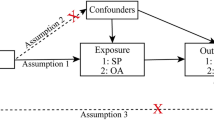
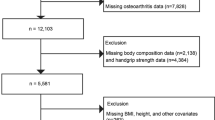
Abstract
Previous epidemiological evidence suggests rheumatoid arthritis is associated with sarcopenia-related features. However, most of the current evidence is from cross-sectional studies, and the causal link of this association is still to be determined. Therefore, this study was committed to a two-sample Mendelian randomization analysis to assess the causal effect of rheumatoid arthritis on sarcopenia-related features. In this two-sample Mendelian randomization study, instrumental variables for rheumatoid arthritis were obtained from the Non-Cancer Disease Study, and data for the five relevant characteristics of sarcopenia were pooled from UKBiobank. Inverse variance weighting is the primary analysis method for assessing causal effects. MR-Egger regression and weighted median are complementary analysis methods for causal effects. Leave-one-out analysis, horizontal pleiotropy test, and Heterogeneity test are applied as a sensitivity analysis to assess the robustness of causal effect estimates. The inverse variance weighted results for the five characteristics associated with sarcopenia and rheumatoid arthritis were: hand grip strength (right) (beta = − 2.309, se = 0.206, p = 3.340E-29), hand grip strength (left) (beta = − 2.046, se = 0.205, p = 2.166E-23), whole body lean mass (beta = − 0.843, se = 0.135, p = 4.67E-10), appendicular lean mass (beta = − 2.444, se = 0.208, p = 6.069E-32), Usual walking pace (OR 0.340, 95% CI (0.238, 0.484), p = 2.471E-09). The sensitivity analyses did not support that horizontal pleiotropy distorted causal effect estimates. The beta coefficient quantifies the number of standard deviations of the continuous outcome variables (hand grip strength, whole body lean mass, and appendicular lean mass) that change on average with each increase in the standard deviation of the binary exposure variable (rheumatoid arthritis). The odds ratios indicate the increased risk of the binary outcome variable (usual walking pace) per rheumatoid arthritis standard deviation increase. This study has demonstrated a negative causal effect of rheumatoid arthritis with five major sarcopenia-related features in a European population.

Similar content being viewed by others
Data Availability
We have provided a catalog and zip file of supplementary files. For all supplemental pictures, tables, and a supplemental file catalog, readers can obtain them from this zip.
Abbreviations
- EWGSOP2:
-
European Working Group on Sarcopenia in Older People 2
- GWAS:
-
Genome-wide association studies
- IVs:
-
Instrumental variables
- IVW:
-
Inverse variance-weighted
- MR:
-
Mendelian randomization
- MR-PRESSO:
-
Mendelian randomization pleiotropy residual sum and outlier
- RA:
-
Rheumatoid arthritis
- SNPs:
-
Single nucleotide polymorphisms
- TNF-α:
-
Tumor necrosis factor-alpha
References
Cruz-Jentoft AJ, Bahat G, Bauer J et al (2019) Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing 48(4):601–601. https://doi.org/10.1093/ageing/afz046
Bhasin S, Travison TG, Manini TM et al (2020) Sarcopenia definition: the position statements of the sarcopenia definition and outcomes consortium. J Am Geriatr Soc 68(7):1410–1418. https://doi.org/10.1111/jgs.16372
Yu H, Luo G, Sun T, Tang Q (2022) Causal effects of homocysteine levels on the components of sarcopenia: a two-sample Mendelian randomization study. Front Genet. https://doi.org/10.3389/fgene.2022.1051047
Firestein GS (2003) Evolving concepts of rheumatoid arthritis. Nature 423(6937):356–361. https://doi.org/10.1038/nature01661
Dao T, Kirk B, Phu S, Vogrin S, Duque G (2021) Prevalence of sarcopenia and its association with antirheumatic drugs in middle-aged and older adults with rheumatoid arthritis: a systematic review and meta-analysis. Calcif Tissue Int 109(5):475–489. https://doi.org/10.1007/s00223-021-00873-w
Santo RCE, Fernandes KZ, Lora PS, Filippin LI, Xavier RM (2018) Prevalence of rheumatoid cachexia in rheumatoid arthritis: a systematic review and meta-analysis: systematic review of RA cachexia prevalence. J Cachexia Sarcopenia Muscle 9(5):816–825. https://doi.org/10.1002/jcsm.12320
Vlietstra L, Stebbings S, Meredith-Jones K, Abbott JH, Treharne GJ, Waters DL (2019) Sarcopenia in osteoarthritis and rheumatoid arthritis: the association with self-reported fatigue, physical function and obesity. PLoS ONE 14(6):e0217462. https://doi.org/10.1371/journal.pone.0217462
Lee K, Lim CY (2019) Mendelian randomization analysis in observational epidemiology. J Lipid Atheroscler 8(2):67. https://doi.org/10.12997/jla.2019.8.2.67
Elsworth B, Lyon M, Alexander T et al (2020) The MRC IEU OpenGWAS data infrastructure. Published online. https://doi.org/10.1101/2020.08.10.244293
Qiu S, Li M, Jin S, Lu H, Hu Y (2021) Rheumatoid arthritis and cardio-cerebrovascular disease: a Mendelian randomization study. Front Genet. https://doi.org/10.3389/fgene.2021.745224
Cox N (2018) UK Biobank shares the promise of big data. Nature 562(7726):194–195. https://doi.org/10.1038/d41586-018-06948-3
Pei YF, Liu YZ, Yang XL et al (2020) The genetic architecture of appendicular lean mass characterized by association analysis in the UK Biobank study. Commun Biol. https://doi.org/10.1038/s42003-020-01334-0
Hemani G, Zheng J, Elsworth B et al (2018) The MR-base platform supports systematic causal inference across the human phenome. Elife. https://doi.org/10.7554/elife.34408
Verbanck M, Chen CY, Neale B, Do R (2018) Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat Genet 50(5):693–698. https://doi.org/10.1038/s41588-018-0099-7
Bowden J, Davey Smith G, Burgess S (2015) Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int J Epidemiol 44(2):512–525. https://doi.org/10.1093/ije/dyv080
Brion MJA, Shakhbazov K, Visscher PM (2013) Calculating statistical power in Mendelian randomization studies. Int J Epidemiol 42(5):1497–1501. https://doi.org/10.1093/ije/dyt179
Boehm FJ, Zhou X (2022) Statistical methods for Mendelian randomization in genome-wide association studies: a review. Comput Struct Biotechnol J 20:2338–2351. https://doi.org/10.1016/j.csbj.2022.05.015
Burgess S, Thompson SG (2017) Interpreting findings from Mendelian randomization using the MR-Egger method. Eur J Epidemiol 32(5):377–389. https://doi.org/10.1007/s10654-017-0255-x
Wiegmann S, Armbrecht G, Borucki D et al (2021) Association between sarcopenia, physical performance and falls in patients with rheumatoid arthritis: a 1-year prospective study. BMC Musculoskelet Disord. https://doi.org/10.1186/s12891-021-04605-x
Suginohara T, Kawaguchi M, Michihara S, Fujita N, Han LK, Takahashi R (2022) Ninjin’yoeito suppressed the onset of arthritis, pain, and muscle atrophy in rheumatoid arthritis model mice. Front Pharmacol. https://doi.org/10.3389/fphar.2022.974380
Alabarse PVG, Lora PS, Silva JMS et al (2018) Collagen-induced arthritis as an animal model of rheumatoid cachexia: CIA as an animal model of RA. J Cachexia Sarcopenia Muscle 9(3):603–612. https://doi.org/10.1002/jcsm.12280
Ono Y, Miyakoshi N, Kasukawa Y et al (2018) Effects of eldecalcitol and ibandronate on secondary osteoporosis and muscle wasting in rats with adjuvant-induced arthritis. Osteoporos Sarcopenia 4(4):128–133. https://doi.org/10.1016/j.afos.2018.11.085
Webster JM, Sagmeister MS, Fenton CG et al (2021) Global deletion of 11β-HSD1 prevents muscle wasting associated with glucocorticoid therapy in polyarthritis. Int J Mol Sci 22(15):7828. https://doi.org/10.3390/ijms22157828
Li J, Yi X, Yao Z, Chakkalakal JV, Xing L, Boyce BF (2020) TNF receptor-associated factor 6 mediates TNFα -induced skeletal muscle atrophy in mice during aging. J Bone Miner Res 35(8):1535–1548. https://doi.org/10.1002/jbmr.4021
Gómez-SanMiguel AB, Martín AI, Nieto-Bona MP, Fernández-Galaz C, Villanúa MÁ, López-Calderón A (2016) The melanocortin receptor type 3 agonistd-Trp(8)-γMSH decreases inflammation and muscle wasting in arthritic rats: <Scp>d</scp>-Trp(8)- MSH decreases inflammatory cachexia. J Cachexia Sarcopenia Muscle 7(1):79–89. https://doi.org/10.1002/jcsm.12036
Vial G, Coudy-Gandilhon C, Pinel A et al (2020) Lipid accumulation and mitochondrial abnormalities are associated with fiber atrophy in the skeletal muscle of rats with collagen-induced arthritis. Biochim Biophys Acta Mol Cell Biol Lipids 1865(2):158574. https://doi.org/10.1016/j.bbalip.2019.158574
Beaudart C, McCloskey E, Bruyère O et al (2016) Sarcopenia in daily practice: assessment and management. BMC Geriatr. https://doi.org/10.1186/s12877-016-0349-4
Tong JJ, Xu SQ, Wang JX et al (2021) Interactive effect of sarcopenia and falls on vertebral osteoporotic fracture in patients with rheumatoid arthritis. Arch Osteoporos. https://doi.org/10.1007/s11657-021-01017-1
Zhang M, Xu S, Zong H et al (2022) Effect of sarcopenia and poor balance on vertebral spinal osteoporotic fracture in female rheumatoid arthritis. Sci Rep. https://doi.org/10.1038/s41598-022-13339-2
Acknowledgements
We thank Professor Jing Zhang from the Department of Geriatrics, Chengdu University of Traditional Chinese Medicine Hospital, for her detailed review of the initial version of this manuscript and the feedback she gave.
Funding
This manuscript received no external funding.
Author information
Authors and Affiliations
Contributions
“Conceptualization, Kaixi Ding. And Wei Jiang.; methodology, Kaixi Ding. And Wei Jiang.; software, Juejue Zhangwang. And Jian Li.; validation, Juejue Zhangwang., Kai Ding.; writing—original draft preparation, Kaixi Ding.; writing—review and editing, Kaixi Ding. And Wei Jiang.; visualization, Wei Jiang.; supervision, Ming Lei.; project administration, Ming Lei. And Jian Li.
Corresponding author
Ethics declarations
Conflict of interest
Kaixi Ding, Wei Jiang, Juejue Zhangwang, Jian Li, and Ming Lei have declare no competing interests.
Consent to Participate
Not applicable.
Consent for Publication
All authors unanimously approved the publication of this manuscript.
Ethical Approval
Not applicable.
Human and Animal Rights
None of the contributing authors in this manuscript conducted research involving human or animal participants.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ding, K., Jiang, W., Zhangwang, J. et al. The Effect of Rheumatoid Arthritis on Features Associated with Sarcopenia: A Mendelian Randomization Study. Calcif Tissue Int 114, 286–294 (2024). https://doi.org/10.1007/s00223-023-01178-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-023-01178-w