Abstract
Purpose of the study
Posterior cortical atrophy is a clinico-radiographical syndrome that presents with higher-order visual dysfunction and is most commonly due to Alzheimer’s disease. Understanding factors associated with atypical presentations of Alzheimer’s disease, such as posterior cortical atrophy (PCA), holds promise to shape our understanding of AD pathophysiology. Thus, we aimed to compare MRI evidence of lobar microbleeds (LMBs) in posterior cortical atrophy (PCA) syndrome to typical AD (tAD) and to assess and compare MRI evidence of cerebral amyloid angiopathy (CAA) in each group.
Findings
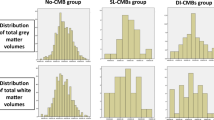
We retrospectively collected clinical and MRI data from participants with PCA (n = 26), identified from an institutional PCA registry, and participants with tAD (n = 46) identified from electronic health records from a single institution. LMBs were identified on susceptibility-weighted imaging (SWI); the Fazekas grade of white matter disease was assessed using FLAIR images, and Boston criteria version 2.0 for cerebral amyloid angiopathy were applied to all data. The proportion of participants with PCA and LMB (7.7%) was lower than for tAD (47.8%) (p = 0.005). The frequency of “probable” CAA was similar in both groups, while “possible” CAA was more frequent in tAD (30.4%) than PCA (0%) (p = 0.001). The Fazekas grades were not different between groups.
Summary
Lobar microbleeds on SWI were not more common in PCA than in typical AD. Clinicopathological investigations are necessary to confirm these findings. The factors that contribute to the posterior cortical atrophy phenotype are unknown.

Similar content being viewed by others
Data Availability
Data available upon reasonable request.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance:
•• Crutch SJ, Schott JM, Rabinovici GD, et al. Consensus classification of posterior cortical atrophy. Alzheimers Dement. 2017;13:870–84. https://doi.org/10.1016/j.jalz.2017.01.014. This manuscript outlines the consensus criteria for the diagnosis of PCA and provides a framework for the incorporation of biomarkers for the underlying disease.
Graff-Radford J, Yong KXX, Apostolova LG, et al. New insights into atypical Alzheimer’s disease in the era of biomarkers. Lancet Neurol. 2021;20:222–34. https://doi.org/10.1016/S1474-4422(20)30440-3.
•• Charidimou A, Boulouis G, Frosch MP, et al. The Boston criteria version 2.0 for cerebral amyloid angiopathy: a multicentre, retrospective. MRI-neuropathology diagnostic accuracy study Lancet Neurol. 2022;21:714–25. https://doi.org/10.1016/S1474-4422(22)00208-3. The authors update the Boston criteria for cerebral amyloid angiopathy. Criteria incorporate white matter changes on MRI and provide.
Reijmer YD, Fotiadis P, Charidimou A, et al. Relationship between white matter connectivity loss and cortical thinning in cerebral amyloid angiopathy. Hum Brain Mapp. 2017;38:3723–31. https://doi.org/10.1002/hbm.23629.
Reijmer YD, Fotiadis P, Martinez-Ramirez S, et al. Structural network alterations and neurological dysfunction in cerebral amyloid angiopathy. Brain. 2015;138:179–88. https://doi.org/10.1093/brain/awu316.
• Reijmer YD, Fotiadis P, Riley GA, et al. Progression of brain network alterations in cerebral amyloid angiopathy. Stroke. 2016;47:2470–5. https://doi.org/10.1161/STROKEAHA.116.014337The authors demonstrate that network impairment progresses over time and follows a pattern of progression going from posterior to frontal.
Pettersen JA, Sathiyamoorthy G, Gao FQ, et al. Microbleed topography, leukoaraiosis, and cognition in probable Alzheimer disease from the Sunnybrook dementia study. Arch Neurol. 2008;65:790–5. https://doi.org/10.1001/archneur.65.6.790.
Martinez-Ramirez S, Greenberg SM, Viswanathan A. Cerebral microbleeds: overview and implications in cognitive impairment. Alzheimers Res Ther. 2014;345:125–30. https://doi.org/10.1186/alzrt263.
Valenti R, Charidimou A, Xiong L, et al. Visuospatial functioning in cerebral amyloid angiopathy: a pilot study. J Alzheimers Dis. 2017;56:1223–7. https://doi.org/10.3233/JAD-160927.
Whitwell JL, Kantarci K, Weigand SD, et al. Microbleeds in atypical presentations of Alzheimer’s disease: a comparison to dementia of the Alzheimer’s type. J Alzheimers Dis. 2015;45:1109–17. https://doi.org/10.3233/JAD-142628.
Sepehry AA, Lang D, Hsiung GY, et al. Prevalence of brain microbleeds in Alzheimer disease: a systematic review and meta-analysis on the influence of neuroimaging techniques. AJNR Am J Neuroradiol. 2016;37:215–22. https://doi.org/10.3174/ajnr.A4525.
Withington CG, Turner RS. Amyloid-related imaging abnormalities with anti-amyloid antibodies for the treatment of dementia due to Alzheimer’s disease. Front Neurol. 2022;13:862369. https://doi.org/10.3389/fneur.2022.862369.
McKhann GM, Knopman DS, Chertkow H, et al. The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011;7:263–79. https://doi.org/10.1016/j.jalz.2011.03.005.
Haller S, Vernooij MW, Kuijer JPA, et al. Cerebral microbleeds: imaging and clinical significance. Radiology. 2018;287:11–28. https://doi.org/10.1148/radiol.2018170803.
Fazekas F, Chawluk JB, Alavi A, et al. MR signal abnormalities at 1.5 T in Alzheimer’s dementia and normal aging. AJR Am J Roentgenol. 1987;149:351–6. https://doi.org/10.2214/ajr.149.2.351.
Desikan RS, Segonne F, Fischl B, et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage. 2006;31:968–80. https://doi.org/10.1016/j.neuroimage.2006.01.021.
Jakel L, De Kort AM, Klijn CJM, et al. Prevalence of cerebral amyloid angiopathy: a systematic review and meta-analysis. Alzheimers Dement. 2022;18:10–28. https://doi.org/10.1002/alz.12366.
Charidimou A, Zonneveld HI, Shams S, et al. APOE and cortical superficial siderosis in CAA: meta-analysis and potential mechanisms. Neurology. 2019;93(4):e358–71. https://doi.org/10.1212/wnl.0000000000007818.
Schott JM, Crutch SJ, Carrasquillo MM, et al. Genetic risk factors for the posterior cortical atrophy variant of Alzheimer’s disease. Alzheimers Dement. 2016;12:862–71. https://doi.org/10.1016/j.jalz.2016.01.010.
Millington RS, James-Galton M, Maia Da Silva MN, et al. Lateralized occipital degeneration in posterior cortical atrophy predicts visual field deficits. Neuroimage Clin. 2017;14:242–9.
Acknowledgements
The editors would like to thank Dr. John Brust for taking the time to review this manuscript. We thank Jerri Lusk, Program Assistant I at the University of Colorado, for help with manuscript preparation and administrative support, and Jody Tanabe, MD, Professor of Radiology and Chief of Neuroradiology at the University of Colorado, for access to the typical AD cohort. We are indebted to our patients and their families and care partners.
Funding
Partial financial support for VSP was received from the University of Colorado School of Medicine (UCSOM) John and Joanne Hare Posterior Cortical Atrophy Research Fund and the UCSOM Brain and Vision Fund. BMB receives research support from NIH (NIA: R01 AG058772; R21AG072153) and the Department of Defense (W81XWH-19–1-0520). PP receives research funding from NIA R61 AG074859-01, NIA R01 AG071151-01, and NIA K23 AG063900-01A1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The following authors have no relevant financial or non-financial interests to disclose: LN, AM, SS, VK, AC, AT. VSP is a member of the editorial board of Current Neurology and Neurosciences Report. All other authors report no competing interests. Financial interests: VSP has received research support from NIH, the John and Joanne Hare Fund for Accelerating PCA Research, University of Colorado Alzheimer’s and Cognition Center, the North American Neuro-ophthalmology Society and institutional support from the Lewy Body Dementia Association as the PI for the LBDA Research Center of Excellence; VSP has also received support from the American Academy of Neurology for editorial work, royalties from Up-to-Date, Inc., as a member of the Alzheimer’s Association for the LEADS Patient Advocacy Committee, as a member of the Medical Advisory Board for Esai, and as a PI for clinical trials affiliated with Biogen and Eisai. PP has received research support from NIH, the Doris Duke Fund to Retain Clinical Scientists, and the Mary Massack Brinker Memorial Dementia Research Fund. BMB has received research support from NIH, the Department of Defense, and the University of Colorado School of Medicine. SHS has received research support from the NIH and the Alzheimer’s Association. ALC is a consultant for Eli Lilly. Non-financial interests: VSP is a member of the following boards and receives no compensation as a member: Board of Directors of the North American Neuro-ophthalmology Society and the Medical Advisory Board of the Benign Essential Blepharospasm Research Foundation. ALC is a Medical Advisory Board Member of the Spinal CSF Leak Foundation and Member for the North American Neuro-ophthalmology Society Illustrated Curriculum, Brain & Life Magazine, StatPearls Behavioral Neurology and Neuropsychiatry Section, Neurology Clinical Practice, and the Journal of Neuro-ophthalmology.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Neuro-Ophthalmology.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pelak, V.S., Krishnan, V., Serva, S. et al. Lobar Microbleeds in the Posterior Cortical Atrophy Syndrome: A Comparison to Typical Alzheimer’s Disease. Curr Neurol Neurosci Rep 24, 27–33 (2024). https://doi.org/10.1007/s11910-024-01330-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11910-024-01330-5