Abstract
Background
Bone marrow mesenchymal stem cells (BMSCs) may be a promising target in the treatment of myocardial infarction (MI). However, the underlying molecular mechanisms of BMSC therapy remain unclear.
Objective
This study sought to evaluate the efficacy of direct intramyocardial transplantation of BMSCs in a mouse model of MI.
Methods
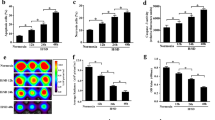
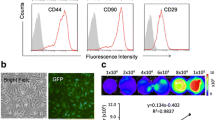
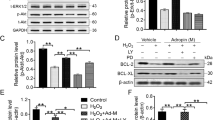
Mouse BMSCs were transfected with small interfering RNA or overexpression plasmid targeting TXNIP. The viability, proliferation, and apoptosis of BMSCs after hypoxia treatment were detected by MTT method, EdU analysis, and flow cytometry, respectively. A mouse model of MI was constructed, after which BSMCs were injected intramyocardially immediately. Cardiac ultrasound, HE staining, TUNEL staining and ELISA, IHC analysis, and Western blot were adopted to evaluate the effects of BSMC therapy on cardiac function, myocardial inflammation, and fibrosis in mice.
Results
In vitro experiments reported that ablating TXNIP increased viability and inhibited apoptosis of hypoxia-treated BMSCs while overexpressing TXNIP did the opposite. In vivo results stated that BSMCs improved cardiac function, myocardial inflammation, and fibrosis after MI, which was further improved by silencing TXNIP but reversed by overexpressing TXNIP. Meanwhile, in vivo cell tracking experiments showed that the retained BMSCs in the myocardium decreased after transplantation, and TXNIP depletion promoted the survival of BMSCs in MI mice, whereas TXNIP overexpression did the opposite.
Conclusion
In conclusion, BMSC transplantation improves cardiac function, myocardial inflammation, and fibrosis after MI by regulating the TXNIP/NLRP3 pathway.






Similar content being viewed by others
Availability of data and materials
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
References
Adluri R, Thirunavukkarasu M, Zhan L, Akita Y, Samuel S, Otani H, Ho Y, Maulik G, Maulik N (2011) Thioredoxin 1 enhances neovascularization and reduces ventricular remodeling during chronic myocardial infarction: a study using thioredoxin 1 transgenic mice. J Mol Cell Cardiol 50(1):239–247
Ashur C, Frishman W (2018) Cardiosphere-derived cells and ischemic heart failure. Cardiol Rev 26(1):8–21
Chen W (2023) TGF-β regulation of T cells. Annu Rev Immunol 26(41):483–512
Chen W, Wang S, Xia J, Huang Z, Tu X, Shen Z (2016) Protein phosphatase 2A plays an important role in migration of bone marrow stroma cells. Mol Cell Biochem 412:173–180
Chen Y, Zhao Y, Chen W, Xie L, Zhao Z, Yang J, Chen Y, Lei W, Shen Z (2017) MicroRNA-133 overexpression promotes the therapeutic efficacy of mesenchymal stem cells on acute myocardial infarction. Stem Cell Res Ther 8(1):268
Chen F, Liang P, Ye F, Hou C, Pi L (2020) Mesenchymal stem cell therapy for patients with ischemic heart failure -past, present, and future. Curr Stem Cell Res Therapy. 2021;16(5):608–621
Dai Y, Wang S, Chang S, Ren D, Shali S, Li C, Yang H, Huang Z, Ge J (2020) M2 macrophage-derived exosomes carry microRNA-148a to alleviate myocardial ischemia/reperfusion injury via inhibiting TXNIP and the TLR4/NF-κB/NLRP3 inflammasome signaling pathway. J Mol Cell Cardiol 142:65–79
Du W, Sang Y, Bai YH (2022) LRG1 expression reduced inflammation of sepsis-renal injury via activation of NLRP3 inflammasome by HIF-1 alpha. Mol Cell Toxicol 18:419–429
Gerbaud E, Elbaz M, Lattuca B (2020) New insights into cardiogenic shock and coronary revascularization after acute myocardial infarction. Arch Cardiovasc Dis 113(4):276–284
Gibb AA, Lazaropoulos MP, Elrod JW (2020) Myofibroblasts and fibrosis: mitochondrial and metabolic control of cellular differentiation. Circ Res 127(3):427–447
Gnecchi M, Melo L (2009) Bone marrow-derived mesenchymal stem cells: isolation, expansion, characterization, viral transduction, and production of conditioned medium. Methods Mol Biol (Clifton, NJ) 482:281–294
Guo L, Du J, Yuan D, Zhang Y, Zhang S, Zhang H, Mi J, Ning Y, Chen M, Wen D et al (2020) Optimal HO preconditioning to improve bone marrow mesenchymal stem cells’ engraftment in wound healing. Stem Cell Res Ther 11(1):434
Han Y, Xu X, Tang C, Gao P, Chen X, Xiong X, Yang M, Yang S, Zhu X, Yuan S et al (2018) Reactive oxygen species promote tubular injury in diabetic nephropathy: the role of the mitochondrial ros-txnip-nlrp3 biological axis. Redox Biol 16:32–46
Isaka Y (2018) Targeting TGF-β signaling in kidney fibrosis. Int J Mol Sci 19(9):2532
Isomi M, Sadahiro T, Ieda M (2019) Progress and challenge of cardiac regeneration to treat heart failure. J Cardiol 73(2):97–101
Jackson A, Rahman G, Yin K, Long S (2021) Enhancing matured stem-cardiac cell generation and transplantation: a novel strategy for heart failure therapy. J Cardiovasc Transl Res 14(3):556–572
Jang K, Tu T, Rosenblatt R, Burks S, Frank J (2020) MR-guided pulsed focused ultrasound improves mesenchymal stromal cell homing to the myocardium. J Cell Mol Med 24(22):13278–13288
Ji L, Long X, Tian H, Liu Y (2013) Effect of transplantation of bone marrow stem cells on myocardial infarction size in a rabbit model. World J Emerg Med 4(4):304–310
Jiang L, Yang A, Li X, Liu K, Tan J (2021) Down-regulation of VCAM-1 in bone mesenchymal stem cells reduces inflammatory responses and apoptosis to improve cardiac function in rat with myocardial infarction. Int Immunopharmacol 101:108180
Ju X, Xue D, Wang T, Ge B, Zhang Y, Li Z (2018) Catalpol promotes the survival and VEGF secretion of bone marrow-derived stem cells and their role in myocardial repair after myocardial infarction in rats. Cardiovasc Toxicol 18(5):471–481
Kim S, Cho J, Lee Y, Lee J, Kim S, Kim M, Lee M, Kang W, Lee K, Ahn Y et al (2018) Improvement in left ventricular function with intracoronary mesenchymal stem cell therapy in a patient with anterior wall st-segment elevation myocardial infarction. Cardiovasc Drugs Ther 32(4):329–338
Ko I, Kim B (2008) Mesenchymal stem cells for treatment of myocardial infarction. Inte J Stem Cells 1(1):49–54
Koitabashi N, Kass D (2011) Reverse remodeling in heart failure–mechanisms and therapeutic opportunities. Nat Rev Cardiol 9(3):147–157
Koyani CN, Plastira I, Sourij H, Hallström S, Schmidt A, Rainer PP, Bugger H, Frank S, Malle E, von Lewinski D (2020) Empagliflozin protects heart from inflammation and energy depletion via AMPK activation. Pharmacol Res 158:104870
Ling X, Wu W, Fan C, Xu C, Liao J, Rich L, Huang R, Repasky E, Wang X, Li F (2018) An ABCG2 non-substrate anticancer agent FL118 targets drug-resistant cancer stem-like cells and overcomes treatment resistance of human pancreatic cancer. J Exp Clin Cancer Res 37(1):240
Liu J, Ling B (2015) Bone marrow-derived mesenchymal stem cells in the treatment of myocardial infarction. Zhongguo Yi Xue Ke Xue Yuan Xue Bao Acta Academiae Medicinae Sinicae 37(1):108–112
Mack M (2018) Inflammation and fibrosis. Matrix Biol 68–69:106–121
McMurray J, Pfeffer M (2005) Heart failure. Lancet (London, England) 365(9474):1877–1889
Miao C, Lei M, Hu W, Han S, Wang Q (2017) A brief review: the therapeutic potential of bone marrow mesenchymal stem cells in myocardial infarction. Stem Cell Res Ther 8(1):242
Morikawa M, Derynck R, Miyazono K (2016) TGF-β and the TGF-β family: context-dependent roles in cell and tissue physiology. Cold Spring Harb Perspect Biol 8(5):a021873
Narita T, Suzuki K (2015) Bone marrow-derived mesenchymal stem cells for the treatment of heart failure. Heart Fail Rev 20(1):53–68
Orlic D, Kajstura J, Chimenti S, Bodine D, Leri A, Anversa P (2003) Bone marrow stem cells regenerate infarcted myocardium. 2003:7 Suppl 3:86–88
Qiu H, Liu W, Lan T, Pan W, Chen X, Wu H, Xu D (2018) Salvianolate reduces atrial fibrillation through suppressing atrial interstitial fibrosis by inhibiting TGF-β1/Smad2/3 and TXNIP/NLRP3 inflammasome signaling pathways in post-MI rats. Phytomedicine: Int J Phytotherapy Phytopharmacol 51:255–265
Sano M, Komiyama H, Shinoda R, Ozawa R, Watanabe H, Karasawa T, Takahashi M, Torii Y, Iwata H, Kuwayama T et al (2022) NLRP3 inflammasome is involved in testicular inflammation induced by lipopolysaccharide in mice. Am J Reproduct Immunol. 2022 Apr;87(4):e13527
Selvaraju V, Suresh S, Thirunavukkarasu M, Mannu J, Foye J, Mathur P, Palesty J, Sanchez J, McFadden D, Maulik N (2017) Regulation of A-kinase-anchoring protein 12 by heat shock protein A12B to prevent ventricular dysfunction following acute myocardial infarction in diabetic rats. J Cardiovasc Transl Res 10(2):209–220
Su H, Ji L, Xing W, Zhang W, Zhou H, Qian X, Wang X, Gao F, Sun X, Zhang H (2013) Acute hyperglycaemia enhances oxidative stress and aggravates myocardial ischaemia/reperfusion injury: role of thioredoxin-interacting protein. J Cell Mol Med 17(1):181–191
Sun D, Huang J, Zhang Z, Gao H, Li J, Shen M, Cao F, Wang H (2012) Luteolin limits infarct size and improves cardiac function after myocardium ischemia/reperfusion injury in diabetic rats. PLoS ONE 7(3):e33491
Sun Y, Xu H, Tan B, Yi Q, Liu H, Tian J, Zhu J (2023) Andrographolide-treated bone marrow mesenchymal stem cells-derived conditioned medium protects cardiomyocytes from injury by metabolic remodeling. Mol Biol Rep 50(3):2651–2662
Teng X, Chen L, Chen W, Yang J, Yang Z, Shen Z (2015) Mesenchymal stem cell-derived exosomes improve the microenvironment of infarcted myocardium contributing to angiogenesis and anti-inflammation. Cell Physiol Biochem Int J Exp Cell Physiol Biochem Pharmacol 37(6):2415–2424
Yue Y, Zong L, Chen Y, Feng Y, Tang N, Xu J, Zhao H (2021) Ling M Liver kinase B1 (LKB1) reduced inflammation and oxidative stress by regulating the AMPK/NLRP3 signaling pathway in LPS-induced lung injury. Mol Cell Toxicol 17:385–395
Zhang Z, Li S, Cui M, Gao X, Sun D, Qin X, Narsinh K, Li C, Jia H, Li C et al (2013) Rosuvastatin enhances the therapeutic efficacy of adipose-derived mesenchymal stem cells for myocardial infarction via PI3K/Akt and MEK/ERK pathways. Basic Res Cardiol 108(2):333
Zhao J, Liu X, Kong F, Qi T, Cheng G, Wang J, Sun C, Luan Y (2014) Bone marrow mesenchymal stem cells improve myocardial function in a swine model of acute myocardial infarction. Mol Med Rep 10(3):1448–1454
Acknowledgement
Not applicable.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
JB, CW, and HY designed the research study. QW performed the research. JZ provided help and advice on the experiments. DS analyzed the data. ZY, BM, and YL wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
JianNan Bai, Cong Wang, HongQiang Yu, QingChao Wang, JinFeng Zhang, DanDan Shao, ZhiQiang Yu, Bo Meng, and You Li have no conflicts of interest to declare.
Ethical approval
The present study was approved by Daqing People's Hospital and all procedures complied with the National Institutes of Health Guide for the Use of Laboratory Animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bai, J., Wang, C., Yu, H. et al. Bone marrow mesenchymal stem cell transplantation protects rats from myocardial infarction by regulating TXNIP/NLRP3 pathway-mediated inflammation and fibrosis. Mol. Cell. Toxicol. (2024). https://doi.org/10.1007/s13273-023-00422-6
Accepted:
Published:
DOI: https://doi.org/10.1007/s13273-023-00422-6