Abstract
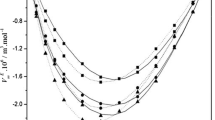
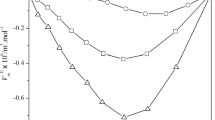
In the present study, measurements of experimental density (ρ), viscosity (η) and speed of sound (u) of binary liquid mixtures of 2-methyl-1-butanol with ethylene glycol monomethyl ether, diethylene glycol monomethyl ether and triethylene glycol monomethyl ether have been carried out within the temperature range of 298.15–308.15 K with an interval of 5 K. Using the experimental data, excess properties such as excess molar volume (\({V}_{m}^{E}\)), excess molar isentropic compressibility (\({K}_{s,m}^{E}\)), excess speed of sound (\({u}^{E}\)), deviation in viscosity (Δη) and excess Gibbs free energy of activation for viscous flow (\(\Delta {G}^{{\ast}E}\)) have been calculated to study the intermolecular interactions present in these liquid mixtures. By comparing the standard deviation values the correlation ability of various proposed viscosity models has also been tested. Further, to rationalize the excess volume data Prigogine–Flory–Patterson (PFP) theory has also been studied.






Similar content being viewed by others
References
M. Jablonský, A. Škulcová, A. Malvis, J. Šima, J. Biotechnol. 46–66, 282 (2018)
A.R. Velez, J.R. Mufari, L.J. Rovetto, Fluid Phase Equilib. 55–63, 497 (2019)
T.H. Kwak, S. Maken, S. Lee, J.W. Park, B.R. Min, Y.D. Yoo, Fuel 2012–2017, 85 (2006)
S. Gahlyan, S. Verma, M. Rani, S. Maken, J. Mol. Liq. 233–240, 244 (2017)
G.P. Dubey, P. Kaur, J. Chem. Eng. Data 2232–2239, 60 (2015)
M. Gupta, I. Vibhu, J.P. Shukla, Fluid Phase Equilib. 26–32, 244 (2006)
I. Mozo, I.G. De La Fuente, J.A. González, J.C. Cobos, J. Chem. Eng. Data 2086–2090, 52 (2007)
H. Piekarski, M. Tkaczyk, P. Goralski, J. Chem. Thermodyn. 259–266, 36 (2004)
E.E. Fenn, D.E. Moilanen, N.E. Levinger, M.D. Fayer, J. Am. Chem. Soc. 5530–5539, 131 (2009)
G.P. Dubey, P. Kaur, J. Chem. Thermodyn. 239–248, 64 (2013)
O. Redlich, A.T. Kister, Ind. Eng. Chem. 345–348, 40 (1948)
M. Papari, H. Ghodrati, F. Fadaei, R. Sadeghi, S. Behrouz, M. Rad, J. Moghadasi, J. Mol. Liq. 121–128, 180 (2013)
F. Kermanpour, H. Jahani, H. Iloukhani, J. Mol. Liq. 29–34, 146 (2009)
L. Vargas-Ibáñez, G. Iglesias-Silva, J. Cano-Gómez, C. Escamilla-Alvarado, M. Berrones-Eguiluz, J. Chem. Eng. Data 2438–2450, 63 (2018)
M. Dzida, Int. J. Thermophys. 55–69, 31 (2010)
A. D’Aprano, D. Donato, V. Agriento, J. Solution Chem. 673–680, 10 (1981)
K. Fukuchi, K. Oginawa, Y. Tashima, S. Yonezawa, Y. Arai, Ube Kogyo Koto Senmon Gakko Kenkyu Hokoku 93–111, 29 (1983)
E. Álvarez, A. Cancela, R. Maceiras, J. Navaza, R. Táboas, J. Chem. Eng. Data 940–945, 51 (2006)
J.M. Resa, J.M. Goenaga, J. Chem. Eng. Data 1892–1895, 51 (2006)
J.M. Resa, C. González, J.M. Goenaga, J. Chem. Eng. Data 73–78, 51 (2006)
J.M. Resa, C. González, J.M. Goenaga, J. Chem. Eng. Data 1570–1575, 50 (2005)
J. George, N.V. Sastry, J. Chem. Eng. Data 977–989, 48 (2003)
I. Mozo, J.A. González, I.G. de la Fuente, J.C. Cobos, N. Riesco, J. Mol. Liq. 87–100, 140 (2008)
L. Sarkar, M.N. Roy, J. Chem. Eng. Data 3307–3312, 54 (2009)
K.V.R. Reddy, K. Rambabu, A. Krishnaiah, M. Ramakrishna, Fluid Phase Equilib. 251–256, 103 (1995)
G.P. Dubey, K. Kumar, J. Chem. Eng. Data 1700–1703, 55 (2010)
A. Pal, B. Kumar, J. Chem. Eng. Data 688–695, 57 (2012)
J.A. Riddick, W.B. Bunger, F.K. Sakano, Organic Solvents Physical Properties and Method of Purification, 4th edn. (Wiley Interscience, New York, 1986)
A. Pal, S. Sharma, J. Chem. Eng. Data 532–536, 43 (1998)
H.A. Zarei, F. Jalili, J. Chem. Thermodyn. 55–66, 39 (2007)
C.M. Kinart, A. Cwiklinska, W.J. Kinart, A. Bald, J. Chem. Eng. Data 1425–1428, 49 (2004)
N. Nishimoto, S. Tabata, K. Tamara, S. Murakamo, Fluid Phase Equilib. 235–247, 136 (1997)
R. Balaji, M.G. Sankar, M.C. Sekhar, M.C. Shekar, J. Mol. Liq. 330–341, 216 (2016)
J.V. Srinivasu, K. Narendra, C. Kavitha, B.S. Rao, J. Solution Chem. 2066–2090, 46 (2017)
M.N. Roy, A. Banerjee, A. Choudhury, Phys. Chem. Liq. 412–436, 47 (2009)
A. Bhattacharjee, M.N. Roy, J. Chem. Eng. Data 5914–5920, 55 (2010)
P.J. Victor, D. Das, D.K. Hazra, J. Indian Chem. Soc. 81, 1045 (2004)
P.K. Muhuri, D.K. Hazara, J. Chem. Eng. Data 582–585, 40 (1995)
C.M. Kinart, A. Ćwiklińska, W.J. Kinart, A. Bald, J. Mol. Liq. 135–139, 126 (2006)
D. Venkatesulu, P. Venkatesu, M.V. Prabhakara Rao, J. Chem. Eng. Data 365–367, 42 (1997)
M.N. Roy, R. Dewan, L. Sarkar, J. Chem. Eng. Data 1347–1353, 55 (2010)
P.J. Victor, D. Das, D.K. Hazra, J. Indian Chem. Soc. 1045–1050, 81 (2004)
M.N. Roy, B.K. Sarkar, R. Chanda, J. Chem. Eng. Data 1630–1637, 52 (2007)
B. Zareena, P.B. Sandhya Sri, D.B. Karuna Kumar, C. Rambabu, J. Mol. Liq. 99–112, 178 (2013)
M.N. Roy, R.S. Sah, P. Pradha, Int. J. Thermophys. 316–326, 31 (2010)
H. Kumar, M. Singla, A. Khosla, J. Solution Chem. 428–440, 42 (2013)
I. Mozo, I.G.D.L. Fuente, J.A. Gonzalez, J.C. Cobos, J. Chem. Eng. Data 2086–2090, 52 (2007)
G.P. Dubey, K.K. Mehra, M. Sharma, J. Chem. Thermodyn. 234–243, 42 (2010)
R. Francesconi, C. Castellari, F. Comelli, J. Chem. Eng. Data 1373–1378, 44 (1999)
X.X. Li, W.D. Zhou, X.Y. Li, J.L. Sun, W. Jiang, J. Mol. Liq. 73–76, 148 (2009)
D. Belhadj, I. Bahadur, A. Negadi, N. Muñoz-Rujas, E. Montero, L. Negadi, J. Chem. Eng. Data 5192–5209, 65 (2020)
C.M. Kinart, K. Nowak, W.J. Kinart, J. Chem. Thermodyn. 423–429, 37 (2005)
A. Pal, S. Sharma, J. Chem. Eng. Data 1067–1070, 44 (1999)
A. Pal, H. Kumar, A. Kumar, G. Dass, Fluid Phase Equilb. 245, 166 (1999)
A. Pal, Y.P. Singh, J. Chem. Eng. Data 425–427, 41 (1996)
G. Douheret, P. Lajoie, M.I. Davis, J.L. Ratliff, J. Ulloa, H. Hoiland, J. Chem. Soc. Faraday Trans. 2291–2298, 91 (1995)
S. Aznarez, M.M.E.F. de Ruiz Holgado, E.L. Arancibia, J. Mol. Liq. 131–137, 139 (2008)
X.X. Li, G.C. Fan, Y.W. Wang, M. Zhang, Y.Q. Lu, J. Mol. Liq. 62–66, 151 (2010)
A. Henni, P. Tontiwachwuthikul, A. Chakma, E.A. Mather, J. Chem. Eng. Data 101–107, 44 (1999)
O. Kiyohara, G.C. Benson, J. Chem. Thermodyn. 861–873, 11 (1979)
G. Douheret, M.I. Davis, J. Loya, Thermochim. Acta 313–328, 207 (1992)
G. Douheret, A. Viallard, J. Phys. Chem. 809–818, 80 (1983)
G. Douheret, M.B. Holczer, R. Peryrelier, M.I. Davis, J. Chem. Eng. Data 868–872, 39 (1994)
G. Douheret, A. Pal, M.I. Davis, J. Chem. Thermodyn. 99–108, 22 (1990)
S. Glasstone, K.J. Laidler, H. Eyring, The Theory of Rate Processes (McGraw-Hill Book Company, New York, 1941)
L. Grunberg, A.H. Nissan, Nature 799–800, 164 (1949)
M. Tamura, M. Kurata, Bull. Chem. Soc. Jpn. 32–38, 25 (1952)
P.K. Katti, M.M. Chaudhri, J. Chem. Eng. Data 442–443, 9 (1964)
R.K. Hind, E. McLaughlin, A.R. Ubbelohde, Trans. Faraday Soc. 328–330, 56 (1960)
R.A. McAllister, AIChE J. 427–431, 6 (1960)
E.L. Heric, J. Chem. Eng. Data 66–68, 11 (1966)
T.M. Aminabhavi, M.I. Aralaguppi, S.B. Horogopped, R.H. Balundgi, J. Chem. Eng. Data 31–39, 38 (1993)
G.P. Dubey, P. Kaur, Fluid Phase Equilib. 114–126, 354 (2013)
P. Kaur, G.P. Dubey, J. Mol. Liq. 479–486, 233 (2017)
G.P. Dubey, P. Kaur, J. Mol. Liq. 10–20, 201 (2015)
J. George, N.V. Sastry, S.R. Patel, M.K. Valand, J. Chem. Eng. Data 262–269, 47 (2002)
R.J. Fort, W.R. Moore, Trans. Faraday Soc. 2102–2114, 61 (1965)
P.J. Flory, J. Am. Chem. Soc. 1833–1838, 87 (1965)
A. Abe, P.J. Flory, J. Am. Chem. Soc. 1838–1846, 87 (1965)
S. Oswal, A. Patel, J. Chem. Eng. Data 366–371, 39 (1994)
M. Zafarani-Moattar, H. Shekaari, J. Chem. Thermodyn. 1377–1384, 38 (2006)
A. Kumar, T. Singh, R.L. Gardas, J.A.P. Coutinho, J. Chem. Thermodyn. 32–39, 40 (2008)
Acknowledgements
One of the authors (Rachana Singh) gratefully acknowledges the Council of Scientific & Industrial Research (CSIR), New Delhi for financial support in the form of Junior Research Fellowship via file No. 09/105(0306)/2020-EMR-I dated 30/5/2020.
Funding
This work was supported by the Council of Scientific & Industrial Research (CSIR), New Delhi [09/105(0306)/2020-EMR-I dated 30/5/2020]. My self Rachana. I am CSIR fellow with file No. 09/105(0306)/2020-EMR-I dated 30/5/2020. My fellowship has been stuck from past three months i.e. from September 2023 to November 2023. On CSIR Fellows portal it is proceeded up to university. It is not further proceeded by EMR DA. Kindly resolve my issue as soon as possible.
Author information
Authors and Affiliations
Contributions
Gyan Prakash Dubey: Conceptualization, validation, investigation, resources, writing—review and editing, Supervision. Rachana Singh: Conceptualization, resources, writing—original draft, visualization, funding acquisition. Aarzoo Ahuja: Conceptualization, resources, visualization.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dubey, G.P., Singh, R. & Ahuja, A. Studies on Intermolecular Interactions in Binary Liquid Mixtures of 2-Methyl-1-butanol with Glycol Ethers {CH3(OCH2CH2)nOH}, n = 1,2,3 at Different Temperatures: A Volumetric, Acoustic and Transport Study. Int J Thermophys 45, 32 (2024). https://doi.org/10.1007/s10765-024-03329-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10765-024-03329-z