis. Phylogenetic analyses split the family into six major clades, among which CMT2 and cpSRP43 formed independent clades, respectively. Two CMT2s are present in the maize genome, while a single cpSRP43 exists in maize. Expression of the CTM2 homologs was elevated 2 and 48 h after heat stress in maize roots, while that of cpSRP43 was increased 2 and 48 h after stress in maize stalks. The genes identified here can be used as molecular targets for breeders to select for thermal-tolerant wheat lines, while phylogenetic and expression analyses of the CHROMO domain genes can be used as a knowledge base for molecular, biochemical and physiological characterization of the genes in monocots.



Similar content being viewed by others
REFERENCES
D’Hont, A., Denoeud, F., Aury, J.M., Baurens, F.C., Carreel, F., Garsmeur, O., Noel, B., Bocs, S., Droc, G., Rouard, M., Da Silva, C., Jabbari, K., Cardi, C., Poulain, J., Souquet, M., Labadie, K., Jourda, C., Lengelle, J., Rodier-Goud, M., Alberti, A., Bernard, M., Correa, M., Ayyampalayam, S., McKain, M.R., Leebens-Mack, J., Burgess, D., Freeling, M., Mbeguie, A.M.D., Chabannes, M., Wicker, T., Panaud, O., Barbosa, J., Hribova, E., Heslop-Harrison, P., Habas, R., Rivallan, R., Francois, P., Poiron, C., Kilian, A., Burthia, D., Jenny, C., Bakry, F., Brown, S., Guignon, V., Kema, G., Dita, M., Waalwijk, C., Joseph, S., Dievart, A., Jaillon, O., Leclercq, J., Argout, X., Lyons, E., Almeida, A., Jeridi, M., Dolezel, J., Roux, N., Risterucci, A.M., Weissenbach, J., Ruiz, M., Glaszmann, J.C., Quetier, F., Yahiaoui, N., and Wincker, P., The banana (Musa acuminata) genome and the evolution of monocotyledonous plants, Nature, 2012, vol. 488, pp. 213–217. https://doi.org/10.1038/nature11241
Dubin, M.J., Zhang, P., Meng, D., Remigereau, M.-S., Osborne, E.J., Paolo Casale, F., Drewe, P., Kahles, A., Jean, G., Vilhjálmsson, B., Jagoda, J., Irez, S., Voronin, V., Song, Q., Long, Q., Rätsch, G., Stegle, O., Clark, R.M., and Nordborg, M., DNA methylation in Arabidopsis has a genetic basis and shows evidence of local adaptation, eLife, 2015, vol. 4, p. e05255. https://doi.org/10.7554/eLife.05255
Edgar, R.C., MUSCLE: multiple sequence alignment with high accuracy and high throughput, Nucleic Acids Res., 2004, vol. 32, pp. 1792–1797. https://doi.org/10.1093/nar/gkh340
Eissenberg, J.C., Molecular biology of the chromo domain: an ancient chromatin module comes of age, Gene, 2001, vol. 275, pp. 19–29. https://doi.org/10.1016/s0378-1119(01)00628-x
Finn, R.D., Bateman, A., Clements, J., Coggill, P., Eberhardt, R.Y., Eddy, S.R., Heger, A., Hetherington, K., Holm, L., Mistry, J., Sonnhammer, E.L., Tate, J., and Punta, M., Pfam: the protein families database, Nucleic Acids Res, 2014, vol. 42, pp. D222–D230. https://doi.org/10.1093/nar/gkt1223
Finn, R.D., Clements, J., and Eddy, S.R., HMMER web server: interactive sequence similarity searching, Nucleic Acids Res, 2011, vol. 39, pp. W29–W37. https://doi.org/10.1093/nar/gkr367
Gallego-Bartolome, J., DNA methylation in plants: mechanisms and tools for targeted manipulation, New Phytol., 2020, vol. 227, pp. 38–44. https://doi.org/10.1111/nph.16529
Guindon, S., Delsuc, F., Dufayard, J.F., and Gascuel, O., Estimating maximum likelihood phylogenies with PhyML, Methods Mol. Biol., 2009, vol. 537, pp. 113–137. https://doi.org/10.1007/978-1-59745-251-9_6
He, J., Jiang, Z., Gao, L., You, C., Ma, X., Wang, X., Xu, X., Mo, B., Chen, X., and Liu, L., Genome-wide transcript and small RNA profiling reveals transcriptomic responses to heat stress, Plant Physiol., 2019, vol. 181, pp. 609–629. https://doi.org/10.1104/pp.19.00403
Henikoff, S. and Comai, L., A DNA methyltransferase homolog with a chromodomain exists in multiple polymorphic forms in Arabidopsis, Genetics, 1998, vol. 149, pp. 307–318. https://doi.org/10.1093/genetics/149.1.307
International Brachypodium I, Genome sequencing and analysis of the model grass Brachypodium distachyon, Nature, 2010, vol. 463, pp. 763–768. https://doi.org/10.1038/nature08747
International Rice Genome Sequencing P, The map-based sequence of the rice genome, Nature, 2005, vol. 436, pp. 793–800. https://doi.org/10.1038/nature03895
Jaru-Ampornpan, P., Liang, F.C., Nisthal, A., Nguyen, T.X., Wang, P., Shen, K., Mayo, S.L., and Shan, S.O., Mechanism of an ATP-independent protein disaggregase: II. Distinct molecular interactions drive multiple steps during aggregate disassembly, J. Biol. Chem., 2013, vol. 288, pp. 13431–13445. https://doi.org/10.1074/jbc.M113.462861
Jaru-Ampornpan, P., Shen, K., Lam, V.Q., Ali, M., Doniach, S., Jia, T.Z., and Shan, S.O., ATP-independent reversal of a membrane protein aggregate by a chloroplast SRP subunit, Nat. Struct. Mol. Biol., 2010, vol. 17, pp. 696–702. https://doi.org/10.1038/nsmb.1836
Ji, S., Siegel, A., Shan, S.O., Grimm, B., and Wang, P., Chloroplast SRP43 autonomously protects chlorophyll biosynthesis proteins against heat shock, Nat. Plants, 2021, vol. 7, pp. 1420–1432. https://doi.org/10.1038/s41477-021-00994-y
Jing, Y., Guo, Q., and Lin, R., The chromatin-remodeling factor PICKLE antagonizes Polycomb repression of FT to promote flowering, Plant Physiol., 2019, vol. 181, pp. 656–668. https://doi.org/10.1104/pp.19.00596
Kawahara, Y., de la Bastide, M., Hamilton, J.P., Kanamori, H., McCombie, W.R., Ouyang, S., Schwartz, D.C., Tanaka, T., Wu, J., Zhou, S., Childs, K.L., Davidson, R.M., Lin, H., Quesada-Ocampo, L., Vaillancourt, B., Sakai, H., Lee, S.S., Kim, J., Numa, H., Itoh, T., Buell, C.R., and Matsumoto, T., Improvement of the Oryza sativa Nipponbare reference genome using next generation sequence and optical map data, Rice, 2013, vol. 6, p. 4. https://doi.org/10.1186/1939-8433-6-4
Kumar, S., Suleski, M., Craig, J.M., Kasprowicz, A.E., Sanderford, M., Li, M., Stecher, G., and Hedges, S.B., TimeTree 5: an expanded resource for species divergence times, Mol. Biol. Evol., 2022, vol. 39. https://doi.org/10.1093/molbev/msac174
Li, Z. and Howell, S.H., Heat stress responses and thermotolerance in maize, Int. J. Mol. Sci., 2021, vol. 22. https://doi.org/10.3390/ijms22020948
Li, Z., Tang, J., Srivastava, R., Bassham, D.C., and Howell, S.H., The transcription factor bZIP60 links the unfolded protein response to the heat stress response in maize, Plant Cell, 2020, vol. 32, pp. 3559–3575
Liu, S., de Jonge, J., Trejo-Arellano, M.S., Santos-Gonzalez, J., Kohler, C., and Hennig, L., Role of H1 and DNA methylation in selective regulation of transposable elements during heat stress, New Phytol., 2021, vol. 229, pp. 2238–2250. https://doi.org/10.1111/nph.17018
Liu, Y., Yang, X., Zhou, M., Yang, Y., Li, F., Yan, X., Zhang, M., Wei, Z., Qin, S., and Min, J., Structural basis for the recognition of methylated histone H3 by the Arabidopsis LHP1 chromodomain, J. Biol. Chem., 2022, vol. 298, p. 101623. https://doi.org/10.1016/j.jbc.2022.101623
Lizaso, J., Ruiz-Ramos, M., Rodríguez, L., Gabaldon-Leal, C., Oliveira, J., Lorite, I., Sánchez, D., García, E., and Rodríguez, A., Impact of high temperatures in maize: phenology and yield components, Field Crops Res., 2018, vol. 216, pp. 129–140.
Lobell, D.B., Bänziger, M., Magorokosho, C., and Vivek, B., Nonlinear heat effects on African maize as evidenced by historical yield trials, Nat. Climate Change, 2011, vol. 1, pp. 42–45. https://doi.org/10.1038/nclimate1043
Nguyen, T.X., Jaru-Ampornpan, P., Lam, V.Q., Cao, P., Piszkiewicz, S., Hess, S., and Shan, S.O., Mechanism of an ATP-independent protein disaggregase: I. Structure of a membrane protein aggregate reveals a mechanism of recognition by its chaperone, J. Biol. Chem., 2013, vol. 288, pp. 13420–13430. https://doi.org/10.1074/jbc.M113.462812
Nozawa, K., Masuda, S., Saze, H., Ikeda, Y., Suzuki, T., Takagi, H., Tanaka, K., Ohama, N., Niu, X., Kato, A., and Ito, H., Regulation of ecotype-specific expression of the heat-activated transposon ONSEN, Front. Plant Sci., 2022, vol. 13, p. 899105. https://doi.org/10.3389/fpls.2022.899105
Park, J., Oh, D.H., Dassanayake, M., Nguyen, K.T., Ogas, J., Choi, G., and Sun, T.P., Gibberellin signaling requires chromatin remodeler PICKLE to promote vegetative growth and phase transitions, Plant Physiol., 2017, vol. 173, pp. 1463–1474. https://doi.org/10.1104/pp.16.01471
Reiser, L., Subramaniam, S., Zhang, P., and Berardini, T., Using the Arabidopsis Information Resource (TAIR) to find information about Arabidopsis genes, Curr. Protoc., 2022, vol. 2, p. e574. https://doi.org/10.1002/cpz1.574
Sang, Q., Pajoro, A., Sun, H., Song, B., Yang, X., Stolze, S.C., Andres, F., Schneeberger, K., Nakagami, H., and Coupland, G., Mutagenesis of a quintuple mutant impaired in environmental responses reveals roles for CHROMATIN REMODELING4 in the Arabidopsis floral transition, Plant Cell, 2020, vol. 32, pp. 1479–1500. https://doi.org/10.1105/tpc.19.00992
Sasaki, E., Kawakatsu, T., Ecker, J.R., and Nordborg, M., Common alleles of CMT2 and NRPE1 are major determinants of CHH methylation variation in Arabidopsis thaliana, PLoS Genet., 2019, vol. 15, p. e1008492. https://doi.org/10.1371/journal.pgen.1008492
Schuenemann, D., Gupta, S., Persello-Cartieaux, F., Klimyuk, V.I., Jones, J.D., Nussaume, L., and Hoffman, N.E., A novel signal recognition particle targets light-harvesting proteins to the thylakoid membranes, Proc. Natl. Acad. Sci. U. S. A., 1998, vol. 95, pp. 10312–10316. https://doi.org/10.1073/pnas.95.17.10312
Shen, X., De Jonge, J., Forsberg, S.K., Pettersson, M.E., Sheng, Z., Hennig, L., and Carlborg, O., Natural CMT2 variation is associated with genome-wide methylation changes and temperature seasonality, PLoS Genet., 2014, vol. 10, p. e1004842. https://doi.org/10.1371/journal.pgen.1004842
Shen, Y., Devic, M., Lepiniec, L., and Zhou, D.X., Chromodomain, helicase and DNA-binding CHD1 protein, CHR5, are involved in establishing active chromatin state of seed maturation genes, Plant Biotechnol. J., 2015, vol. 13, pp. 811–820. https://doi.org/10.1111/pbi.12315
Van Buren, R., Bryant, D., Edger, P.P., Tang, H., Burgess, D., Challabathula, D., Spittle, K., Hall, R., Gu, J., Lyons, E., Freeling, M., Bartels, D., Ten Hallers, B, Hastie, A., Michael, T.P., and Mockler, T.C., Single-molecule sequencing of the desiccation-tolerant grass Oropetium thomaeum, Nature, 2015 vol. 527, pp. 508–511. https://doi.org/10.1038/nature15714
Wang, P. and Grimm, B., Comparative analysis of light-harvesting antennae and state transition in chlorina and cpSRP mutants, Plant Physiol., 2016, vol. 172, pp. 1519–1531. https://doi.org/10.1104/pp.16.01009
Wang, P., Liang, F.C., Wittmann, D., Siegel, A., Shan, S.O., and Grimm, B., Chloroplast SRP43 acts as a chaperone for glutamyl-tRNA reductase, the rate-limiting enzyme in tetrapyrrole biosynthesis, Proc. Natl. Acad. Sci. U. S. A., 2018, vol. 115, pp. E3588–E3596. https://doi.org/10.1073/pnas.1719645115
Yan, B., Lv, Y., Zhao, C., and Wang, X., Knowing when to silence: roles of polycomb-group proteins in SAM maintenance, root development, and developmental phase transition, Int. J. Mol. Sci., 2020, vol. 21. https://doi.org/10.3390/ijms21165871
Yates, A.D., Allen, J., Amode, R.M., Azov, A.G., Barba, M., Becerra, A., Bhai, J., Campbell, L.I., Carbajo Martinez, M., Chakiachvili, M., Chougule, K., Christensen, M., Contreras-Moreira, B., Cuzick, A., Da Rin Fioretto, L., Davis, P., De Silva, N.H., Diamantakis, S., Dyer, S., Elser, J., Filippi, C.V., Gall, A., Grigoriadis, D., Guijarro-Clarke, C., Gupta, P., Hammond-Kosack, K.E., Howe, K.L., Jaiswal, P., Kaikala, V., Kumar, V., Kumari, S., Langridge, N., Le, T., Luypaert, M., Maslen, G.L., Maurel, T., Moore, B., Muffato, M., Mushtaq, A., Naamati, G., Naithani, S., Olson, A., Parker, A., Paulini, M., Pedro, H., Perry, E., Preece, J., Quinton-Tulloch, M., Rodgers, F., Rosello, M., Ruffier, M., Seager, J., Sitnik, V., Szpak, M., Tate, J., Tello-Ruiz, M.K., Trevanion, S.J., Urban, M., Ware, D., Wei, S., Williams, G., Winterbottom, A., Zarowiecki, M., Finn, R.D., and Flicek, P., Ensembl Genomes 2022: an expanding genome resource for non-vertebrates, Nucleic Acids Res., 2022, vol. 50, pp. D996–D1003. https://doi.org/10.1093/nar/gkab1007
Yu, Y., Zhang, H., Long, Y., Shu, Y., and Zhai, J., Plant Public RNA-seq Database: a comprehensive online database for expression analysis of ~45 000 plant public RNA-Seq libraries, Plant Biotechnol. J., 2022, vol. 20, pp. 806–808. https://doi.org/10.1111/pbi.13798
Zhang, H., Lang, Z., and Zhu, J.K., Dynamics and function of DNA methylation in plants, Nat. Rev. Mol. Cell Biol., 2018a, vol. 19, pp. 489–506. https://doi.org/10.1038/s41580-018-0016-z
Zhang, Y., Harris, C.J., Liu, Q., Liu, W., Ausin, I., Long, Y., Xiao, L., Feng, L., Chen, X., Xie, Y., Chen, X., Zhan, L., Feng, S., Li, J.J., Wang, H., Zhai, J., and Jacobsen, S.E., Large-scale comparative epigenomics reveals hierarchical regulation of non-CG methylation in Arabidopsis, Proc. Natl. Acad. Sci. U. S. A., 2018b, vol. 115, pp. E1069–E1074. https://doi.org/10.1073/pnas.1716300115
Zhang, Y., Wendte, J.M., Ji, L., and Schmitz, R.J., Natural variation in DNA methylation homeostasis and the emergence of epialleles, Proc. Natl. Acad. Sci. U. S. A., 2020, vol. 117, pp. 4874–4884. https://doi.org/10.1073/pnas.1918172117
Zou, B., Sun, Q., Zhang, W., Ding, Y., Yang, D.L., Shi, Z., and Hua, J., The Arabidopsis chromatin-remodeling factor CHR5 regulates plant immune responses and nucleosome occupancy, Plant Cell Physiol., 2017, vol. 58, pp. 2202–2216. https://doi.org/10.1093/pcp/pcx155
Funding
This work is supported by Special Funds for Key Research Development and Promotion of Henan Province (222102110231) and the Key Research and Development Projects of Henan Province (231111113000).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This work does not contain any studies involving human and animal subjects.
CONFLICT OF INTEREST
The authors of this work declare that they have no conflicts of interest.
Additional information
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pengfei Duan, Kan, Y., Zhao, D. et al. Genome-Wide Identification and Characterization of CHROMO Domain Family Genes Reveal Roles of the Maize Genes in Heat Stress Response. Biol Bull Russ Acad Sci 50 (Suppl 3), S289–S297 (2023). https://doi.org/10.1134/S1062359022603494
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1062359022603494