Abstract
Background
Contrast-induced neurotoxicity (CIN) is an increasingly recognised complication following endovascular procedures utilising contrast. It remains poorly understood with heterogenous clinical management strategies. The aim of this review was to identify commonly employed treatments for CIN to enhance clinical decision making.
Methods
A systematic search of Embase (1947–2022) and Medline (1946–2022) was conducted. Articles describing (i) patients with a clinical diagnosis of CIN, (ii) with radiological exclusion of other pathologies, (iii) detailed report of treatments, and (iv) discharge outcomes, were included. Data relating to demographics, procedure, symptoms, treatment and outcomes were extracted.
Results
A total of 73 patients were included, with a median age of 64 years. The most common procedures were cerebral angiography (42.5%) and coronary angiography (42.5%), and the median volume of contrast administered was 150 ml. The most common symptoms were cortical blindness (38.4%) and reduced consciousness (28.8%), and 84.9% of patients experienced complete resolution at the time of discharge. Management included intravenous fluids to dilute contrast in the cerebrovasculature (54.8%), corticosteroids to reduce blood–brain barrier damage (47.9%), antiseizure (16.4%) and sedative (16.4%) medications. Mannitol (13.7%) was also utilised to reduce cerebral oedema. Intensive care admission was required for 19.2% of patients. No statistically significant differences were observed between treatment and discharge outcomes.
Conclusions
The clinical management of CIN should be considered on a patient-by-patient basis, but may consist of aggressive fluid therapy alongside corticosteroids, as well as other supportive therapy as required. Further examination of CIN management is required to define best practice.
Similar content being viewed by others
Introduction
With advancements in technology and techniques allowing access to a growing spectrum of pathologies, the rate of endovascular procedures in clinical practice has experienced exponential growth [1, 2]. Contrast-induced neurotoxicity (CIN) is an increasingly recognised complication of procedures requiring iodinated contrast, that presents as a range of neurological symptoms that typically mimics ischaemic stroke, including sensory and motor deficit, aphasia, cortical blindness, and reduced consciousness [3,4,5].
Contrast-induced neurotoxicity remains a poorly understood clinical entity, with a lack of formalised diagnostic criteria and evidence-base regarding management. A recent survey of clinicians demonstrated that less than 25% were comfortable in treating CIN, and 82.1% agreed that further investigation was required to enhance treatment strategies [6]. In light of this, we conducted this systematic review with the aim of characterising currently employed management strategies, with the ultimate goal of enhancing clinical decision making and patient outcomes.
Methods
Ethical approval
This study was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [7]. Ethical approval and patient consent were not required for this study.
Search strategy
A comprehensive literature search of Medline (1946 to December 2022) and Embase (1947 to December 2022) was performed from inception. Key search terms included “contrast”, “neurotoxicity”, “encephalopathy”, “blindness’, and “deficit”, with Boolean operators employed as appropriate. Reference lists of selected papers were also screened to identify additional publications, and duplicate articles were removed.
Eligibility criteria
Studies were selected for analysis based on the following inclusion criteria: publications reporting (i) patients with a clinical diagnosis of CIN, (ii) with radiological exclusion (CT or MR brain imaging) of other pathologies (most notably ischaemic or haemorrhagic stroke), (iii) sufficient reporting of treatments administered, and (iv) reporting of discharge outcomes. Reports of patients < 18 years of age were excluded from analysis, as well as conference abstracts, case series from which it was not possible to extract individual data, and non-English publications. For the purposes of this review, CIN was defined as the onset of neurological symptoms following iodinated contrast administration, with clinical, biochemical and radiological exclusion of other pathologies, most notably ischaemic stroke. Reports in which it was unclear if CIN was the primary explanation for the symptoms presented (i.e., other differentials were not adequately investigated) were also excluded.
Screening process
Two investigators (FM and JXC) independently evaluated studies for eligibility according to the eligibility criteria. Titles and abstracts were screened initially. Full text reports were then examined. Where consensus was not able to achieved, a third investigator was consulted. The systematic review platform Covidence (www.covidence.org; Veritas Health Innovation, Melbourne, Australia) was used to facilitate the screening process. Publications that fulfilled eligibility criteria underwent data extraction.
Data extraction
Data were extracted by two independent investigators (FM and JXC), and were crosschecked. In the event of discrepancy, further discussion and examination was conducted until consensus was reached. An additional investigator was consulted when consensus was not able to be achieved. Extracted data including demographic and procedural variables including age, sex, country of publication, comorbidities, procedure, indication of procedure, contrast type, and contrast volumes were collected. The clinical signs and symptoms of CIN, along with relevant imaging findings, and discharge outcomes were also extracted. Details of clinical management were collected including medications administered, procedures performed and requirement of intensive care admission.
Outcome measures
The clinical presentation of CIN was separated into individual symptoms. Likewise, management regimens were categorised into individual medications and treatment. Patients requiring mechanical ventilation were assumed to require intensive care support. Patient outcomes were based on symptoms at time of discharge from hospital. Favourable outcome was defined as complete resolution at time of discharge from hospital. Unfavourable outcome was defined as ongoing symptoms at time of discharge or death.
Quality assessment
Quality assessment of included publications was conducted using a modified version of the Methodological Quality and Synthesis of Case Series and Case Reports eight-item questionnaire proposed by Murad et al. [8], which encompasses the domains of selection, ascertainment, causality, and reporting. Reporting items included in the modified tool were the type of contrast administered, time to onset of CIN, radiological exclusion of other pathology, volume of contrast administered, and time course of CIN symptoms. Two independent investigators (FM and JXC) individually assessed all included publications according to the modified tool.
Statistical analysis
Included cases were pooled and descriptive analysis was performed for patient demographics, comorbidities, procedural details, clinical symptoms of CIN, administered treatments, and discharge outcomes. Administered treatments were assessed for association with length of CIN symptoms and discharge outcomes. Fisher’s exact test and Chi-squared tests were utilised where appropriate to evaluate association between categorical variables. Statistical significance was defined as a p value < 0.05. All statistical analyses were performed with Stata/BE (StataCorp LLC, College Station, Texas, USA).
Results
Study selection
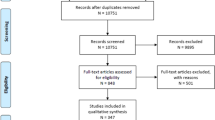
Our search strategy yielded 1059 articles, and after removal of duplicates, 733 references were screened by title and abstract (Fig. 1). A total of 169 were eligible for full text screening, of which 110 were excluded with reasons. Finally, 59 articles (Supplementary Data 1) were included for final analysis, with a pooled sample size of 73 patients.
Population characteristics
The median patient age was 64 years (range: 22–89), with 37 (50.7%) males, and 36 (49.3%) females (Table 1). The year of publication ranged from 1995 to 2022, with cases originating from 27 countries. The most commonly reported comorbidities included hypertension (58.9%), diabetes (27.4%), and hyperlipidaemia (24.7%).
Procedural characteristics
Patients underwent a variety of contrast-requiring procedures (Table 2), most notably cerebral angiography with or without intracranial intervention (42.5%), and coronary angiography with or without intervention (42.5%). The most commonly administered contrast agents were iodixanol (20.5%) iopamidol, (17.8%), and iohexol (16.4%). The median volume of contrast administered was 150 ml, with cases reported following as little as 18 ml of contrast.
Clinical course
Commonly reported symptoms of CIN included cortical blindness (38.4%), reduced consciousness (28.8%), hemiparesis (27.4%), confusion (26.0%), and aphasia (23.3%) (Table 3). The median time of symptom onset was 1-h post-procedure, with symptoms appearing as early as intraprocedurally and as late as 27 h following the procedure. At the time of discharge, complete resolution of symptoms was reported in 62 patients (84.9%), nine (12.3%) were discharged with residual deficits, and two patients (2.7%) were deceased. In patients with complete resolution of symptoms, 17 (23.3%) lasted 24 h or less, 25 (34.2%) between 24 and 72 h, and 16 (21.9%) longer than 72 h, with 4 (5.5%) not reporting a timeframe.
Management
Several treatment options were reported (Table 4). The most commonly employed treatments included intravenous fluids (54.8%), corticosteroids (47.9%), sedatives (16.4%) and antiseizure medications (ASMs) (16.4%). ASMs were initiated as seizure prophylaxis in 33.3%. Mannitol was utilised in 13.7% of patients, presumably for management of intracranial pressure and cerebral oedema. Seven (9.6%) patients received calcium channel blockers (CCBs). Four patients (5.5%) underwent haemodialysis, all of whom had pre-existing chronic kidney disease. Antipsychotics were administered in 4 patients (5.5%) for management of agitation and confusion. A total of 14 patients (19.2%) required intensive care admission, and 11 (15.1%) required intubation.
The correlation between administered treatments and the clinical outcomes of CIN was assessed. Univariate analysis demonstrated no significant associations between treatments and the outcomes of patients at the time of hospital discharge (Table 5). When limited to a cohort of patients with complete resolution of their symptoms at discharge, administration of mannitol administration was associated with a longer CIN clinical course (p = 0.044), with no association detected with other medications (Table 6). The volume of contrast was also not associated with length of symptoms (p = 0.774) or overall discharge outcome (p > 0.999). Furthermore, a comparison between cardiac and cerebral interventions demonstrated no difference in length of symptoms (p = 0.537), or outcomes (p = 0.053).
Quality assessment
Of the 59 included publications, 6 (10.2%) reported the selection criteria and specified the incidence at their institution (Table 7). The type of contrast was reported by 89.8% of publications, and the timing of symptom onset was reported in 79.7%. All included publications provided radiological exclusion of other acute intracranial pathologies. The volume of contrast was reported in 89.8% and the outcome and duration of CIN was specified in 86.4% of papers. Only 34 publications (57.6%) were deemed to provide sufficient details to replicate practice.
Discussion
Summary of evidence
In this review of 59 articles describing 73 cases of CIN, we found that the mainstays of clinical management consisted of intravenous fluids (54.8%), corticosteroid therapy (47.9%), with other frequently described medications, including mannitol, ASMs and sedatives. One in four patients (19.2%) were admitted into an intensive care unit, and 11 patients (15.1%) required tracheal intubation. Reported treatments had no observed statistical effect on discharge outcomes.
The commonest symptoms of CIN were hemiparesis, cortical blindness, and reduced consciousness, with a median onset of symptoms 1-h post-procedure. Complete resolution of symptoms was reported in 84.9% of cases.
Pathophysiological mechanisms
The pathophysiology of CIN is unclear, although it has been suggested that the blood–brain barrier (BBB) plays a vital role [9, 10]. Disruption of the BBB allows passage of contrast agents into the central nervous system, allowing it to potentiate neurotoxic effects. One theory for this relates to the oncotic action of hyperosmolar contrast agents leading to shrinkage of endothelial cells and subsequent opening of the tight junctions [11, 12]. Nonetheless, this is unlikely to be the only cause of BBB dysfunction, with CIN observed in patients administered low/iso-osmolar contrast agents. Other factors including increased shear stress caused by hypertension [13], as well as reduced BBB integrity following ischaemic stroke have also been suggested to cause disruption of the BBB [9], allowing passage of contrast agents. In the current review, almost two in three patients were reported to have hypertension, and 10.3% were reported to have suffered a prior ischaemic stroke.
Principles of management
Intravenous fluids
The clinical manifestation of CIN is thought to occur due to the direct effects of contrast on neural cellular function [14]. In addition, several studies have suggested that damage to the BBB secondary to contrast media is directly proportional to the concentration and the length of time the cerebrovasculature is exposed to contrast [15]. As such, reducing the exposure and concentration of contrast agents in cerebral vessels may minimise the effects of contrast on the CNS. Aggressive administration of intravenous fluids may act to dilute and subsequently accelerate the removal contrast agents from the cerebral vascular system. In the current study, intravenous fluids were reportedly used in 55% of patients. Given the widespread use of intravenous fluid therapy in normal clinical practice, it is likely that this number is much higher and was not specified in some case reports.
Corticosteroids
Corticosteroids were administered in 47.9% of CIN patients. Glucocorticoids are known to reduce inflammation, and have long been used in inflammatory conditions affecting the CNS, including infective and autoimmune disease processes [16,17,18,19]. Corticosteroids have also been demonstrated to increase the integrity of the BBB by enhancing recovery and upregulating synthesis of BBB tight junction proteins [20, 21]. As aforementioned, the BBB appears to play a key role in the pathogenesis of CIN. By reducing damage to the BBB, and potentially preventing the entry of contrast agents, corticosteroids may play an important role in CIN management.
Mannitol
In some instances, the signs and symptoms of CIN have reported to be associated with cerebral oedema [22, 23], which is likely due to changes in oncotic pressure following the extravasation of contrast. Following breakdown and passage of contrast agents through the BBB, the relative hyperosmolarity of contrast media will cause a shift between fluid compartments [24]. In the current study, the use of mannitol was seen to be associated with prolonged CIN symptom course (> 72 h). The most likely explanation for this would be that mannitol was utilised in patients with more severe or prolonged CIN. The use of mannitol to lower intracranial pressure and cerebral oedema is already a standard therapeutic option in the management of neurological conditions [25, 26]. The targeted use of mannitol in patients with cerebral oedema secondary to CIN would be expected to improve symptoms associated with the localised effects of cerebral oedema as well as the compression of neural structures secondary to raised intracranial pressure. The findings of this review, ultimately, cannot confirm the benefits of mannitol use in CIN, but may be able to guide management in patients with cerebral oedema.
Calcium channel blockers
Calcium channel blockers were reported in approximately 9.6% of cases, and were presumably used to prevent vasospasm. Although the current definition of CIN remains unclear, cerebral vasospasm is a neurological pathology in and of itself, being a major cause of mortality and neurological morbidity [27, 28]. It may be possible that contrast agents induce vasospasm, however, patients with suspected vasospasm following endovascular procedures should be categorised separately to CIN patients, with an arsenal of treatment options and increasingly evidence-based management strategies available to clinicians [27,28,29,30]. Further study and understanding of the pathophysiology is required to rationalise the use of CCBs in CIN.
Antiseizure medications
The administration of ASMs were primarily used in the management of patients experiencing seizure activity, with two-thirds of patients in this review who received ASM therapy experiencing active seizures as part of their clinical manifestation. The remaining 33.3% were administered ASMs for seizure prophylaxis. Sedating agents, such as benzodiazepines, were also reported in the treatment of seizures associated with CIN. Antiseizure medications are already commonly used prophylactically in neurosurgery and neurotrauma [31,32,33,34]. In the current review, seizures were experienced by 16.4% of patients, and forms a recognised part of the clinical picture of CIN. The use of ASMs for seizure prophylaxis in patients with CIN may be warranted, although requires careful consideration of the patient’s clinical state and possible adverse effects.
Clinical implications
In the clinical management of CIN, the most important step is to exclude other acute intracranial pathologies that require emergent treatment, most notably ischaemic stroke. Once a diagnosis of CIN is clear, several supportive and therapeutic options may be available. Corticosteroids to reduce inflammation and reduce BBB damage, as well as aggressive intravenous fluids to dilute and remove contrast from the cerebrovascular are likely to form the foundation of CIN management moving forward. Depending on the clinical scenario, mannitol may also be appropriate to reduce cerebral oedema. Other medications including ASMs for seizure prophylaxis should also be considered. In patients with high clinical concern or reduced consciousness, admission into an intensive care unit for supportive care and close observation would be appropriate.
In clinical practice, it is vital to individualise treatment. This principle is all the more relevant in the context of CIN, given the relative paucity of literature, and the lack of definitive evidence for treatment strategies. As a result, our recommendation would be to evaluate the clinical manifestation of CIN in each patient and decide on therapy on a case-by-case basis. The literature presented may act as a guide to enhance decision-making, but ultimately, each patient should be treated according to their symptoms and clinical state, until further evidence emerges.
Study strengths
This review has several strengths. We applied a very strict eligibility criteria to ensure that all included cases of CIN were as accurate as possible, to increase the certainty from which conclusions could be drawn. Patients with insufficient investigation of other neurological pathologies were excluded. By including cases from a variety of procedures, the findings of this study are more widely applicable to a range of specialties and clinicians. Furthermore, there is a widespread of demographics represented in the cohort of included patients, with cases originating from 27 countries across Asia, Europe, North America, and the Middle East.
Study limitations
This study also has several limitations. A major limitation was the variable quality in reporting of cases. According to our quality assessment, it was deemed that only 57.6% of articles presented sufficient clinical information to replicate practice. As a result of the variability in reporting, the intricacies of individual management options, such as dosage, were not able to be accurately characterised or examined. Furthermore, the vast majority of articles were case reports without a specific focus on treatment, it is possible that certain administered treatments were not reported, such as intravenous fluids. Moreover, comparison between treatment options and patient discharge outcomes showed no statistically significant effect. This may be partially due to the small sample size of this study, but may also be attributable to the heterogeneity in the reporting of patient outcomes. Another limitation of note was that non-English articles were excluded from this study, which may have provided insightful data.
Gaps in knowledge
This systematic review focussing on clinical management has brought to light several important deficiencies in the current understanding of CIN. Although we were able to identify the most commonly reported treatments utilised, the effectiveness and ultimate impact on patient prognosis and recovery has not been established. Furthermore, this investigation was not able to elucidate appropriate dosing and timing of reported treatments. Additionally, improved understanding of the pathophysiology of CIN will aid in the formulation of optimal clinical management strategies. Ultimately, this review only presents the currently utilised treatments for CIN from within the literature, and may not represent optimal management.
Future directions
This study highlights the significant need for further investigation into the treatment of CIN. The overwhelming majority of the literature is formed by case reports and case series, which are low-level evidence, are highly biased, and are very difficult to draw practical conclusions from. Large cohort studies of patients who develop CIN following contrast-requiring procedures are required. In particular, studies should focus on specific treatment strategies and their effects on the short-term recovery, as well as the long-term outcomes of patients. Additional prognostic factors, including procedural details, comorbidities and risk factors, and contrast characteristics should also be methodically examined. Further investigation into the pathophysiology of CIN would not only improve our understanding of it as a clinical entity, but would provide an underpinning to treatment decision making.
Conclusion
The findings of this review suggest that the clinical management of CIN could include aggressive intravenous fluids to reduce cerebrovascular exposure to contrast, corticosteroids to decrease inflammation and BBB disruption, ASMs to control seizure activity, and mannitol to reduce cerebral oedema. Nonetheless, we recommend each patient be treated on a case-by-case basis. Ultimately, the efficacy of different treatment options remain unclear, and larger cohort studies with a specific focus on management are required to define optimal treatment strategies.
Data availability
Not applicable.
References
Andaluz N, Zuccarello M (2008) Recent trends in the treatment of cerebral aneurysms: analysis of a nationwide inpatient database. J Neurosurg 108(6):1163–1169
Salem MM, Maragkos GA, Gomez-Paz S, Ascanio LC, Ngo LH, Ogilvy CS et al (2021) Trends of ruptured and unruptured aneurysms treatment in the United States in post-ISAT era: a national inpatient sample analysis. J Am Heart Assoc 10(4):e016998
Vazquez S, Graifman G, Spirollari E, Ng C, Uddin A, Feldstein E et al (2022) Incidence and risk factors for acute transient contrast-induced neurologic deficit: a systematic review with meta-analysis. Stroke 2(1):e000142
Quintas-Neves M, Araújo JM, Xavier SA, Amorim JM, Silva VCE, Pinho J (2020) Contrast-induced neurotoxicity related to neurological endovascular procedures: a systematic review. Acta Neurol Belg 120:1419–1424
Meijer FJ, Steens SC, Tuladhar AM, van Dijk ED, Boogaarts HD (2022) Contrast-induced encephalopathy—neuroimaging findings and clinical relevance. Neuroradiology 64(6):1265–1268
Mariajoseph FP, Lai LT, Moore J, Chandra RV, Goldschlager T, Praeger A et al (2023) Current knowledge and perspectives of contrast-induced neurotoxicity: a survey of Australian clinicians. J Clin Neurosci 116:8–12
Moher D, Liberati A, Tetzlaff J, Altman DG, Group* P (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 151(4):264–269
Murad MH, Sultan S, Haffar S, Bazerbachi F (2018) Methodological quality and synthesis of case series and case reports. BMJ Evid Based Med 23(2):60–63
Liao M-T, Lin T-T, Lin L-Y, Hwang J-J, Tseng C-D (2013) Contrast-induced encephalopathy after percutaneous coronary intervention. Acta Cardiol Sin 29(3):277
Muruve D, Steinman T (1996) Contrast-induced encephalopathy and seizures in a patient with chronic renal insufficiency. Clin Nephrol 45(6):406–409
Waldron RL, Bridenbaugh RB, Dempsey EW (1974) Effect of angiographic contrast media at the cellular level in the brain: hypertonic vs chemical action. Am J Roentgenol 122(3):469–476
Rapoport S (1973) Reversible opening of the blood-brain barrier by osmotic shrinkage of the cerebrovascular endothelium: opening of the tight junctions as related to carotid arteriography. Small vessel angiography. Mosby, St. Louis, pp 137–151
Harnish PP, Hagberg DJ (1988) Contrast media-induced blood-brain barrier damage potentiation by hypertension. Invest Radiol 23(6):463–465
Junck L, Marshall WH (1983) Neurotoxicity of radiological contrast agents. Ann Neurol 13(5):469–484
Rapoport S, Thompson H, Bidinger JM (1974) Equi-osmolal opening of the blood-brain barrier in the rabbit by different contrast media. Acta Radiol Diagn 15(1):21–32
De Gans J, Van de Beek D (2002) Dexamethasone in adults with bacterial meningitis. N Engl J Med 347(20):1549–1556
Hodzic E, Hasbun R, Granillo A, Tröscher AR, Wagner H, von Oertzen TJ et al (2023) Steroids for the treatment of viral encephalitis: a systematic literature review and meta-analysis. J Neurol 270:3603–3615
Beck RW, Cleary PA, Trobe JD, Kaufman DI, Kupersmith MJ, Paty DW et al (1993) The effect of corticosteroids for acute optic neuritis on the subsequent development of multiple sclerosis. N Engl J Med 329(24):1764–1769
Rudick RA, Cohen JA, Weinstock-Guttman B, Kinkel RP, Ransohoff RM (1997) Management of multiple sclerosis. N Engl J Med 337(22):1604–1611
McMahon D, Oakden W, Hynynen K (2020) Investigating the effects of dexamethasone on blood-brain barrier permeability and inflammatory response following focused ultrasound and microbubble exposure. Theranostics 10(4):1604
Hue CD, Cho FS, Cao S, Bass CRD, Meaney DF, Morrison B III (2015) Dexamethasone potentiates in vitro blood-brain barrier recovery after primary blast injury by glucocorticoid receptor-mediated upregulation of ZO-1 tight junction protein. J Cereb Blood Flow Metab 35(7):1191–1198
Zevallos CB, Dandapat S, Ansari S, Farooqui M, Quispe-Orozco D, Mendez-Ruiz A et al (2020) Clinical and imaging features of contrast-induced neurotoxicity after neurointerventional surgery. World Neurosurg 142:e316–e324
Li J, Qi G, Zhang H, Chen G, Wang S, Yan M et al (2021) Contrast-induced encephalopathy mimicking stroke after a second cerebral DSA: an unusual case report. BMC Neurol 21(1):430
Chu Y-T, Lee K-P, Chen C-H, Sung P-S, Lin Y-H, Lee C-W et al (2020) Contrast-induced encephalopathy after endovascular thrombectomy for acute ischemic stroke. Stroke 51(12):3756–3759
Shawkat H, Westwood M-M, Mortimer A (2012) Mannitol: a review of its clinical uses. Contin Educ Anaesth Crit Care Pain 12(2):82–85
Schwarz S, Schwab S, Bertram M, Aschoff A, Hacke W (1998) Effects of hypertonic saline hydroxyethyl starch solution and mannitol in patients with increased intracranial pressure after stroke. Stroke 29(8):1550–1555
Charpentier C, Audibert G, Guillemin F, Civit T, Ducrocq X, Bracard S et al (1999) Multivariate analysis of predictors of cerebral vasospasm occurrence after aneurysmal subarachnoid hemorrhage. Stroke 30(7):1402–1408
Bauer AM, Rasmussen PA (2014) Treatment of intracranial vasospasm following subarachnoid hemorrhage. Front Neurol 5:72
Komotar RJ, Zacharia BE, Valhora R, Mocco J, Connolly ES Jr (2007) Advances in vasospasm treatment and prevention. J Neurol Sci 261(1–2):134–142
Newell DW, Eskridge JM, Mayberg MR, Grady MS, Winn HR (1989) Angioplasty for the treatment of symptomatic vasospasm following subarachnoid hemorrhage. J Neurosurg 71(5):654–660
Schierhout G, Roberts I (1998) Prophylactic antiepileptic agents after head injury: a systematic review. J Neurol Neurosurg Psychiatry 64(1):108–112
Iuchi T, Kuwabara K, Matsumoto M, Kawasaki K, Hasegawa Y, Sakaida T (2015) Levetiracetam versus phenytoin for seizure prophylaxis during and early after craniotomy for brain tumours: a phase II prospective, randomised study. J Neurol Neurosurg Psychiatry 86(10):1158–1162
Temkin NR (2002) Prophylactic anticonvulsants after neurosurgery. Epilepsy Currents 2(4):105–107
Zafar SN, Khan AA, Ghauri AA, Shamim MS (2012) Phenytoin versus leviteracetam for seizure prophylaxis after brain injury–a meta analysis. BMC Neurol 12:1–8
Acknowledgements
This research did not receive any specific grants from funding agencies in the public, commercial, or not-for-profit sectors.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors has any conflict of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mariajoseph, F.P., Chung, J.X., Lai, L.T. et al. Clinical management of contrast-induced neurotoxicity: a systematic review. Acta Neurol Belg (2024). https://doi.org/10.1007/s13760-024-02474-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13760-024-02474-4