Abstract
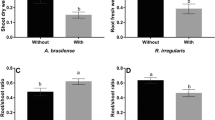
Root rot of hydroponically grown Welsh onion has caused considerable economic losses every year since commercial cultivation began in 1988 in Hiroshima Prefecture, Japan. The primary pathogens, which have not been reported to form sexual structures, are Pythium spp. including Pythium clusters B2a sp., which is most prevalent, and B1d sp. and Pythium clade A sp. These pathogens infect Welsh onion roots by producing sporangia and zoospores. The objective of this study was to elucidate the inhibitory effects of low-pH solutions on root-rot pathogens of hydroponically grown Welsh onion. Naturally occurring disease was significantly lower after treatment with nutrient solutions with a pH maintained at 4.3–4.9 than with conventional unmanaged solutions (pH 5.4–6.4). In a greenhouse after inoculation of infected Welsh onion with Pythium cluster B2a sp., low-pH solutions (pH 4.0 and 4.5) significantly suppressed zoospore production and reduced the percentage of root infections compared to the conventional solution (pH 5.5). In other greenhouse inoculation tests with Pythium clusters B2a sp. and B1d sp. and Pythium clade A sp., the low-pH solution (pH 4.0) substantially decreased zoospore density and root infection compared to the conventional solution (pH 5.5). The low-pH solutions did not negatively impact crop yield in the greenhouse tests and thus can be used to suppress root rot in hydroponically grown Welsh onions.








Similar content being viewed by others
Change history
01 April 2024
A Correction to this paper has been published: https://doi.org/10.1007/s10327-024-01173-5
References
Araujo JL, Faquin V, Baliza DP, de Ávila FW, Guerrero AC (2016) Growth and mineral nutrition of green onions grown hydroponically under different N, P and K concentrations. Rev Ceres Vicosa 63:232–240. https://doi.org/10.1590/0034-737X201663020015
Arnon DI, Johnson CM (1942) Influence of hydrogen ion concentration on the growth of higher plants under controlled conditions. Plant Physiol 17:525–539
Bates ML, Stanghellini ME (1984) Root rot of hydroponically grown spinach caused by Pythium aphanidermatum and P. dissotocum. Plant Dis 68:989–991
Blaker NS, MacDonald JD (1983) Influence of container medium pH on sporangium formation, zoospore release, and infection of rhododendron by Phytophthora cinnamomi. Plant Dis 67:259–263
Cao Y, Li Y, Li J, Wang L, Cheng Z, Wang H, Fan Z, Li H (2016) Rapid and quantitative detection of Pythium inflatum by real-time fluorescence loop-mediated isothermal amplification assay. Eur J Plant Pathol 144:83–95. https://doi.org/10.1007/s10658-015-0752-2
Dimova MA, Büttner C, Gablers J, Grosch R, Bar-Yosef B, Kläring HP (2010) Control of root zone pH is not effective in preventing Pythium aphanidermatum disease in cucumber. J Plant Dis Prot 117:244–247. https://doi.org/10.1007/BF03356368
Favrin RJ, Rahe JE, Mauza B (1988) Pythium spp. associated with crown rot of cucumbers in British Columbia greenhouses. Plant Dis 72:683–687
Garibaldi A, Gilardi G, Matic S, Gullino ML (2017) First report of stem rot caused by a Pythium cluster B2a species on lettuce in Italy. Plant Dis 101:1681. https://doi.org/10.1094/PDIS-01-17-0124-PDN
NARO Genebank (2023) Database of plant diseases in Japan. https://www.gene.affrc.go.jp/databases-micro_pl_diseases.php. Accessed 30 Oct 2023
Gillespie PG, Kubota C, Miller SA (2020) Effects of low pH of hydroponic nutrient solution on plant growth, nutrient uptake, and root rot disease incidence of basil (Ocimum basilicum L.). Hort Sci 55:1251–1258. https://doi.org/10.21273/HORTSCI14986-20
Gold SE, Stanghellini ME (1985) Effects of temperature on Pythium root rot of spinach grown under hydroponic conditions. Phytopathology 75:333–337
Hickman CJ, Ho HH (1966) Behavior of zoospores in plant-pathogenic Phycomycetes. Annu Rev Phytopathol 4:195–220
Huang R, Tu J (1998) Effects of hydrogen ion concentration and temperature on attachment of zoospores of Pythium aphanidermatum to tomato roots. Meded-Fac Landbouwkd Univ Gent 63:855–859
Kong P, Moorman GW, Lea-Cox JD, Ross DS, Richardson PA, Hong C (2009) Zoosporic tolerance to pH stress and its implications for Phytophthora species in aquatic ecosystems. Appl Environ Microbiol 75:4307–4314. https://doi.org/10.1128/AEM.00119-09
Krasnow CS, Hausbeck MK (2017) Influence of pH and etridiazole on Pythium species. HortTechnology 27:367–374. https://doi.org/10.21273/HORTTECH03633-16
Kusakari S, Okada K, Kawaradani M (1996) Dispersal of spinach (Spinacia oleracea) damping-off caused by Pythium aphanidermatum and Pythium ultimum in hydroponics (in Japanese). Kinki Chugoku Agric Res 92:3–7
Lévesque CA, De Cock AWAM (2004) Molecular phylogeny and taxonomy of the genus Pythium. Mycol Res 108:1363–1383. https://doi.org/10.1017/S0953756204001431
Miller PM (1955) V-8 juice agar as a general-purpose medium for fungi and bacteria. Phytopathology 45:461–462
Morita Y, Tojo M (2007) Modifications of PARP medium using fluazinam, miconazole, and nystatin for detection of Pythium spp. in soil. Plant Dis 91:1591–1599. https://doi.org/10.1094/PDIS-91-12-1591
Nagashima S, You X, Uzuhashi S, Tojo M (2021) Stem and root rot in hydrangea caused by Pythium myriotylum and Globisporangium spp. and susceptibility of hydrangea cultivars. J Gen Plant Pathol 87:148–153. https://doi.org/10.1007/s10327-021-00989-9
Ochi M, Okano H, Nakamura K, Yokoyama N, Hashimoto K, Abe T, Furukawa N, Imai S (2012) Study of cultivation method using reusable medium to Welsh onion in hydroponic culture (in Japanese). Hiroshima Pref Tech Res Inst Agr Res Cent Rep 88:15–20
Pantelides IS, Tsolakidou M-D, Chrysargyris A, Tzortzakis N (2017) First report of root rot of hydroponically grown lettuce (Lactuca sativa) caused by a Pythium species from the cluster B2a species complex in Cyprus. Plant Dis 101:636. https://doi.org/10.1094/PDIS-07-16-0972-PDN
Robideau GP, De Cock AWAM, Coffey MD, Voglmayr H, Brouwer H, Bala K, Chitty DW, Désaulniers N, Eggertson QA, Gachon CMM, Hu CH, Küpper FC, Rintoul TL, Sarhan E, Verstappen ECP, Zhang Y, Bonants PJM, Ristaino JB, Lévesque CA (2011) DNA barcoding of oomycetes with cytochrome c oxidase subunit I and internal transcribed spacer. Mol Ecol Resour 11:1002–1011. https://doi.org/10.1111/j.1755-0998.2011.03041.x
Sako I, Tojo M, Kato M, Taguchi Y (1997) Root rot of Welsh onion caused by Pythium irregulare (Abstract in Japanese). Ann Phytopathol Soc Jpn 63:203
Shimizu S, Tojo M (2022a) Association of two unknown Pythium spp. without forming sexual structures on root rot of hydroponically grown Welsh onion. Asian Res J Agric 15:86–91. https://doi.org/10.9734/arja/2022/v15i430170
Shimizu S, Tojo M (2022b) First report of Pythium cluster B2a species causing root rot in Welsh onion. Plant Dis 106:336. https://doi.org/10.1094/PDIS-06-21-1211-PDN
Shimizu S, Nakano M, Kohguchi T, Ikeda H, Tojo M (2012) Species identity, host range and occurrence conditions of Pythium species causing root rot of Welsh onion grown in a hydroponic culture (Abstract in Japanese). Jpn J Phytopathol 78:191
Shimizu S, Uzuhashi S, Nishimura S, Mise R, Shinozaki S, Tojo M (2020) Morphological and molecular identification of Pythium aphanidermatum isolates from a root-rotted hydroponic spinach culture in Hiroshima Prefecture. Ann Rept Kansai Pl Prot 62:137–139. https://doi.org/10.4165/kapps.62.137
Stanghellini ME, Kronland WC (1986) Yield loss in hydroponically grown lettuce attributed to subclinical infection of feeder rootlets by Pythium dissotocum. Plant Dis 70:1053–1056
Stanghellini ME, Rasmussen SL (1994) Hydroponics: a solution for zoosporic pathogens. Plant Dis 78:1129–1138
Sutton JC, Sopher CR, Owen-Going TN, Liu W, Grodzinski B, Hall JC, Benchimol RL (2006) Etiology and epidemiology of Pythium root rot in hydroponic crops: current knowledge and perspectives. Summa Phytopathol 32:307–321. https://doi.org/10.1590/S0100-54052006000400001
Uzuhashi S, Kakishima M, Tojo M (2010) Phylogeny of the genus Pythium and description of new genera. Mycoscience 51:337–365. https://doi.org/10.1007/s10267-010-0046-7
van der Plaats-Niterink AJ (1981) Monograph of the genus Pythium. Stud Mycol 21:1–242
Yamamoto K (2018) Welsh onion. In: Hydroponic Society of Japan (ed) Handbook for practical hydroponic cultivations (in Japanese). Seibundo-shinkosha, Tokyo, pp 88–89 (ISBN: 978-4-416-91800-5)
Yu YN (1998) Peronosporales. In: Editorial Committee of Flora of China (ed) Flora fungorum sinicolum (in Chinese), vol 6. Science Press, Beijing, pp 111–131
Acknowledgements
We thank Yuko Ishiburo, Masahiro Nishimito, Chizuko Nagai, Naoki Hikasa, Takahiro Yamada, and Akira Miyawaki for their technical assistance. We thank commercial farmers in Hiroshima Prefecture for investigating and collecting samples, and Aya Imamura for sampling support. We also thank Masahiro Suzuki for his statistical recommendations. Part of this study was financially supported by the Hiroshima Prefecture government. We thank Editage (www.editage.com) for English language editing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised to correct the value in the sentence beginning “Throughout the experiment, ...”
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shimizu, S., Tojo, M. Suppressive effects of low-pH solutions on root rot in hydroponically grown Welsh onion. J Gen Plant Pathol 90, 151–162 (2024). https://doi.org/10.1007/s10327-024-01168-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10327-024-01168-2