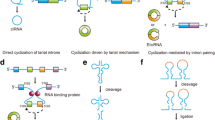
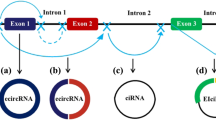
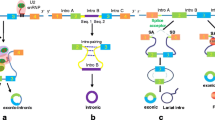
Abstract
Pre-messenger RNA molecules are back-spliced to create circular RNAs, which are non-coding RNA molecules. After a thorough investigation, it was discovered that these circRNAs have critical biological roles. CircRNAs have a variety of biological functions, including their ability to operate as microRNA sponges, interact with proteins to alter their stabilities and activities, and provide templates for the translation of proteins. Evidence supports a link between the emergence of numerous diseases, including various cancer types, and dysregulated circRNA expression. It is commonly known that a significant contributing element to cancer development is the disruption of numerous molecular pathways essential for preserving cellular and tissue homeostasis. The dysregulation of multiple biological processes is one of the hallmarks of cancer, and the molecular pathways linked to these processes are thought to be promising targets for therapeutic intervention. The biological and carcinogenic effects of circRNAs in the context of cancer are thoroughly reviewed in this article. Specifically, we highlight circRNAs’ involvement in signal transduction pathways and their possible use as novel biomarkers for the early identification and prognosis of human cancer.

Similar content being viewed by others
Data availability
Not applicable.
Abbreviations
- ncRNAs:
-
Non-coding RNAs
- mRNA:
-
Messenger RNA
- LncRNA:
-
Long non-coding RNA
- piRNA:
-
PIWI-interacting RNAs
- miRNA:
-
microRNA
- siRNA:
-
small interfering RNA
- circRNA:
-
Circular RNA
- ceRNA:
-
Competing endogenous RNA
- RBPs:
-
RNA-binding proteins
- MBNL1:
-
Muscleblind-like protein 1
- EIciRNA:
-
Exon–intron circRNA
- IRES:
-
Internal ribosome entry site
- MET:
-
Epithelial-mesenchymal transition
- NSCLC:
-
Non-small cell lung cancer
- PTK2:
-
Protein tyrosine kinase 2
- TGFβ:
-
Transforming growth factor-β
- TIF1γ:
-
Transcriptional intermediary factor 1 gamma
- SMARCA5:
-
SWI/SNF-elated, matrix-associated, actin dependent regulator of chromatin, subfamily A, member 5
- GC:
-
Gastric cancer
- TNBC:
-
Triple-negative breast cancer
- FoxO3:
-
Forkhead box O-3
- CDH19:
-
Cadherin-19
- GLIS3:
-
GLIS family zinc finger 3 isoform
- AIF1L:
-
Allograft inflammatory factor 1 like
- TNM:
-
Tumor, nodes, and metastases
- CRC:
-
Colorectal cancer
- LATS1:
-
large tumor suppressor gene 1
- EPHA3:
-
Ephrin tyrosine kinase a3
- PDE4D:
-
Phosphodiesterase 4D
- BMP-2:
-
Bone morphogenetic protein 2
- DNMT3A:
-
DNA methyltransferase 3 alpha
- SEMA5A:
-
Semaphorin 5A
- CCNE1:
-
Cyclin E1
- CDR1:
-
Cerebellum degeneration-related antigen 1
- TLK1:
-
Tousled-like kinase 1
- HCC:
-
Hepatocellular carcinoma
- PD-L1:
-
Programmed cell death ligand 1
- FGFR1:
-
Fibroblast growth factor receptor 1 protein
- CXCR-4:
-
C-X-C chemokine receptor type 4
- MAPK:
-
Mitogen-activated protein kinase
- ERK:
-
Extracellular signal-regulated kinase
- PI3K:
-
Phosphatidylinositol 3-kinase
- AKT:
-
Protein kinase B
- NF-κB:
-
Nuclear factor kappa B
- WNT5A:
-
Wnt family member 5A
- NIPBL:
-
Nipped-B-like protein
- ZEB1:
-
Zinc finger E‐box binding homeobox 1
- PHF16:
-
PHD finger protein 16
- RNF128:
-
Ring finger protein 128
- FZD5:
-
Frizzled class receptor 5
- ITCH:
-
Itchy E3 ubiquitin protein ligase
- FUS:
-
Fused in sarcoma
- SP:
-
Specificity protein 1
- EIF4A3:
-
Eukaryotic translation initiation factor 4A3
- CAPG:
-
Capping actin protein, gelsolin-like
- NFATC3:
-
Nuclear factor of activated T cells 3
- CTNNB1:
-
Catenin beta 1
- PLAGL2:
-
Pleomorphic adenoma gene like-2
- MTSS1:
-
Metastasis suppressor
- NEK6:
-
NIMA-related kinase 6
- CDK13:
-
Cyclin-dependent kinase 13
- NOL10:
-
Nucleolar protein 10
- SOCS2:
-
Suppressors of cytokine signaling 2
- SPARC:
-
Secreted protein acidic and rich in cysteine
- OSCC:
-
Oral squamous cell carcinoma
- STAT3:
-
Signal transducers and activators of transcription 3
- JAK:
-
Janus kinase
- ANKIB1:
-
Ankyrin repeat and IBR domain containing 1
- NRIP1:
-
Nuclear receptor-interacting protein 1
- MAPK:
-
Mitogen activated protein kinases
- TFAP2C:
-
Transcription factor AP‐2 gamma
- TRIM29:
-
Tripartite motif containing 29
- IL4R:
-
Interleukin 4 receptor
- IGF1R:
-
Insulin-like growth factor 1 receptor
- PIP5K1A:
-
Phosphatidylinositol-4-phosphate 5-kinase type 1 alpha
- TCF12:
-
Transcription factor 12
- HIPK3:
-
Homeodomain interacting protein kinase 3
- NUPR1:
-
Nuclear protein 1
- CSPP1:
-
Centrosome and spindle pole associated protein
- ITGB1:
-
Integrin beta 1
- mTOR:
-
Mammalian target of rapamycin
- PTEN:
-
Phosphatase and TENsin homolog deleted on chromosome 10
- ACTR2:
-
Actin related protein 2
- TAZ:
-
Tafazzin
- eEF2:
-
Eukaryotic translation elongation factor 2
- F-actin:
-
Filamentous actin
- LATS2:
-
Large tumor suppressor kinase 2
- PPP1R12A:
-
Protein phosphatase 1, regulatory subunit 12a
- XRN2:
-
5′-3′-exoribonuclease 2
- YAP:
-
Yes-associated protein
- ROCK1:
-
Rho-associated protein kinase 1
- IPO7:
-
Importin 7
- CEP128:
-
Centrosomal protein 128
- MYD88:
-
Myeloid differentiation primary response 88
- PVT1:
-
Plasmacytoma variant translocation 1 gene
- SLC7A11:
-
Solute carrier family 7 member 11
- UBE4B:
-
Ubiquitination factor E4B
- CORO1C:
-
Coronin‐like actin‐binding protein 1C
- GLIS2:
-
GLIS family zinc finger 2
- IKBKB:
-
Inhibitor of nuclear factor kappa B kinase subunit beta
- IKK:
-
IκB kinase
- IκBα:
-
Nuclear factor-kappa B inhibitor alpha
- ABHD5:
-
α/β-Hydrolase domain-containing protein 5
- ATGL:
-
Adipose triglyceride lipase
- TNF-α:
-
Tumor necrosis factor alpha
- TLR4:
-
Toll-like receptors
- CSC:
-
Cancer stem cell
- IPO11:
-
Importin-11
- TOP1:
-
DNA topoisomerase I
- GLI1:
-
Glioma-associated oncogene homolog 1
- CYR61:
-
Cysteine-rich angiogenic inducer 61
- DCAF6:
-
DDB1 and CUL4 associated factor 6
- SLC6A6:
-
Solute carrier family 6 member 6
- HSP90:
-
Heat shock protein 90
- NFIX:
-
Nuclear factor I X
- APLP2:
-
Amyloid precursor-like protein 2
- CRISPR:
-
Clustered regularly interspaced short palindromic repeats
- Cas:
-
CRISPR-associated protein
- MMP:
-
Matrix metallopeptidase
- APLP2:
-
Amyloid precursor-like protein 2
References
Palazzo AF, Lee ES. Non-coding RNA: what is functional and what is junk? Front Genet. 2015;6:2.
Anastasiadou E, Jacob LS, Slack FJ. Non-coding RNA networks in cancer. Nat Rev Cancer. 2018;18:5–18.
Kos A, Dijkema R, Arnberg A, Van der Meide P, Schellekens H. The hepatitis delta (δ) virus possesses a circular RNA. Nature. 1986;323:558–60.
Rybak-Wolf A, Stottmeister C, Glažar P, Jens M, Pino N, Giusti S, Hanan M, Behm M, Bartok O, Ashwal-Fluss R. Circular RNAs in the mammalian brain are highly abundant, conserved, and dynamically expressed. Mol Cell. 2015;58:870–85.
Pisignano G, Michael DC, Visal TH, Pirlog R, Ladomery M, Calin GA. Going circular: history, present, and future of circRNAs in cancer. Oncogene. 2023;42:1–18.
Garikipati VNS, Verma SK, Cheng Z, Liang D, Truongcao MM, Cimini M, Yue Y, Huang G, Wang C, Benedict C. Circular RNA CircFndc3b modulates cardiac repair after myocardial infarction via FUS/VEGF-A axis. Nat Commun. 2019;10:4317.
Wu F, Han B, Wu S, Yang L, Leng S, Li M, Liao J, Wang G, Ye Q, Zhang Y. Circular RNA TLK1 aggravates neuronal injury and neurological deficits after ischemic stroke via miR-335-3p/TIPARP. J Neurosci. 2019;39:7369–93.
Zhang F, Zhang R, Zhang X, Wu Y, Li X, Zhang S, Hou W, Ding Y, Tian J, Sun L. Comprehensive analysis of circRNA expression pattern and circRNA-miRNA-mRNA network in the pathogenesis of atherosclerosis in rabbits. Aging (Albany NY). 2018;10:2266.
Hu F, Peng Y, Fan X, Zhang X, Jin Z. Circular RNAs: implications of signaling pathways and bioinformatics in human cancer. Cancer Biol Med. 2023;20:104.
Wang C, Liu W-R, Tan S, Zhou J-K, Xu X, Ming Y, Cheng J, Li J, Zeng Z, Zuo Y. Characterization of distinct circular RNA signatures in solid tumors. Mol Cancer. 2022;21:1–16.
Jeck WR, Sorrentino JA, Wang K, Slevin MK, Burd CE, Liu J, Marzluff WF, Sharpless NE. Circular RNAs are abundant, conserved, and associated with ALU repeats. RNA. 2013;19:141–57.
Ashwal-Fluss R, Meyer M, Pamudurti NR, Ivanov A, Bartok O, Hanan M, Evantal N, Memczak S, Rajewsky N, Kadener S. circRNA biogenesis competes with pre-mRNA splicing. Mol Cell. 2014;56:55–66.
Li X, Yang L, Chen L-L. The biogenesis, functions, and challenges of circular RNAs. Mol Cell. 2018;71:428–42.
Tagawa T, Gao S, Koparde VN, Gonzalez M, Spouge JL, Serquiña AP, Lurain K, Ramaswami R, Uldrick TS, Yarchoan R. Discovery of Kaposi’s sarcoma herpesvirus-encoded circular RNAs and a human antiviral circular RNA. Proc Natl Acad Sci. 2018;115:12805–10.
Qiao Y, Zhao X, Liu J, Yang W. Epstein-Barr virus circRNAome as host miRNA sponge regulates virus infection, cell cycle, and oncogenesis. Bioengineered. 2019;10:593–603.
Schmidt CA, Giusto JD, Bao A, Hopper AK, Matera AG. Molecular determinants of metazoan tricRNA biogenesis. Nucleic Acids Res. 2019;47:6452–65.
Zeng K, Chen X, Xu M, Liu X, Hu X, Xu T, Sun H, Pan Y, He B, Wang S. CircHIPK3 promotes colorectal cancer growth and metastasis by sponging miR-7. Cell Death Dis. 2018;9:417.
Huang A, Zheng H, Wu Z, Chen M, Huang Y. Circular RNA-protein interactions: functions, mechanisms, and identification. Theranostics. 2020;10:3503–17.
Zhang Y, Wang X. Targeting the Wnt/β-catenin signaling pathway in cancer. J Hematol Oncol. 2020;13:1–16.
Ma J-h, Qin L, Li X. Role of STAT3 signaling pathway in breast cancer. Cell Commun Signal. 2020;18:1–13.
Zou Z, Tao T, Li H, Zhu X. mTOR signaling pathway and mTOR inhibitors in cancer: progress and challenges. Cell Biosci. 2020;10:1–11.
Wang S, Cheng L, Wu H, Li G. Mechanisms and prospects of circular RNAs and their interacting signaling pathways in colorectal cancer. Front Oncol. 2022;12: 949656.
Yang Z, Xie L, Han L, Qu X, Yang Y, Zhang Y, He Z, Wang Y, Li J. Circular RNAs: regulators of cancer-related signaling pathways and potential diagnostic biomarkers for human cancers. Theranostics. 2017;7:3106.
Wang L, Tong X, Zhou Z, Wang S, Lei Z, Zhang T, Liu Z, Zeng Y, Li C, Zhao J. Circular RNA hsa_circ_0008305 (circPTK2) inhibits TGF-β-induced epithelial-mesenchymal transition and metastasis by controlling TIF1γ in non-small cell lung cancer. Mol Cancer. 2018;17:1–18.
Tong S. Circular RNA SMARCA5 may serve as a tumor suppressor in non-small cell lung cancer. J Clin Lab Anal. 2020;34: e23195.
Zhang J, Liu H, Hou L, Wang G, Zhang R, Huang Y, Chen X, Zhu J. Circular RNA_LARP4 inhibits cell proliferation and invasion of gastric cancer by sponging miR-424-5p and regulating LATS1 expression. Mol Cancer. 2017;16:1–16.
Lu J, Zhang P-y, Li P, Xie J-w, Wang J-b, Lin J-x, Chen Q-y, Cao L-l, Huang C-m, Zheng C-h. Circular RNA hsa_circ_0001368 suppresses the progression of gastric cancer by regulating miR-6506–5p/FOXO3 axis. Biochem Biophys Res Commun. 2019;512:29–33.
Shi Y, Han T, Liu C. CircRNA hsa_circ_0006220 acts as a tumor suppressor gene by regulating miR-197–5p/CDH19 in triple-negative breast cancer. Ann Transl Med. 2021;9:1236.
Cao S, Yin Y, Hu H, Hong S, He W, Lv W, Liu R, Li Y, Yu S, Xiao H. CircGLIS3 inhibits thyroid cancer invasion and metastasis through miR-146b-3p/AIF1L axis. Cell Oncol. 2023;43:1–13.
Deng YY, Min YJ, Zhou K, Yang QS, Peng M, Cui ZR, Zhu XL, Liu H, Wang M, Zhang X, Liu LX. Identification of the tumor-suppressive role of circular RNA-FOXO3 in colorectal cancer via regulation of miR-543/LATS1 axis. Oncol Rep. 2021. https://doi.org/10.3892/or.2021.8190.
Deng Y-Y, Min Y-J, Zhou K, Yang Q-S, Peng M, Cui Z-R, Zhu X-L, Liu H, Wang M, Zhang X. Identification of the tumor-suppressive role of circular RNA-FOXO3 in colorectal cancer via regulation of miR-543/LATS1 axis. Oncol Rep. 2021;46:1–11.
Li J, Lv J, Chen Y, Li L. Tumor suppressor circPDE4D inhibits the progression of colorectal cancer and regulates oxaliplatin chemoresistance. Gene. 2023;864: 147323.
Feng H, Deng Z, Peng W, Wei X, Liu J, Wang T. Circular RNA EPHA3 suppresses progression and metastasis in prostate cancer through the miR-513a-3p/BMP2 axis. J Transl Med. 2023;21:1–15.
Zhang B, Yang S, Wang J. Circ_0084615 is an oncogenic circular RNA in colorectal cancer and promotes DNMT3A expression via repressing miR-599. Pathol Res Pract. 2021;224: 153494.
Ye Q, Liu S, Lin S, Xie W. Circular RNA circSEMA5A facilitates colorectal cancer development by regulating microRNA-195-5p to target CCNE1 axis. Cell Signal. 2023;107: 110649.
Yi H, Han Y, Li S. Oncogenic circular RNA circ_0007534 contributes to paclitaxel resistance in endometrial cancer by sponging miR-625 and promoting ZEB2 expression. Front Oncol. 2022;12: 985470.
Yu L, Gong X, Sun L, Zhou Q, Lu B, Zhu L. The circular RNA Cdr1as act as an oncogene in hepatocellular carcinoma through targeting miR-7 expression. PLoS ONE. 2016;11: e0158347.
Lu Y, Liu Y, Zhang K, Jiang L. Circular RNA TLK1 exerts oncogenic functions in hepatocellular carcinoma by acting as a ceRNA of miR-138–5p. J Oncol. 2022;2022:1–9.
Qu D, Yan B, Xin R, Ma T. A novel circular RNA hsa_circ_0020123 exerts oncogenic properties through suppression of miR-144 in non-small cell lung cancer. Am J Cancer Res. 2018;8:1387.
Lu W, Li L, Li L, Guo N, Ma X. Circular RNA circ_0101675 promotes NSCLC cell proliferation, migration, invasion, angiogenesis and immune evasion by sponging miR-607/PDL1 axis. Biochem Genet. 2023. https://doi.org/10.1007/s10528-023-10493-8.
Zhang Q, Chen S, Zhen Y, Gao P, Zhang Z, Guo H, Wang Y. Circular RNA circFGFR1 functions as an oncogene in glioblastoma cells through sponging to hsa-miR-224–5p. J Immunol Res. 2022;2022:1–14.
Zhan T, Rindtorff N, Boutros M. Wnt signaling in cancer. Oncogene. 2017;36:1461–73.
Kikuchi A, Yamamoto H, Sato A, Matsumoto S. Wnt5a: its signalling, functions and implication in diseases. Acta Physiol. 2012;204:17–33.
Li Y, Kong Y, An M, Luo Y, Zheng H, Lin Y, Chen J, Yang J, Liu L, Luo B. ZEB1-mediated biogenesis of circNIPBL sustains the metastasis of bladder cancer via Wnt/β-catenin pathway. J Exp Clin Cancer Res. 2023;42:191.
Ding L, Lin Y, Chen X, Wang R, Lu H, Wang H, Luo W, Lu Z, Xia L, Zhou X. circPHF16 suppresses prostate cancer metastasis via modulating miR-581/RNF128/Wnt/β-catenin pathway. Cell Signal. 2023;102: 110557.
Huang J, Cai Y, Guo L, Huang W, Yan J, Lai J, Wang Y, Jiang D, Peng L. hsa_circ_0136839 regulates the malignant phenotypes of nasopharyngeal carcinoma via the Wnt/β-catenin signaling pathway. Pathol Res Pract. 2023;245: 154433.
Huang G, Liang M, Liu H, Huang J, Li P, Wang C, Zhang Y, Lin Y, Jiang X. CircRNA hsa_circRNA_104348 promotes hepatocellular carcinoma progression through modulating miR-187-3p/RTKN2 axis and activating Wnt/β-catenin pathway. Cell Death Dis. 2020;11:1065.
Zhu Q, Lu G, Luo Z, Gui F, Wu J, Zhang D, Ni Y. CircRNA circ_0067934 promotes tumor growth and metastasis in hepatocellular carcinoma through regulation of miR-1324/FZD5/Wnt/β-catenin axis. Biochem Biophys Res Commun. 2018;497:626–32.
Peng Y, Wang HH. Cir-ITCH inhibits gastric cancer migration, invasion and proliferation by regulating the Wnt/β-catenin pathway. Sci Rep. 2020;10:17443.
Zhang X, Yao J, Shi H, Gao B, Zhou H, Zhang Y, Zhao D, Gao S, Wang C, Zhang L. Hsa_circ_0026628 promotes the development of colorectal cancer by targeting SP1 to activate the Wnt/β-catenin pathway. Cell Death Dis. 2021;12:802.
Chen Z, Duan X. hsa_circ_0000177-miR-638-FZD7-Wnt signaling cascade contributes to the malignant behaviors in glioma. DNA Cell Biol. 2018;37:791–7.
Zhou Q, Fu Q, Shaya M, Kugeluke Y, Li S, Dilimulati Y. Knockdown of circ_0055412 promotes cisplatin sensitivity of glioma cells through modulation of CAPG and Wnt/β-catenin signaling pathway. CNS Neurosci Ther. 2022;28:884–96.
Gao N, Ye B. Circ-SOX4 drives the tumorigenesis and development of lung adenocarcinoma via sponging miR-1270 and modulating PLAGL2 to activate WNT signaling pathway. Cancer Cell Int. 2020;20:1–12.
Peng L, Liu Q, Wu T, Li P, Cai Y, Wei X, Zeng Y, Ye J, Chen P, Huang J. Hsa_circ_0087302, a circular RNA, affects the progression of osteosarcoma via the Wnt/β-catenin signaling pathway. Int J Med Sci. 2022;19:1377.
Wu M, Qiu Q, Zhou Q, Li J, Yang J, Zheng C, Luo A, Li X, Zhang H, Cheng X. circFBXO7/miR-96-5p/MTSS1 axis is an important regulator in the Wnt signaling pathway in ovarian cancer. Mol Cancer. 2022;21:1–19.
Chen F, Feng Z, Zhu J, Liu P, Yang C, Huang R, Deng Z. Emerging roles of circRNA_NEK6 targeting miR-370-3p in the proliferation and invasion of thyroid cancer via Wnt signaling pathway. Cancer Biol Ther. 2018;19:1139–52.
Xin P, Xu X, Deng C, Liu S, Wang Y, Zhou X, Ma H, Wei D, Sun S. The role of JAK/STAT signaling pathway and its inhibitors in diseases. Int Immunopharmacol. 2020;80: 106210.
Lin Q, Ling Y-B, Chen J-W, Zhou C-R, Chen J, Li X, Huang M-S. Circular RNA circCDK13 suppresses cell proliferation, migration and invasion by modulating the JAK/STAT and PI3K/AKT pathways in liver cancer. Int J Oncol. 2018;53:246–56.
Wang F, Wang X, Li J, Lv P, Han M, Li L, Chen Z, Dong L, Wang N, Gu Y. CircNOL10 suppresses breast cancer progression by sponging miR-767-5p to regulate SOCS2/JAK/STAT signaling. J Biomed Sci. 2021;28:1–16.
Gu Y, Wang Y, He L, Zhang J, Zhu X, Liu N, Wang J, Lu T, He L, Tian Y. Circular RNA circIPO11 drives self-renewal of liver cancer initiating cells via Hedgehog signaling. Mol Cancer. 2021;20:1–20.
Huang C, Li H, Zhou L, Li D. Circ_0005050 promotes the proliferation of oral squamous cell carcinoma and inhibits the apoptosis by activating JAK/STAT3 signaling pathway. Pathol Res Pract. 2022;238: 154058.
Du Y-x, Guo L-x, Pan H-s, Liang Y-m, Li X. Circ_ANKIB1 stabilizes the regulation of miR-19b on SOCS3/STAT3 pathway to promote osteosarcoma cell growth and invasion. Hum Cell. 2020;33:252–60.
Li C, Zhu L, Fu L, Han M, Li Y, Meng Z, Qiu X. CircRNA NRIP1 promotes papillary thyroid carcinoma progression by sponging mir-195-5p and modulating the P38 MAPK and JAK/STAT pathways. Diagn Pathol. 2021;16:1–11.
Molinaro A, Becattini B, Mazzoli A, Bleve A, Radici L, Maxvall I, Sopasakis VR, Molinaro A, Bäckhed F, Solinas G. Insulin-driven PI3K-AKT signaling in the hepatocyte is mediated by redundant PI3Kα and PI3Kβ activities and is promoted by RAS. Cell Metab. 2019;29(1400–1409): e1405.
Fruman DA, Chiu H, Hopkins BD, Bagrodia S, Cantley LC, Abraham RT. The PI3K pathway in human disease. Cell. 2017;170:605–35.
Okkenhaug K, Graupera M, Vanhaesebroeck B. Targeting PI3K in cancer: impact on tumor cells, their protective stroma, angiogenesis, and immunotherapy. Cancer Discov. 2016;6:1090–105.
Vara JÁF, Casado E, de Castro J, Cejas P, Belda-Iniesta C, González-Barón M. PI3K/Akt signalling pathway and cancer. Cancer Treat Rev. 2004;30:193–204.
Xue C, Li G, Lu J, Li L. Crosstalk between circRNAs and the PI3K/AKT signaling pathway in cancer progression. Signal Transduct Target Ther. 2021;6:400.
Jiang T, Wang H, Liu L, Song H, Zhang Y, Wang J, Liu L, Xu T, Fan R, Xu Y. CircIL4R activates the PI3K/AKT signaling pathway via the miR-761/TRIM29/PHLPP1 axis and promotes proliferation and metastasis in colorectal cancer. Mol Cancer. 2021;20:1–24.
Wang Y, Yin L, Sun X. CircRNA hsa_circ_0002577 accelerates endometrial cancer progression through activating IGF1R/PI3K/Akt pathway. J Exp Clin Cancer Res. 2020;39:1–16.
Zheng K, Xie H, Wu W, Wen X, Zeng Z, Shi Y. CircRNA PIP5K1A promotes the progression of glioma through upregulation of the TCF12/PI3K/AKT pathway by sponging miR-515-5p. Cancer Cell Int. 2021;21:1–13.
Jiang W, Zhang C, Zhang X, Sun L, Li J, Zuo J. CircRNA HIPK3 promotes the progression of oral squamous cell carcinoma through upregulation of the NUPR1/PI3K/AKT pathway by sponging miR-637. Ann Transl Med. 2021;9:860.
Yang W, Xie T. Hsa_circ_CSPP1/MiR-361-5p/ITGB1 regulates proliferation and migration of cervical cancer (CC) by modulating the PI3K-Akt signaling pathway. Reprod Sci. 2020;27:132–44.
Wu D, Li Y, Xu A, Tang W, Yu B. CircRNA RNA hsa_circ_0008234 promotes colon cancer progression by regulating the miR-338-3p/ETS1 axis and PI3K/AKT/mTOR signaling. Cancers. 2023;15:2068.
Xu C, Ye Q, Ye C, Liu S. circACTR2 attenuates gemcitabine chemoresiatance in pancreatic cancer through PTEN mediated PI3K/AKT signaling pathway. Biol Direct. 2023;18:14.
Fang K, Liang S, Liu D, Yi Q, Li Y, Zhu R. Hsa_circ_0003596 enhances the development of cell renal clear cell carcinoma through the miR-502-5p/IGF1/PI3K/AKT axis. J Gene Med. 2023;25:e3562.
Zhou J, Jin S. Circ_0058063 contributed to oral squamous cell carcinoma development by sponging miR-145 and regulating PI3K/AKT pathway. Mol Biotechnol. 2023;65:1–12.
Yu F-X, Zhao B, Guan K-L. Hippo pathway in organ size control, tissue homeostasis, and cancer. Cell. 2015;163:811–28.
Wang X, Ji C, Hu J, Deng X, Zheng W, Yu Y, Hua K, Zhou X, Fang L. Hsa_circ_0005273 facilitates breast cancer tumorigenesis by regulating YAP1-hippo signaling pathway. J Exp Clin Cancer Res. 2021;40:1–17.
Geng Z, Wang W, Chen H, Mao J, Li Z, Zhou J. Circ_0001667 promotes breast cancer cell proliferation and survival via Hippo signal pathway by regulating TAZ. Cell Biosci. 2019;9:1–10.
Gao P, Yang Y, Li X, Zhao Q, Liu Y, Dong C, Zhang Y, Liu D. Circular RNA hsa_circ_0098181 inhibits metastasis in hepatocellular carcinoma by activating the Hippo signaling pathway via interaction with eEF2. Ann Hepatol. 2023;28:101124.
Peng Q-S, Cheng Y-N, Zhang W-B, Fan H, Mao Q-H, Xu P. circRNA_0000140 suppresses oral squamous cell carcinoma growth and metastasis by targeting miR-31 to inhibit Hippo signaling pathway. Cell Death Dis. 2020;11:112.
Zheng X, Chen L, Zhou Y, Wang Q, Zheng Z, Xu B, Wu C, Zhou Q, Hu W, Wu C. A novel protein encoded by a circular RNA circPPP1R12A promotes tumor pathogenesis and metastasis of colon cancer via Hippo-YAP signaling. Mol Cancer. 2019;18:1–13.
Xie B, Lin J, Chen X, Zhou X, Zhang Y, Fan M, Xiang J, He N, Hu Z, Wang F. CircXRN2 suppresses tumor progression driven by histone lactylation through activating the Hippo pathway in human bladder cancer. Mol Cancer. 2023;22:151.
Wu P, Hou X, Peng M, Deng X, Yan Q, Fan C, Mo Y, Wang Y, Li Z, Wang F. Circular RNA circRILPL1 promotes nasopharyngeal carcinoma malignant progression by activating the Hippo-YAP signaling pathway. Cell Death Differ. 2023;30:1–16.
Wang Q, Feng J, Tang L. Non-coding RNA related to MAPK signaling pathway in liver cancer. Int J Mol Sci. 2022;23:11908.
He J, Huang Z, He M, Liao J, Zhang Q, Wang S, Xie L, Ouyang L, Koeffler HP, Yin D. Circular RNA MAPK4 (circ-MAPK4) inhibits cell apoptosis via MAPK signaling pathway by sponging miR-125a-3p in gliomas. Mol Cancer. 2020;19:1–17.
Jiang T, Xia Y, Lv J, Li B, Li Y, Wang S, Xuan Z, Xie L, Qiu S, He Z. A novel protein encoded by circMAPK1 inhibits progression of gastric cancer by suppressing activation of MAPK signaling. Mol Cancer. 2021;20:1–18.
Liu T, Zhou L, He Z, Chen Y, Jiang X, Xu J, Jiang J. Circular RNA hsa_circ_0006117 facilitates pancreatic cancer progression by regulating the miR-96–5p/KRAS/MAPK signaling pathway. J Oncol. 2021;2021:1–16.
Sun M, Zhao W, Chen Z, Li M, Li S, Wu B, Bu R. Circular RNA CEP128 promotes bladder cancer progression by regulating Mir-145-5p/Myd88 via MAPK signaling pathway. Int J Cancer. 2019;145:2170–81.
Wang S, Li W, Yang L, Yuan J, Wang L, Li N, Zhao H. CircPVT1 facilitates the progression of oral squamous cell carcinoma by regulating miR-143-3p/SLC7A11 axis through MAPK signaling pathway. Funct Integr Genom. 2022;22:891–903.
Lyu Y, Tan B, Li L, Liang R, Lei K, Wang K, Wu D, Lin H, Wang M. A novel protein encoded by circUBE4B promotes progression of esophageal squamous cell carcinoma by augmenting MAPK/ERK signaling. Cell Death Dis. 2023;14:346.
Wang B, Chen H, Deng Y, Chen H, Xing L, Guo Y, Wang M, Chen J. CircDNAJC11 interacts with TAF15 to promote breast cancer progression via enhancing MAPK6 expression and activating the MAPK signaling pathway. J Transl Med. 2023;21:186.
Baud V, Karin M. Is NF-κB a good target for cancer therapy? Hopes and pitfalls. Nat Rev Drug Discov. 2009;8:33–40.
Wu F, Sun G, Zheng W, Tang W, Cheng Y, Wu L, Li X, Tao J, Ma S, Cao H. circCORO1C promotes the proliferation and metastasis of hepatocellular carcinoma by enhancing the expression of PD-L1 through NF-κB pathway. J Clin Lab Anal. 2021;35: e24003.
Chen J, Yang X, Liu R, Wen C, Wang H. Circular RNA GLIS2 promotes colorectal cancer cell motility via activation of the NF-κB pathway. Cell Death Dis. 2020;11:788.
Xu Y, Zhang S, Liao X, Li M, Chen S, Li X, Wu X, Yang M, Tang M, Hu Y. Circular RNA circIKBKB promotes breast cancer bone metastasis through sustaining NF-κB/bone remodeling factors signaling. Mol Cancer. 2021;20:1–18.
Zhou Q, Sun Y. Circular RNA cMras suppresses the progression of lung adenocarcinoma through ABHD5/ATGL axis using NF-κB signaling pathway. Cancer Biother Radiopharm. 2023;38:336–46.
Ai Y, Song J, Wei H, Tang Z, Li X, Lv X, Luo H, Wu S, Zou C. circ_0001461 promotes oral squamous cell carcinoma progression through miR-145/TLR4/NF-κB axis. Biochem Biophys Res Commun. 2021;566:108–14.
Robbins DJ, Fei DL, Riobo NA. The Hedgehog signal transduction network. Sci signal. 2012;5:re6–re6.
Wang L, Li B, Yi X, Xiao X, Zheng Q, Ma L. Circ_0036412 affects the proliferation and cell cycle of hepatocellular carcinoma via hedgehog signaling pathway. J Transl Med. 2022;20:1–18.
Zhang P, Gao H, Yan R, Yu L, Xia C, Yang D. has_circ_0070512 promotes prostate cancer progression by regulating the miR-338-3p/hedgehog signaling pathway. Cancer Sci. 2023;114:1491.
Chen J, Zhou X, Yang J, Sun Q, Liu Y, Li N, Zhang Z, Xu H. Circ-GLI1 promotes metastasis in melanoma through interacting with p70S6K2 to activate Hedgehog/GLI1 and Wnt/β-catenin pathways and up-regulate Cyr61. Cell Death Dis. 2020;11:596.
Ye G, Pan R, Zhu L, Zhou D. Circ_DCAF6 potentiates cell stemness and growth in breast cancer through GLI1-Hedgehog pathway. Exp Mol Pathol. 2020;116: 104492.
Allenspach EJ, Maillard I, Aster JC, Pear WS. Notch signaling in cancer. Cancer Biol Ther. 2002;1:466–76.
Katoh M, Katoh M. Precision medicine for human cancers with notch signaling dysregulation. Int J Mol Med. 2020;45:279–97.
Hui Y, Wenguang Y, Wei S, Haoran W, Shanglei N, Ju L. circSLC4A7 accelerates stemness and progression of gastric cancer by interacting with HSP90 to activate NOTCH1 signaling pathway. Cell Death Dis. 2023;14:452.
Xu H, Zhang Y, Qi L, Ding L, Jiang H, Yu H. NFIX circular RNA promotes glioma progression by regulating miR-34a-5p via notch signaling pathway. Front Mol Neurosci. 2018;11:225.
Wu H-B, Huang S-S, Lu C-G, Tian S-D, Chen M. CircAPLP2 regulates the proliferation and metastasis of colorectal cancer by targeting miR-101-3p to activate the notch signalling pathway. Am J Transl Res. 2020;12:2554.
Holdt LM, Kohlmaier A, Teupser D. Circular RNAs as therapeutic agents and targets. Front Physiol. 2018;9:1262.
Liang D, Tatomer DC, Luo Z, Wu H, Yang L, Chen L-L, Cherry S, Wilusz JE. The output of protein-coding genes shifts to circular RNAs when the pre-mRNA processing machinery is limiting. Mol Cell. 2017;68(940–954): e943.
Pan H, Li T, Jiang Y, Pan C, Ding Y, Huang Z, Yu H, Kong D. Overexpression of circular RNA ciRS-7 abrogates the tumor suppressive effect of miR-7 on gastric cancer via PTEN/PI3K/AKT signaling pathway. J Cell Biochem. 2018;119:440–6.
Meganck RM, Liu J, Hale AE, Simon KE, Fanous MM, Vincent HA, Wilusz JE, Moorman NJ, Marzluff WF, Asokan A. Engineering highly efficient backsplicing and translation of synthetic circRNAs. Mol Ther-Nucleic Acids. 2021;23:821–34.
Pamudurti NR, Patop IL, Krishnamoorthy A, Ashwal-Fluss R, Bartok O, Kadener S. An in vivo strategy for knockdown of circular RNAs. Cell Discov. 2020;6:52.
Okano M, Oshi M, Butash AL, Asaoka M, Katsuta E, Peng X, Qi Q, Yan L, Takabe K. Estrogen receptor positive breast cancer with high expression of androgen receptor has less cytolytic activity and worse response to neoadjuvant chemotherapy but better survival. Int J Mol Sci. 2019;20:2655.
Hu J, Peng X, Du W, Huang Y, Zhang C, Zhang X. circSLC6A6 sponges miR-497-5p to promote endometrial cancer progression via the PI4KB/hedgehog axis. J Immunol Res. 2021;2021:1–13.
Acknowledgements
None.
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
ROS, SISA-H, BDO, AH: Conceptualization, Writing—Original Draft. KHO, MJA, SSJ: Conceptualization, Writing—Review & editing, Visualization. A Alawadi, A Alsalamy: Conceptualization. SAJ: Supervision.
Corresponding author
Ethics declarations
Conflict of interests
The authors have not disclosed any competing interests.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saleh, R.O., Al-Hawary, S.I.S., Jasim, S.A. et al. A therapeutical insight into the correlation between circRNAs and signaling pathways involved in cancer pathogenesis. Med Oncol 41, 69 (2024). https://doi.org/10.1007/s12032-023-02275-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-023-02275-4