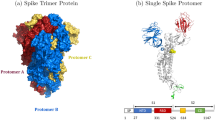
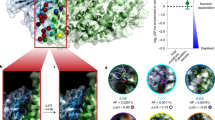
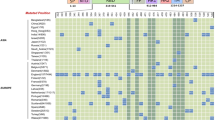
Abstract
SARS-CoV-2, the virus that causes COVID-19, led to a global health emergency that claimed the lives of millions. Despite the widespread availability of vaccines, the virus continues to exist in the population in an endemic state which allows for the continued emergence of new variants. Most of the current vaccines target the spike glycoprotein interface of SARS-CoV-2, creating a selection pressure favoring viral immune evasion. Antivirals targeting other molecular interactions of SARS-CoV-2 can help slow viral evolution by providing orthogonal selection pressures on the virus. GRP78 is a host auxiliary factor that mediates binding of the SARS-CoV-2 spike protein to human cellular ACE2, the primary pathway of cell infection. As GRP78 forms a ternary complex with SARS-CoV-2 spike protein and ACE2, disrupting the formation of this complex is expected to hinder viral entry into host cells. Here, we developed a model of the GRP78-Spike RBD-ACE2 complex. We then used that model together with hot spot mapping of the GRP78 structure to identify the putative binding site for spike protein on GRP78. Next, we performed structure-based virtual screening of known drug/candidate drug libraries to identify binders to GRP78 that are expected to disrupt spike protein binding to the GRP78, and thereby preventing viral entry to the host cell. A subset of these compounds has previously been shown to have some activity against SARS-CoV-2. The identified hits are starting points for the further development of novel SARS-CoV-2 therapeutics, potentially serving as proof-of-concept for GRP78 as a potential drug target for other viruses.








Similar content being viewed by others
Data availability
All data and material are available in the supplementary information.
References
Nicola M, Alsafi Z, Sohrabi C et al (2020) The socio-economic implications of the coronavirus pandemic (COVID-19): a review. Int J Surg 78:185–193. https://doi.org/10.1016/j.ijsu.2020.04.018
Harvey WT, Carabelli AM, Jackson B et al (2021) SARS-CoV-2 variants, spike mutations and immune escape. Nat Rev Microbiol 19:409–424. https://doi.org/10.1038/s41579-021-00573-0
Van Egeren D, Novokhodko A, Stoddard M et al (2021) Risk of rapid evolutionary escape from biomedical interventions targeting SARS-CoV-2 spike protein. PLoS ONE 16:e0250780. https://doi.org/10.1371/journal.pone.0250780
Gaurav A, Agrawal N, Al-Nema M, Gautam V (2022) Computational approaches in the discovery and development of therapeutic and prophylactic agents for viral diseases. Curr Top Med Chem 22:2190–2206. https://doi.org/10.2174/1568026623666221019110334
Sahoo BM, Bhattamisra SK, Das S et al (2022) Computational approach to combat COVID-19 infection: emerging tools for accelerating drug research. Curr Drug Discov Technol 19:e170122200314. https://doi.org/10.2174/1570163819666220117161308
Shanmugam A, Venkattappan A, Gromiha MM (2022) Structure based drug designing approaches in SARS-CoV-2 spike inhibitor design. Curr Top Med Chem 22:2396–2409. https://doi.org/10.2174/1568026623666221103091658
Mehmood A, Nawab S, Wang Y et al (2022) Discovering potent inhibitors against the Mpro of the SARS-CoV-2: a medicinal chemistry approach. Comput Biol Med 143:105235. https://doi.org/10.1016/j.compbiomed.2022.105235
Mehmood A, Nawab S, Jia G et al (2023) Supervised screening of Tecovirimat-like compounds as potential inhibitors for the monkeypox virus E8L protein. J Biomol Struct Dyn. https://doi.org/10.1080/07391102.2023.2245042
Lan J, Ge J, Yu J et al (2020) Structure of the SARS-CoV-2 spike receptor-binding domain bound to the ACE2 receptor. Nature 581:215–220. https://doi.org/10.1038/s41586-020-2180-5
Shang J, Wan Y, Luo C et al (2020) Cell entry mechanisms of SARS-CoV-2. Proc Natl Acad Sci U S A 117:11727–11734. https://doi.org/10.1073/pnas.2003138117
Papageorgiou AC, Mohsin I (2020) The SARS-CoV-2 spike glycoprotein as a drug and vaccine target: structural insights into its complexes with ACE2 and antibodies. Cells 9:2343. https://doi.org/10.3390/cells9112343
Yuen K-S, Ye Z-W, Fung S-Y et al (2020) SARS-CoV-2 and COVID-19: the most important research questions. Cell Biosci 10:40. https://doi.org/10.1186/s13578-020-00404-4
Mostafa-Hedeab G et al (2020) ACE2 as drug target of COVID-19 virus treatment, simplified updated review. rbmb.net 9:97–105. https://doi.org/10.29252/rbmb.9.1.97
Rangel HR, Ortega JT, Maksoud S et al (2020) SARS-CoV-2 host tropism: an in silico analysis of the main cellular factors. Virus Res 289:198154. https://doi.org/10.1016/j.virusres.2020.198154
Elfiky AA, Ibrahim IM, Ismail AM, Elshemey WM (2021) A possible role for GRP78 in cross vaccination against COVID-19. J Infect 82:282–327. https://doi.org/10.1016/j.jinf.2020.09.004
Carlos AJ, Ha DP, Yeh D-W et al (2021) The chaperone GRP78 is a host auxiliary factor for SARS-CoV-2 and GRP78 depleting antibody blocks viral entry and infection. J Biol Chem 296:100759. https://doi.org/10.1016/j.jbc.2021.100759
Yang J, Nune M, Zong Y et al (2015) Close and allosteric opening of the polypeptide-binding site in a human Hsp70 chaperone BiP. Structure 23:2191–2203. https://doi.org/10.1016/j.str.2015.10.012
Ibrahim IM, Abdelmalek DH, Elshahat ME, Elfiky AA (2020) COVID-19 spike-host cell receptor GRP78 binding site prediction. J Infect 80:554–562. https://doi.org/10.1016/j.jinf.2020.02.026
Dominguez C, Boelens R, Bonvin AMJJ (2003) HADDOCK: a protein−protein docking approach based on biochemical or biophysical information. J Am Chem Soc 125:1731–1737. https://doi.org/10.1021/ja026939x
van Zundert GCP, Rodrigues JPGLM, Trellet M et al (2016) The HADDOCK2.2 web server: user-friendly integrative modeling of biomolecular complexes. J Mol Biol 428:720–725. https://doi.org/10.1016/j.jmb.2015.09.014
Yang J, Zong Y, Su J et al (2017) Conformation transitions of the polypeptide-binding pocket support an active substrate release from Hsp70s. Nat Commun 8:1201. https://doi.org/10.1038/s41467-017-01310-z
Yoneda Y, Steiniger SCJ, Čapková K et al (2008) A cell-penetrating peptidic GRP78 ligand for tumor cell-specific prodrug therapy. Bioorg Med Chem Lett 18:1632–1636. https://doi.org/10.1016/j.bmcl.2008.01.060
Kim Y, Lillo AM, Steiniger SCJ et al (2006) Targeting heat shock proteins on cancer cells: selection, characterization, and cell-penetrating properties of a peptidic GRP78 ligand. Biochemistry 45:9434–9444. https://doi.org/10.1021/bi060264j
Brenke R, Kozakov D, Chuang GY, Beglov D, Hall D, Landon MR, Mattos C, Vajda S (2009) Fragment-based identification of druggable “hot spots” of proteins using Fourier domain correlation techniques. Bioinformatics. https://doi.org/10.1093/bioinformatics/btp036
Kozakov D, Grove LE, Hall DR et al (2015) The FTMap family of web servers for determining and characterizing ligand-binding hot spots of proteins. Nat Protoc 10:733–755. https://doi.org/10.1038/nprot.2015.043
Kozakov D, Hall DR, Napoleon RL et al (2015) New frontiers in druggability. J Med Chem 58:9063–9088. https://doi.org/10.1021/acs.jmedchem.5b00586
Friesner RA, Banks JL, Murphy RB et al (2004) Glide: a new approach for rapid, accurate docking and scoring—1: method and assessment of docking accuracy. J Med Chem 47:1739–1749. https://doi.org/10.1021/jm0306430
Friesner RA, Murphy RB, Repasky MP et al (2006) Extra precision glide: docking and scoring incorporating a model of hydrophobic enclosure for protein−ligand complexes. J Med Chem 49:6177–6196. https://doi.org/10.1021/jm051256o
Halgren TA, Murphy RB, Friesner RA et al (2004) Glide: a new approach for rapid, accurate docking and scoring—2: enrichment factors in database screening. J Med Chem 47:1750–1759. https://doi.org/10.1021/jm030644s
Sterling T, Irwin JJ (2015) ZINC 15—ligand discovery for everyone. J Chem Inf Model 55:2324–2337. https://doi.org/10.1021/acs.jcim.5b00559
Gaulton A, Bellis LJ, Bento AP et al (2012) ChEMBL: a large-scale bioactivity database for drug discovery. Nucleic Acids Res 40:D1100–D1107. https://doi.org/10.1093/nar/gkr777
Schrödinger release 2022–3: QikProp (2021) Schrödinger, LLC, New York
Di L, Kerns EH (2016) Drug-like properties: concepts, structure design and methods from ADME to toxicity optimization, 2nd edn. Academic Press
Duan J, Dixon SL, Lowrie JF, Sherman W (2010) Analysis and comparison of 2D fingerprints: insights into database screening performance using eight fingerprint methods. J Mol Graph Model 29:157–170. https://doi.org/10.1016/j.jmgm.2010.05.008
Sastry M, Lowrie JF, Dixon SL, Sherman W (2010) Large-scale systematic analysis of 2D fingerprint methods and parameters to improve virtual screening enrichments. J Chem Inf Model 50:771–784. https://doi.org/10.1021/ci100062n
Weston S, Coleman CM, Haupt R et al (2020) Broad anti-coronaviral activity of FDA approved drugs against SARS-CoV-2 in vitro and SARS-CoV in vivo. Microbiology 26:865
MARSH W (2007) Tricyclic antidepressants. In: Enna SJ, Bylund DB, Elsevier Science (FIRM) (eds) xPharm: the comprehensive pharmacology reference. Amsterdam: Elsevier, pp 1–3. https://doi.org/10.1016/B978-008055232-3.61066-9
Quinones QJ et al (2008) GRP78: a chaperone with diverse roles beyond the endoplasmic reticulum. Histol histopathol 23(11):1409–1416. https://doi.org/10.14670/HH-23.1409
Gurusinghe KRDSNS, Mishra A, Mishra S (2018) Glucose-regulated protein 78 substrate-binding domain alters its conformation upon EGCG inhibitor binding to nucleotide-binding domain: molecular dynamics studies. Sci Rep 8:5487. https://doi.org/10.1038/s41598-018-22905-6
Halford B (2022) How Pfizer scientists transformed an old drug lead into a COVID-19 antiviral Behind the scenes of the medicinal chemistry campaign that led to the pill Paxlovid
Focosi D, McConnell S, Shoham S et al (2023) Nirmatrelvir and COVID-19: development, pharmacokinetics, clinical efficacy, resistance, relapse, and pharmacoeconomics. Int J Antimicrob Agents 61:106708. https://doi.org/10.1016/j.ijantimicag.2022.106708
De Clercq E, Li G (2016) Approved antiviral drugs over the past 50 years. Clin Microbiol Rev 29:695–747. https://doi.org/10.1128/CMR.00102-15
Acknowledgements
We thank Kenji C. Walker for thoughtful discussions related to the properties of intranasally delivered drugs.
Funding
The research leading to these results received funding the National Science Foundation under grant NSF-2200052 to DJ-M.
Author information
Authors and Affiliations
Contributions
ML, AC and DJM are responsible for the study conception and design. ML and JRH were responsible for all computations. ML, AC, and DJM contributed to the interpretation of the results and edited the manuscript. DJM oversaw the project.
Corresponding author
Ethics declarations
Competing interest
The authors have no competing interests to declare that are relevant to the content of this article.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lazou, M., Hutton, J.R., Chakravarty, A. et al. Identification of a druggable site on GRP78 at the GRP78-SARS-CoV-2 interface and virtual screening of compounds to disrupt that interface. J Comput Aided Mol Des 38, 6 (2024). https://doi.org/10.1007/s10822-023-00546-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10822-023-00546-w