Abstract
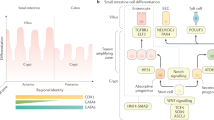
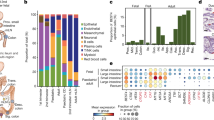
A key aspect of nutrient absorption is the exquisite division of labour across the length of the small intestine, with individual nutrients taken up at different proximal:distal positions. For millennia, the small intestine was thought to comprise three segments with indefinite borders: the duodenum, jejunum and ileum. By examining the fine-scale longitudinal transcriptional patterns that span the mouse and human small intestine, we instead identified five domains of nutrient absorption that mount distinct responses to dietary changes, and three regional stem cell populations. Molecular domain identity can be detected with machine learning, which provides a systematic method to computationally identify intestinal domains in mice. We generated a predictive model of transcriptional control of domain identity and validated the roles of Ppar-δ and Cdx1 in patterning lipid metabolism-associated genes. These findings represent a foundational framework for the zonation of absorption across the mammalian small intestine.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The datasets generated and analysed in the current study are available in the Gene Expression Omnibus (GEO; BioProject accession code GSE201859) and can be visualized using the Chan Zuckerberg CELLxGENE tool (https://cellxgene.cziscience.com/collections/3db5617e-9f12-4eb4-8416-94893a0d7c46). Previously published single-cell sequencing data6 analysed here are available under accession code GSE92332. Source data and analysis outputs are provided in the Supplementary Information associated with this publication. All other data supporting the findings of this study are available from the corresponding author on reasonable request.
Code availability
Custom code developed for this manuscript is available in Zenodo under record no. 10223562.
References
San Roman, A. K. & Shivdasani, R. A. Boundaries, junctions and transitions in the gastrointestinal tract. Exp. Cell. Res. 317, 2711–2718 (2011).
Brown, H. & Esterhazy, D. Intestinal immune compartmentalization: implications of tissue specific determinants in health and disease. Mucosal Immunol. 14, 1259–1270 (2021).
Esterhazy, D. et al. Compartmentalized gut lymph node drainage dictates adaptive immune responses. Nature 569, 126–130 (2019).
Altmann, G. G. & Leblond, C. P. Factors influencing villus size in the small intestine of adult rats as revealed by transposition of intestinal segments. Am. J. Anat. 127, 15–36 (1970).
Bates, M. D. et al. Novel genes and functional relationships in the adult mouse gastrointestinal tract identified by microarray analysis. Gastroenterology 122, 1467–1482 (2002).
Haber, A. L. et al. A single-cell survey of the small intestinal epithelium. Nature 551, 333–339 (2017).
Elmentaite, R. et al. Cells of the human intestinal tract mapped across space and time. Nature 597, 250–255 (2021).
Burclaff, J. et al. A proximal-to-distal survey of healthy adult human small intestine and colon epithelium by single-cell transcriptomics. Cell Mol. Gastroenterol. Hepatol. 13, 1554–1589 (2022).
Wang, Y. et al. Single-cell transcriptome analysis reveals differential nutrient absorption functions in human intestine. J. Exp. Med. 217, jem.20191130 (2020).
Hickey, J. W. et al. Organization of the human intestine at single-cell resolution. Nature 619, 572–584 (2023).
Fawkner-Corbett, D. et al. Spatiotemporal analysis of human intestinal development at single-cell resolution. Cell 184, 810–826 (2021).
Zwick, R. K., Ohlstein, B. & Klein, O. D. Intestinal renewal across the animal kingdom: comparing stem cell activity in mouse and Drosophila. Am. J. Physiol. Gastrointest. Liver Physiol. 316, G313–G322 (2019).
Buchon, N. et al. Morphological and molecular characterization of adult midgut compartmentalization in Drosophila. Cell Rep. 3, 1725–1738 (2013).
Marianes, A. & Spradling, A. C. Physiological and stem cell compartmentalization within the Drosophila midgut. eLife 2, e00886 (2013).
Driver, I. & Ohlstein, B. Specification of regional intestinal stem cell identity during Drosophila metamorphosis. Development 141, 1848–1856 (2014).
Hudry, B. et al. Sex differences in intestinal carbohydrate metabolism promote food intake and sperm maturation. Cell 178, 901–918 (2019).
Middendorp, S. et al. Adult stem cells in the small intestine are intrinsically programmed with their location-specific function. Stem Cells 32, 1083–1091 (2014).
Kayisoglu, O. et al. Location-specific cell identity rather than exposure to GI microbiota defines many innate immune signalling cascades in the gut epithelium. Gut 70, 687–697 (2021).
Kraiczy, J. et al. DNA methylation defines regional identity of human intestinal epithelial organoids and undergoes dynamic changes during development. Gut 68, 49–61 (2019).
McGinnis, C. S. et al. MULTI-seq: sample multiplexing for single-cell RNA sequencing using lipid-tagged indices. Nat. Methods 16, 619–626 (2019).
Moor, A. E. et al. Spatial reconstruction of single enterocytes uncovers broad zonation along the intestinal villus axis. Cell 175, 1156–1167 (2018).
Tibshirani, R., Walther, G. & Hastie, T. Estimating the number of clusters in a data set via the gap statistic. J. R. Stat. Soc. Ser. B Stat. Methodol. 63, 411–423 (2001).
Peng, M., Li, Y., Wamsley, B., Wei, Y. & Roeder, K. Integration and transfer learning of single-cell transcriptomes via cFIT. Proc. Natl Acad. Sci. USA 118, e2024383118 (2021).
Sullivan, Z. A. et al. γδ T cells regulate the intestinal response to nutrient sensing. Science 371, eaba8310 (2021).
Enriquez, J. R. et al. A dietary change to a high-fat diet initiates a rapid adaptation of the intestine. Cell Rep. 41, 111641 (2022).
Goda, T. Regulation of the expression of carbohydrate digestion/absorption-related genes. Br. J. Nutr. 84, S245–S248 (2000).
Clara, R. et al. Metabolic adaptation of the small intestine to short- and medium-term high-fat diet exposure. J. Cell. Physiol. 232, 167–175 (2017).
Ko, C.-W., Qu, J., Black, D. D. & Tso, P. Regulation of intestinal lipid metabolism: current concepts and relevance to disease. Nat. Rev. Gastroenterol. Hepatol. 17, 169–183 (2020).
Gebert, N. et al. Region-specific proteome changes of the intestinal epithelium during aging and dietary restriction. Cell Rep. 31, 107565 (2020).
Biton, M. et al. T helper cell cytokines modulate intestinal stem cell renewal and differentiation. Cell 175, 1307–1320 (2018).
Maimets, M. et al. Mesenchymal-epithelial crosstalk shapes intestinal regionalisation via Wnt and Shh signalling. Nat. Commun. 13, 715 (2022).
Spence, J. R., Lauf, R. & Shroyer, N. F. Vertebrate intestinal endoderm development. Dev. Dyn. 240, 501–520 (2011).
Thompson, C. A., DeLaForest, A. & Battle, M. A. Patterning the gastrointestinal epithelium to confer regional-specific functions. Dev. Biol. 435, 97–108 (2018).
Thompson, C. A. et al. GATA4 is sufficient to establish jejunal versus ileal identity in the small intestine. Cell Mol. Gastroenterol. 3, 422–446 (2017).
Chen, C., Fang, R. X., Davis, C., Maravelias, C. & Sibley, E. Pdx1 inactivation restricted to the intestinal epithelium in mice alters duodenal gene expression in enterocytes and enteroendocrine cells. Am. J. Physiol. Gastrintest. Liver Physiol. 297, G1126–G1137 (2009).
Battle, M. A. et al. GATA4 is essential for jejunal function in mice. Gastroenterology 135, 1676–1686 (2008).
Bosse, T. et al. Gata4 is essential for the maintenance of jejunal-ileal identities in the adult mouse small intestine. Mol. Cell. Biol. 26, 9060–9070 (2006).
Keenan, A. B. et al. ChEA3: transcription factor enrichment analysis by orthogonal omics integration. Nucleic Acids Res. 47, W212–W224 (2019).
Aibar, S. et al. SCENIC: single-cell regulatory network inference and clustering. Nat. Methods 14, 1083–1086 (2017).
Zorn, A. M. & Wells, J. M. Vertebrate endoderm development and organ formation. Annu. Rev. Cell Dev. Biol. 25, 221–251 (2009).
Verzi, M. P., Shin, H., Ho, L. L., Liu, X. S. & Shivdasani, R. A. Essential and redundant functions of caudal family proteins in activating adult intestinal genes. Mol. Cell. Biol. 31, 2026–2039 (2011).
Hryniuk, A., Grainger, S., Savory, J. G. A. & Lohnes, D. Cdx function is required for maintenance of intestinal identity in the adult. Dev. Biol. 363, 426–437 (2012).
Bonhomme, C. et al. Cdx1, a dispensable homeobox gene for gut development with limited effect in intestinal cancer. Oncogene 27, 4497–4502 (2008).
Doktorova, M. et al. Intestinal PPARdelta protects against diet-induced obesity, insulin resistance and dyslipidemia. Sci. Rep. 7, 846 (2017).
Beyaz, S. et al. High-fat diet enhances stemness and tumorigenicity of intestinal progenitors. Nature 531, 53–58 (2016).
Mana, M. D. et al. High-fat diet-activated fatty acid oxidation mediates intestinal stemness and tumorigenicity. Cell Rep. 35, 109212 (2021).
Seiler, K. M. et al. Single-cell analysis reveals regional reprogramming during adaptation to massive small bowel resection in mice. Cell Mol. Gastroenterol. Hepatol. 8, 407–426 (2019).
Nusse, Y. M. et al. Parasitic helminths induce fetal-like reversion in the intestinal stem cell niche. Nature 559, 109–113 (2018).
Schneider, C. et al. A metabolite-triggered tuft cell-ILC2 circuit drives small intestinal remodeling. Cell 174, 271–284 (2018).
Cheng, C. W. et al. Ketone body signaling mediates intestinal stem cell homeostasis and adaptation to diet. Cell 178, 1115–1131 (2019).
Stine, R. R. et al. PRDM16 maintains homeostasis of the intestinal epithelium by controlling region-specific metabolism. Cell Stem Cell 25, 830–845 (2019).
Obniski, R., Sieber, M. & Spradling, A. C. Dietary lipids modulate notch signaling and influence adult intestinal development and metabolism in Drosophila. Dev. Cell 47, 98–111 (2018).
Gajendran, M., Loganathan, P., Catinella, A. P. & Hashash, J. G. A comprehensive review and update on Crohn’s disease. Dis. Mon. 64, 20–57 (2018).
Pan, S. Y. & Morrison, H. Epidemiology of cancer of the small intestine. World J. Gastrointest. Oncol. 3, 33–42 (2011).
Schottenfeld, D., Beebe-Dimmer, J. L. & Vigneau, F. D. The epidemiology and pathogenesis of neoplasia in the small intestine. Ann. Epidemiol. 19, 58–69 (2009).
Brown, E. M., Clardy, J. & Xavier, R. J. Gut microbiome lipid metabolism and its impact on host physiology. Cell Host Microbe 31, 173–186 (2023).
Tian, H. et al. A reserve stem cell population in small intestine renders Lgr5-positive cells dispensable. Nature 478, 255–259 (2011).
Huch, M. et al. In vitro expansion of single Lgr5+ liver stem cells induced by Wnt-driven regeneration. Nature 494, 247–250 (2013).
Platt, R. J. et al. CRISPR-Cas9 knockin mice for genome editing and cancer modeling. Cell 159, 440–455 (2014).
Madisen, L. et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat. Neurosci. 13, 133–140 (2010).
Smillie, C. S. et al. Intra- and inter-cellular rewiring of the human colon during ulcerative colitis. Cell 178, 714–730 (2019).
Melsted, P. et al. Modular, efficient and constant-memory single-cell RNA-seq preprocessing. Nat. Biotechnol. 39, 813–818 (2021).
Lun, A. T. L. et al. EmptyDrops: distinguishing cells from empty droplets in droplet-based single-cell RNA sequencing data. Genome Biol. 20, 63 (2019).
Zhu, Q., Conrad, D. N. & Gartner, Z. J. deMULTIplex2: Robust Sample Demultiplexing for scRNA-seq (Cold Spring Harbor Laboratory, 2023).
Alquicira-Hernandez, J., Sathe, A., Ji, H. P., Nguyen, Q. & Powell, J. E. scPred: accurate supervised method for cell-type classification from single-cell RNA-seq data. Genome Biol. 20, 264 (2019).
Stuart, T. et al. Comprehensive integration of single-cell data. Cell 177, 1888–1902 (2019).
Hao, Y. et al. Integrated analysis of multimodal single-cell data. Cell 184, 3573–3587 (2021).
Hafemeister, C. & Satija, R. Normalization and variance stabilization of single-cell RNA-seq data using regularized negative binomial regression. Genome Biol. 20, 296 (2019).
Cunningham, F. et al. Ensembl 2022. Nucleic Acids Res. 50, D988–D995 (2022).
Kolde, R. Pheatmap: pretty heatmaps. R package version 1.2 (CRAN.R Project, 2012); https://cran.r-project.org/package=pheatmap
Lin, J. H. Divergence measures based on the Shannon entropy. IEEE Trans. Inf. Theory 37, 145–151 (1991).
Drost, H.-G. Philentropy: information theory and distance quantification with R.J. Open Source Softw 3, 765 (2018).
Kotliar, D. et al. Identifying gene expression programs of cell-type identity and cellular activity with single-cell RNA-Seq. eLife 8, e43803 (2019).
Wu, T. Z. et al. clusterProfiler 4.0: a universal enrichment tool for interpreting omics data. Innovation 2, 100141 (2021).
van de Sande, B. et al. A scalable SCENIC workflow for single-cell gene regulatory network analysis. Nat. Protoc. 15, 2247–2276 (2020).
Street, K. et al. Slingshot: cell lineage and pseudotime inference for single-cell transcriptomics. BMC Genomics 19, 477 (2018).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Bankhead, P. et al. QuPath: Open source software for digital pathology image analysis. Sci. Rep. 7, 16878 (2017).
Castillo-Azofeifa, D. et al. Atoh1+ secretory progenitors possess renewal capacity independent of Lgr5+ cells during colonic regeneration. EMBO J. 38, e99984 (2019).
McKinley, K. L. Employing CRISPR/Cas9 genome engineering to dissect the molecular requirements for mitosis. Methods Cell. Biol. 144, 75–105 (2018).
Gilbert, L. A. et al. Genome-scale CRISPR-mediated control of gene repression and activation. Cell 159, 647–661 (2014).
Adamson, B. et al. A multiplexed single-cell CRISPR screening platform enables systematic dissection of the unfolded protein response. Cell 167, 1867–1882 (2016).
Sanjana, N. E., Shalem, O. & Zhang, F. Improved vectors and genome-wide libraries for CRISPR screening. Nat. Methods 11, 783–784 (2014).
Koo, B. K. et al. Controlled gene expression in primary Lgr5 organoid cultures. Nat. Methods 9, 81–83 (2012).
Bray, N. L., Pimentel, H., Melsted, P. & Pachter, L. Near-optimal probabilistic RNA-seq quantification. Nat. Biotechnol. 34, 525–527 (2016).
Risso, D., Ngai, J., Speed, T. P. & Dudoit, S. Normalization of RNA-seq data using factor analysis of control genes or samples. Nat. Biotechnol. 32, 896–902 (2014).
Acknowledgements
We are grateful to H. Miyazaki, D. Castillo-Azofeifa and other members of the Klein laboratory for valuable discussions, experimental assistance and protocol development. We thank M. Helmrath, N. Shroyer, Y.-H. Lo and members of the Intestinal Stem Cell Consortium for critical scientific input throughout this project. We also thank B. Ohlstein, I. Chen, K. Bahar Halpern, Z. Sullivan, D. Conrad, J. Sheu-Gruttadauria, J. Bush, and E. Chow for sharing data, resources and expertise. This study benefited from the following cores and facilities at UCSF: the Center for Advanced Technology, the Institute for Human Genetics, Parnassus Flow Cytometry Core, ViraCore, VIable Tissue Acquisition Lab, the Biological Imaging Development CoLab, and the Laboratory Animal Resource Center. Portions of schematic figure panels were created with BioRender.com. This work was funded by NIH R35-DE026602 and U01DK103147 from the Intestinal Stem Cell Consortium, a collaborative research project funded by the National Institute of Diabetes and Digestive and Kidney Diseases and the National Institute of Allergy and Infectious Diseases (to O.D.K.). R.K.Z. was supported by NIH F32 DK125089 and an American Cancer Society—South Florida Research Council Postdoctoral Fellowship (PF-20-037-01-DDC). Finally, our most sincere gratitude goes to Donor Network West, and to the organ donors and their families for their generosity in supporting basic science research.
Author information
Authors and Affiliations
Contributions
R.K.Z. and O.D.K. conceived and developed the study. R.K.Z. conceived and planned experiments. D.B. conceived several computational approaches, and supervised and verified the analytical methods. R.K.Z., C.S.M., S.I., D.V. and D.B. developed the analysis strategy and performed data analysis. R.K.Z., P.K., B.P., S.V., L.W., K.L.M., A.R., V.N., C.T. and E.R. carried out experiments. A.R.G. and J.M.G. facilitated the human intestinal tissue donation. Z.J.G., R.M.L., J.M.G. and S.I. provided intellectual review of the project content. R.K.Z., D.B. and O.D.K. wrote the manuscript with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Cell Biology thanks Alessandro Ori and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Quality control and initial processing of mouse scRNA-seq data.
a,b Quality control metrics of data, including number of genes detected (‘nFeature_RNA’), number of unique molecular identifiers detected (‘nCount_RNA’), and percent mitochondrial reads (‘% mito GE) before (a) and after (b) processing data. c-e UMAP of total murine epithelial cells sequenced post-QC, coloured according to mouse identity (c), cell type annotation (d), or cell cycle phase (e). f Frequency of epithelial cells of indicated subtype by segment. QC, quality control, mito, mitochondrial; GE, gene expression; ISC, intestinal stem cell; TA, transit amplifying; G1, growth 1; G2M, growth 2 mitosis; S, synthesis.
Extended Data Fig. 2 Quality control and initial processing of human scRNA-seq data from human subject 2.
a,b Quality control metrics of data, including number of genes detected (‘nFeature_RNA’), number of unique molecular identifiers detected (‘nCount_RNA’), and percent mitochondrial reads (‘% mito GE’) before (a) and after (b) processing data. c UMAP of total human cells sequenced post-QC, highlighting cell type annotation. d Frequency of cells of all epithelial subtypes by segment pair. QC, quality control; mito, mitochondrial; GE, gene expression; ISC, intestinal stem cell; TA, transit amplifying.
Extended Data Fig. 3 Zonation across multiple axes of the small intestine.
a UMAP of absorptive lineage cells coloured by segment number along the proximal to distal axis in mouse and human donors. Major epithelial cell types are labeled. b-e Villus zonation across murine enterocytes. b UMAP plots coloured according to summed expression of previously reported21 landmarks of the villus tip (left) or base of villus (right). An equal number of enterocytes were assigned to each of 6 crypt:villus zones, zones 1–6. c UMAP plots coloured according to the expression of select top and bottom villus markers. d UMAP plots coloured according to villus zonation scores (left) compared to segment positions (right). Villus zonation scores represent the ratio of the summed expression of bottom and top landmark genes. e Expression of select villus zonation markers across crypt:villus zones. Center lines represent zone mean, and are coloured by domain with surrounding grey standard error bands. M-, microfold.
Extended Data Fig. 4 Stability and features of five domains across the mouse and human small intestine.
a Right: Average expression of the top 150 upregulated genes in enterocytes from human donor 1 in each segment, with segment order and hierarchical clustering based on expression distance between segments. Vertical white lines show the five domains that divide the small intestine, based on (center) gap statistics for hierarchical clusters of enterocytes in regional gene expression distance. Data bars are presented as mean values +/− confidence interval, based on all cells within the sample. Right: Cuts of dendrogram with optimal cluster number (magenta bracket, center). b Most highly regionalized genes expressed by enterocytes in mouse and donor 2 as in Fig. 1f,g but with a smaller number of genes displayed (75–100), as indicated on the y-axis. c Jensen-Shannon Divergence between enterocytes from segment pairs across the intestine of each individual mouse, with segment pair order and hierarchical clustering based on divergence values between segments. d Murine villus height by domain, presented as mean values +/− standard error of mean. Villus base to tip distances were measured for 3–5 villi in each segment, for each of 4 mice. Statistical significance was calculated using one-way ANOVA followed by Tukey’s multiple comparisons test for villus heights across all segments in each domain. *P < 0.05, ****P < 0.0001, ns not significant. e Domain-defining gene expression scores for human donor 1, as in Fig. 2c,d, coloured by domain with surrounding grey standard error bounds, across intestinal segments. Positions of domain boundaries calculated in b are noted with dotted lines and brackets. f Expression of key domain marker genes in mouse enterocytes across segments. The segment positions of each domain designation are indicated (bottom).
Extended Data Fig. 5 Single-molecule ISH validation of additional domain markers.
a,b Full-length murine intestinal tissue coiled from the proximal (outside) end to the distal (inside) end, probed with single-molecule ISH for select marker genes of domains as indicated. Channels are shown both individually and merged with pseudocolouring. White boxes indicate insets. Scale bars are 2 mm, and 100 μm for insets. Similar results obtained with 3 mice.
Extended Data Fig. 6 Domain marker expression in human tissue.
a Single channels of multi-channel images in Fig. 3b. Data are human tissue sections from indicated domains probed using single-molecule ISH with domain marker genes. Scale bars are 100 μm. b Quantification of mean fluorescence per domain for each donor, presented as mean values +/− standard error of mean. n = 3 or 4 donors per domain as indicated by number of datapoints. One-way ANOVA was performed to compare mean fluorescence in each donor by domain, p values for each marker are labeled.
Extended Data Fig. 7 Functional pathways enriched in domain-associated NMF gene modules in mouse and human.
a,b Selected enriched functional pathways in each NMF gene module displayed in Fig. 2e,f in (a) mouse and (b) human. All gene modules with a regionally variable expression profile across segments that contained genes that encode aspects of nutrient metabolism are displayed (8 modules per species, dotted vertical lines). Module labels (bottom) are the domain(s) most closely-associated with each module, as determined by regional expression profile and rank of key domain-associated signature genes. Pathways were edited to remove redundancy. P values were adjusted for multiple comparisons using the Benjamini-Hochberg procedure.
Extended Data Fig. 8 Divisions between regional intestinal stem cells (ISCs).
a Jensen-Shannon Divergence between ISCs from segment pairs across the intestine, with segment pair order and hierarchical clustering based on divergence values between segments. Dotted red line indicates level of hierarchical tree of domain divisions. b,c Full-length murine intestinal tissue coiled from the proximal (outside) end to the distal (inside) end, probed with single-molecule ISH for select regional ISC marker genes (as in Fig. 5d) as indicated. Channels are shown both individually and merged with pseudocolouring. White boxes indicate insets. Scale bars are 2 mm, and 100 μm for insets. Similar results obtained with 3 mice. d Expression of regional ISC marker genes in absorptive lineage cells. Dot colour reflects average expression, dot size reflects the percent of cells of each type expressing the marker. 'Mature' and 'progenitor' refer to enterocyte state. e Expression of ISC region 1 genes (Gkn3 and Hmgcs2) and ISC region 3 genes (Bex1 and Hoxb6) across ISCs from 15 segments collected from the small intestines of mice fed chow, high-carbohydrate, or high-fat diets as indicated by colour. (n = 3 mice per diet group). ISC, intestinal stem cell; TA, transit amplifying.
Extended Data Fig. 9 Top candidate regulators of domain identity.
a,b Domain-wise expression levels of 5 candidate regulators of domain A and B identities (a) and 15 candidate regulators of domain D and E identities (b), identified using ChEA3 and SCENIC analyses. c,d Expression trajectories of indicated factors, coloured according to inferred differentiation stage in Fig. 6c. Transcription factor expression trajectories were plotted for cells in domain E. Plots are grouped according to expression by early-lineage cells (c) or differentiated cells (d).
Extended Data Fig. 10 Generation and analysis of Ppar-δ and Cdx1 mutant domain E organoids.
a, b Schematics of CRISPR/Cas9 gene targeting strategy. Cas9 endonuclease was encoded in an endogenous genomic locus and 4-hydroxytamoxifen-induced (strategy 1) or delivered by lentiviral vector (strategy 2). Target-specific sgRNAs were delivered by lentiviral vectors (strategies 1 and 2) to induce mutations in the protein coding regions of the target genes. Following mutagenesis, selected clones were expanded and genotyped. Clones containing exclusively deleterious alleles were used for downstream analysis. c Cdx1 mutant organoid sequences from CRISPR editing strategy 1 (‘batch 1’, n = 1 mutant line from mouse 1) and 2 (‘batch 2’, n = 3 unique mutant lines from mouse 2), and Ppar-δ mutant organoid sequences from editing strategy 1 (‘batch 1’, n = 2 unique mutant lines from mouse 1) and 2 (‘batch 2’, n = 3 unique mutant lines from mouse 2). Indel mutations are specified. d Trend towards decreased expression of Fabp6 in Cdx1 mutant lines in both batches of mRNAseq expression data from editing strategies 1 and 2, which could not be merged. Line represents median. e Expression of differentially expressed genes in individual Ppar-δ mutant organoid lines from batch 1 mutants (red dots) and control organoid lines (black dots). Batch 2 expression data of these and other DEGs in Fig. 6f,h. f Normalized mRNA levels of select DEGs of interest in Ppar-δ mutant organoids, validated with real time PCR. (n = 2–4 technical replicates per one control and two mutant organoid lines as indicated). bp, base pair; DEGs, differentially expressed genes.
Supplementary information
Supplementary Information
Supplementary Figs. 1–9 with legends.
Supplementary Table 1
Significantly variable marker genes of mouse and human secretory cells.
Supplementary Table 2
Differentially expressed gene signatures of mouse small intestinal domains identified using Wilcoxon rank sum test.
Supplementary Table 3
Differentially expressed gene signatures of human small intestinal domains identified using Wilcoxon rank sum test.
Supplementary Table 4
Non-negative matrix factorization (NMF) gene modules.
Supplementary Table 5
Functional profiles of mouse intestinal domain gene signatures.
Supplementary Table 6
Differentially expressed gene signatures of murine regional ISCs.
Supplementary Table 7
Candidate TF regulators of domain gene signatures in enterocytes predicted with ChEA3.
Supplementary Table 8
Candidate TF regulators of gene signatures in enterocytes predicted with SCENIC.
Supplementary Table 9
Datasets for cFIT analysis.
Supplementary Table 10
Primer sequences.
Source data
Source Data
Source data
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zwick, R.K., Kasparek, P., Palikuqi, B. et al. Epithelial zonation along the mouse and human small intestine defines five discrete metabolic domains. Nat Cell Biol 26, 250–262 (2024). https://doi.org/10.1038/s41556-023-01337-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41556-023-01337-z