Abstract
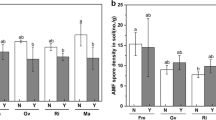
Arbuscular mycorrhizal fungi (AMF) are one of the environment-friendly organisms that enhance plant performance. AMF affect the herbivorous insect community by indirectly modifying host plant nutrient uptake, growth, and defense, also known as priming. In the current study, under greenhouse conditions, the effects of inoculating tomato seedlings with four species of AMF, i.e., Funneliformis mosseae, Rhizophagus intraradices, Rhizophagus irregularis, and Glomus iranicus, were studied in relation to tomato plant growth parameters, plant defense enzymes, and total phenol content, and additionally, the life table of Bemisia tabaci (Gennadius) (Hemiptera: Aleyrodidae) feeding on these plants was determined. The results demonstrated that the growth parameters of tomato plants, including plant height, stem diameter, number of leaves, root volume, leaf surface area, weight of the root, and aerial organs (containing the leaves and stem), were greater and larger in the AMF-inoculated plants compared to the non-inoculated plants. Furthermore, there were higher defense enzyme activities, including peroxidase, phenylalanine ammonia lyase and polyphenol oxidase, and also higher total phenol contents in the AMF-inoculated plants. The whitefly life table characteristics were decreased in the group feeding on the AMF-inoculated plants. All together, the AMF colonization made the tomato plants more resistant against B. tabaci by improving plant growth and increasing defense enzymes. The degree of priming observed here suggests the potential of AMF to have expansive applications, including their implementation in sustainable agriculture.





Similar content being viewed by others
Data Availability
All data underlying the results are available as part of the article and no additional source data are required.
References
Agrawal AA, Fishbein M (2006) Plant defense syndromes. J Ecol 87(sp7):S132–S149. https://doi.org/10.1890/0012-9658(2006)87[132:PDS]2.0.CO;2
Alagar M, Suresh S, Saravanakumar D, Samiyappan R (2010) Feeding-induced changes in defence enzymes and PR proteins and their implications in host resistance to Nilaparvata lugens. J Appl Entomol 134(2):123–131. https://doi.org/10.1111/j.1439-0418.2009.01461.x
Ameye M, Allmann S, Verwaeren J, Smagghe G, Haesaert G, Schuurink RC, Audenaert K (2018) Green leaf volatile production by plants: a meta-analysis. New Phytol 220(3):666–683
An C, Sheng L, Du X, Wang Y, Zhang Y, Song A et al (2019) Overexpression of CmMYB15 provides chrysanthemum resistance to aphids by regulating the biosynthesis of lignin. J Hortic Res 6:84. https://doi.org/10.1038/s41438-019-0166-y
Avio L, Sbrana C, Giovannetti M, Frassinetti S (2017) Arbuscular mycorrhizal fungi affect total phenolics content and antioxidant activity in leaves of oak leaf lettuce varieties. Sci Hortic 224:265–271. https://doi.org/10.1016/j.scienta.2017.06.022
Bennett AE, Bever JD, Bowers MD (2009) Arbuscular mycorrhizal fungal species suppress inducible plant responses and alter defensive strategies following herbivory. Oecologia 160:771–779. https://doi.org/10.1007/s00442-009-1338-5
Bernaola L, Stout MJ (2020) The effect of mycorrhizal seed treatments on rice growth, yield, and tolerance to insect herbivores. J Pestic Sci 94(2):375–392
Bever J (2002) Negative feedbacks within a mutualism: host-specific growth of mycorrhizal fungi reduces plant benefit. Proc R Soc 269:2595–2601. https://doi.org/10.1098/rspb.2002.2162
Bezemer TM, Van Dam NM (2005) Linking aboveground and belowground interactions via induced plant defenses. Trends Ecol Evol 20:617–624. https://doi.org/10.1016/j.tree.2005.08.006
Borowicz VA (1997) A fungal root symbiont modifies plant resistance to an insect herbivore. Oecologia 112:534–542. https://doi.org/10.1007/s004420050342
Bowers MD, Puttick GM (1988) Response of generalist and specialist insects to qualitative allelochemical variation. J Chem Ecol 14:319–334. https://doi.org/10.1007/BF01022549
Byrne DN, Bellows TS Jr (1991) Whitefly biology. Annu Rev Entomol 36:431–457
Caparros Megido R, Brostaux Y, Haubruge E, Verheggen FJ (2013) Propensity of the tomato leafminer, Tuta absoluta (Lepidoptera: Gelechiidae), to develop on four potato plant varieties. Am J Potato Res 90:255–260. https://doi.org/10.1007/s12230-013-9300-9
Chen F (2015) Colonization by arbuscular mycorrhizal and endophytic fungi enhanced terpene production in tomato plants and their defense against a herbivorous insect. Symbiosis 65:65–74
Chen L, Li X, He T, Li P, Liu Y, Zhou S, Wu Q, Chen T, Lu Y, Hou Y (2021) Comparative biochemical and transcriptome analyses in tomato and eggplant reveal their differential responses to Tuta absoluta infestation. Genomics 113(4):2108–2121. https://doi.org/10.1016/j.ygeno.2021.05.002
Chen X, Zhang YX, Zhang YP, Wei H, Lin JZ, Sun L, Chen F (2017) Relative fitness of avermectin-resistant strain of Neoseiulus cucumeris (Oudemans) (Acari: Phytoseiidae). Syst Appl Acarol 22(2):184–193. https://doi.org/10.11158/saa.22.2.3
Chi H, Liu H (1985) Two new methods for the study of insect population ecology. Bull Inst Math Acad Sin 24:225–240
Chi H (1988) Life-table analysis incorporating both sexes and variable development rates among individuals. Environ Entomol 17:26–34. https://doi.org/10.1093/ee/17.1.26
Chi H, You M, Atlıhan R, Smith CL, Kavousi A, Özgökçe MS, Güncan A, Tuan S-J, Fu J-W, Xu Y-Y, Zheng F-Q, Ye B-H, Chu D, Yu Y, Gharekhani G, Saska P, Gotoh T, Schneider MI, Bussaman P, Gökçe A, Liu T-X (2020) Age-Stage, two-sex life table: an introduction to theory, data analysis, and application. Entomol Gen 40:103–124. https://doi.org/10.1127/entomologia/2020/0936
Commare RR, Nandakumar R, Kandan A, Suresh S, Bharathi M, Raguchander T, Samiyappan R (2002) Pseudomonas fluorescens based bio-formulation for the management of sheath blight disease and leaf folder insect in rice. J Crop Prot 21:671–677. https://doi.org/10.1016/S0261-2194(02)00020-0
Cozzolino V, Di Meo V, Piccolo A (2013) Impact of arbuscular mycorrhizal fungi applications on maize production and soil phosphorus availability. J Geochem Explor 129:40–44. https://doi.org/10.1016/j.gexplo.2013.02.006
Currie AF, Murray PJ, Gange AC (2011) Is a specialist root-feeding insect affected by arbuscular mycorrhizal fungi? Appl Soil Ecol 47:77–83. https://doi.org/10.1016/j.apsoil.2010.12.002
Czerniewicz P, Sytykiewicz H, Durak R, Borowiak-Sobkowiak B, Chrzanowski G (2017) Role of phenolic compounds during antioxidative responses of winter triticale to aphid and beetle attack. Plant Physiol Biochem 118:529–540. https://doi.org/10.1016/j.plaphy.2017.07.024
Falco B, Manzo D, Incerti G, Garonna AP, Ercolano M, Lanzotti V (2019) Metabolomics approach based on NMR spectroscopy and multivariate data analysis to explore the interaction between the leafminer Tuta absoluta and tomato (Solanum lycopersicum). Phytochem Anal 30(5):556–563. https://doi.org/10.1002/pca.2850
Dicke M (2005) Insect-plant biology. Oxford University Press on Demand, pp 446
Dreher D, Baldermann S, Schreiner M, Hause B (2019) An arbuscular mycorrhizal fungus and a root pathogen induce different volatiles emitted by Medicago truncatula roots. J Adv Res 19:85–90. https://doi.org/10.1016/0048-4059(84)90050-X
Duan C, Yu J, Bai J, Zhu Z, Wang X (2014) Induced defense responses in rice plants against small brown planthopper infestation. Crop J 2:55–62
Eichholtzer J, Ballina-Gómez HS, Gómez-Tec K, Medina-Dzul K (2021) Arbuscular mycorrhizal fungi influence whitefly abundance by modifying habanero pepper tolerance to herbivory. Arthropod Plant Interact 15(6):861–874. https://doi.org/10.1007/s11829-021-09868-8
Elsen A, Gervacio D, Swennen R, De Waele D (2008) AMF-induced biocontrol against plant parasitic nematodes in Musa sp.: a systemic effect. Mycorrhiza 18(5):251–256. https://doi.org/10.1007/s00572-008-0173-6
Formenti L, Rasmann S (2019) Mycorrhizal fungi enhance resistance to herbivores in tomato plants with reduced jasmonic acid production. J Agron 9(3):131. https://doi.org/10.3390/agronomy9030131
Gange AC, West HM (1994) Interactions between arbuscular mycorrhizal fungi and foliar-feeding insects in Plantago lanceolata L. New Phytol 128(1):79–87
Gange AC (2007) Insect-mycorrhizal interactions: patterns, processes and consequences. In: Ohgushi T, Craig T, Price PW (eds) Indirect interaction webs: nontrophic linkages through induced plant traits. Cambridge University Press, Cambridge, pp 124–144
Gilbert L, Johnson D (2015) Plant-mediated ‘apparent effects’ between mycorrhiza and insect herbivores. Curr Opin Plant Biol 26:100–105
Giovannetti M, Mosse B (1980) Evaluation of techniques for measuring vesicular arbuscular mycorrhizal infection in roots. New Phytol 84:489–500
Hao Z, Xie W, Chen B (2019) Arbuscular mycorrhizal symbiosis affects plant immunity to viral infection and accumulation. Viruses 11(6):534. https://doi.org/10.3390/v11060534
Harley JL, Smith SE (2008) Mycorrhizal symbiosis, 3rd edn. Academic Press, Massachusetts
Hartley R, Harris P, Russell G (1978) Degradability and phenolic components of cell walls of wheat in relation to susceptibility to Puccinea striiformis. Ann Appl Biol 88:153–158
Hildebrand DF, Rodriguez JG, Brown GC, Luu KT, Volden CS (1986) Peroxidative responses of leaves in two soybean genotypes injured by two spotted spider mites (Acari: Tetranychidae). J Econ Entomol 79(6):1459–1465
Hori K, Wada A, Shibuta T (1997) Change in phenoloxidase activities of the gals on leaves of Ulmus davidana formed by Tetraneura fusiformis (Homoptera: Eriosomatidae). J Appl Entomol 32:365–371
House GL, Ekanayake S, Ruan Y, Schütte UM, Kaonongbua W, Fox G et al (2016) Phylogenetically structured differences in rRNA gene sequence variation among species of arbuscular mycorrhizal fungi and their implications for sequence clustering. Appl Environ Microbiol 82(16):4921–4930. https://doi.org/10.1128/AEM.00816-16
Jung SC, Martinez-Medina A, Lopez-Raez JA, Pozo MJ (2012) Mycorrhiza-induced resistance and priming of plant defenses. J Chem Ecol 38:651–664
Klironomos JN, McCune J, Moutoglis P (2004) Species of arbuscular mycorrhizal fungi affect mycorrhizal responses to simulated herbivory. Appl Soil Ecol 26(2):133–141
Kohler W, Schachtel W, Voleske P (2002) Biostatistik. Springer-Verlag, Berlin, p 301
Krainacker DA, Carey JR, Vargas RI (1987) Effect of larval host on life history traits on the Mediterranean fruit fly Ceratitis capitata. Oecologia 73:583–590
Krüger M, Krüger C, Walker C, Stockinger H, Schüßler A (2012) Phylogenetic reference data for systematics and phylotaxonomy of arbuscular mycorrhizal fungi from phylum to species level. New Phytol 193(4):970–984. https://doi.org/10.1111/j.1469-8137.2011.03962.x
Kormanik PP, McGraw AC (1982) Quantification of vesicular-arbuscular mycorrhizae in plant roots. In Methods and Principles of Mycorrhizal Research; Schenck, N.C., Ed.; The American Phytopathological Society: St. Paul, MN, USA, pp 37–45
Kumari A, Goyal M, Mittal A, Kumar R (2021) Defensive capabilities of contrasting sorghum genotypes against Atherigona soccata (Rondani) infestation. Protoplasma 1–14. https://doi.org/10.1007/s00709-021-01703-4
Lee EH, Eo JK, Ka KH, Eom AH (2013) Diversity of arbuscular mycorrhizal fungi and their roles in ecosystems. Mycobiology 41(3):121–125
Lokhandwala A, Hoeksema JD (2019) Priming by arbuscular mycorrhizal fungi of plant antioxidant enzyme production: a meta-analysis. Annu Rev Plant Biol 4:1069–1084. https://doi.org/10.1002/9781119312994.apr0680
MacDonald MJ, D’Cunha GB (2007) A modern view of phenylalanine ammonia lyase. Biochem Cell Biol 85(3):273–282. https://doi.org/10.1139/O07-018
Malik RJ, Ali JG, Bever JD (2018) Mycorrhizal composition influences plant anatomical defense and impacts herbivore growth and survival in a life-stage dependent manner. Pedobiologia 66:29–35
Mandal SM, Chakraborty D, Dey S (2010) Phenolic acids act as signaling molecules in plant-microbe symbioses. Plant Signal Behav 5(4):359–368. https://doi.org/10.4161/psb.5.4.10871
Marschner H, Dell B (1994) Nutrient uptake in mycorrhizal symbiosis. Plant Soil 159:89–102
Mayer AM (2006) Polyphenol oxidases in plants and fungi: going places? A Review. Phytochem 67(21):2318–2331. https://doi.org/10.1016/j.phytochem.2006.08.006
Minton MM, Barber NA, Gordon LL (2016) Effects of arbuscular mycorrhizal fungi on herbivory defense in two Solanum (Solanaceae) species. Plant Ecol Evol 149(2):157–164. https://doi.org/10.5091/plecevo.2016.1176
Narula N, Kothe E, Behl RK (2009) Role of root exudates in plant–microbe interactions. J Appl Bot Food Qual 82:122–130
Naskar S, Roy C, Ghosh S, Mukhopadhyay A, Hazarika LK, Chaudhuri RK et al (2021) Elicitation of biomolecules as host defense arsenals during insect attacks on tea plants (Camellia sinensis (L.) Kuntze). Appl Microbiol Biotechnol 1–13. https://doi.org/10.1007/s00253-021-11560-z
Oliveira MRV, Henneberry TJ, Anderson P (2001) History, current status, and collaborative research projects for Bemisia tabaci. J Crop Prot 20:709–723. https://doi.org/10.1016/S0261-2194(01)00108-9
Phillips JM, Hayman DS (1970) Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans Br Mycol Soc 55(1):158–161
Pieterse CM, Zamioudis C, Berendsen RL, Weller DM, Van Wees SC, Bakker PA (2014) Induced systemic resistance by beneficial microbes. Annu Rev Phytopathol 52:347–375. https://doi.org/10.1146/annurev-phyto-082712-102340
Pineda A, Zheng SJ, Van Loon JJA, Pieterse CMJ, Dicke M (2010) Helping plants to deal with insects: the role of beneficial soilborne microbes. Trends Plant Sci 15:507–514. https://doi.org/10.1016/j.tplants.2010.05.007
Rai M, Acharya D, Singh A, Varma A (2001) Positive growth responses of the medicinal plants Spilanthes calva and Withania somnifera to inoculation by Piriformospora indica in a field trial. Mycorrhiza 11(3):123–128. https://doi.org/10.1007/s005720100115
Rasheed VA, Rao SK, Babu TR, Murali T, Krishna BV, Naidu GM (2018) Biochemical constituents of different tomato genotypes responsible for resistance/susceptibility to South American tomato leaf miner, Tuta absoluta (Meyrick). J Pharmacogn Phytother 7(6):1122–1129
Razmjou J, Naseri B, Hemati SA (2014) Comparative performance of the cotton bollworm, Helicoverpa armigera (Hübner) (Lepidoptera: Noctuidae) on various host plants. J Pestic Sci 87:29–37. https://doi.org/10.1007/s10340-013-0515-9
Rivero J, Lidoy J, Llopis-Giménez Á, Herrero S, Flors V, Pozo MJ (2021) Mycorrhizal symbiosis primes the accumulation of antiherbivore compounds and enhances herbivore mortality in tomato. J Exp Bot 72(13):5038–5050. https://doi.org/10.1093/jxb/erab171
Rousseau JVD, Reid CPP (1991) Effects of phosphorus fertilization and mycorrhizal development on phosphorus nutrition and carbon balance of loblolly pine. New Phytol 117:319–326. https://doi.org/10.1111/j.1469-8137.1991.tb04913.x
Sbrana C, Avio L, Giovannetti M (2014) Beneficial mycorrhizal symbionts affecting the production of health-promoting phytochemicals. Electrophor 35(11):1535–1546. https://doi.org/10.1002/elps.201300568
Scheublin TR, Ridgway KP, Young JPW, van der Heijden MGA (2004) Nonlegumes, legumes, and root nodules harbor different arbuscular mycorrhizal fungal communities. Appl Environ Microbiol Appl Environ Microb 70:6240–6246. https://doi.org/10.1128/AEM.70.10.6240-6246.2004
Schoenherr AP, Rizzo E, Jackson N, Manosalva P, Gomez SK (2019) Mycorrhiza-induced resistance in potato involves priming of defense responses against cabbage looper (Noctuidae: Lepidoptera). Environ Entomol 48(2):370–381. https://doi.org/10.1093/ee/nvy195
Selvaraj A, Thangavel K, Uthandi S (2020) Arbuscular mycorrhizal fungi (Glomus intraradices) and Diazotrophic bacterium (Rhizobium BMBS) primed defense in blackgram against herbivorous insect (Spodoptera litura) infestation. Microbiol Res 231:126355. https://doi.org/10.1016/j.micres.2019.126355
Shakarami J, Bazgir F (2017) Effect of temperature on life table parameters of Phytoseius plumifer (Phyto-seiidae) fed on Eotetranychus hirsti (Tetranychidae). Syst Appl Acarol 22(3):410–423. https://doi.org/10.11158/saa.22.3.7
Sheng M, Tang M, Chen H, Yang B, Zhang F, Huang Y (2008) Influence of arbuscular mycorrhizae on photosynthesis and water status of maize plants under salt stress. Mycorrhiza 18:287–296. https://doi.org/10.1007/s00572-008-0180-7
Shrivastava G, Ownley BH, Aug´e RM, Toler H, Dee M, Vu A, Köllner TG, Chen F (2015) Colonization by arbuscular mycorrhizal and endophytic fungi enhanced terpene production in tomato plants and their defense against a herbivorous insect. Symbiosis 65 (2):65–74
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic 16(3):144–158
Song YY, Zeng RS, Xu JF, Li J, Shen X, Yihdego WG (2010) Interplant communication of tomato plants through underground common mycorrhizal networks. PLoS ONE 5(10):e13324. https://doi.org/10.1371/journal.pone.0013324
Song YY, Ye M, Li CY, Wang RL, Wei XC, Luo SM, Zeng RS (2013) Priming of anti-herbivore defense in tomato by arbuscular mycorrhizal fungus and involvement of the jasmonate pathway. J Chem Ecol 39(7):1036–1044. https://doi.org/10.1007/s10886-013-0312-1
Song YY, Xia M, Lin YB, Lin XH, Ding CH, Wang J et al (2018) Colonization with arbuscular mycorrhizal fungus Funneliformis mosseae enhanced the responses of tomato plants to mechanical wounding. J Appl Ecol 29(11):3811–3818. https://doi.org/10.13287/j.1001-9332.201811.035
Song Y, Wang M, Zeng R, Groten K, Baldwin IT (2019) Priming and filtering of antiherbivore defences among Nicotiana attenuata plants connected by mycorrhizal networks. Plant Cell Environ 42(11):2945–2961. https://doi.org/10.1111/pce.13626
Srinivasan R, Uthamasamy S (2004) Feeding induced changes in phenolic and pathogenesis-related proteins: implications in host resistance to Bemisia tabaci Genn. and Helicoverpa armigera Hub. in tomato accessions. Pest Manag Hortic Ecosyst 10(2):95–106
Van der Heyde M, Bennett JA, Pither J, Hart M (2017) Longterm effects of grazing on arbuscular mycorrhizal fungi. Agric Ecosyst Environ 243:27–33
Vandenborre G, Miersch O, Hause B, Smagghe G, Wasternack C, Van Damme EJM (2009) Spodoptera littoralis-induced lectin expression in tobacco. Plant Cell Physiol 50:1142–1155
Vogt T (2010) Phenyl propanoid biosynthesis. Mol Plant 3:2–20
Wahab A, Muhammad M, Munir A, Abdi G, Zaman W, Ayaz A, ... & Reddy SPP (2023) Role of arbuscular mycorrhizal fungi in regulating growth, enhancing productivity, and potentially influencing ecosystems under abiotic and biotic stresses. Plants 12(17): 3102. https://doi.org/10.3390/plants12173102
Wahing I, Van W, Houba VJG, Van der lee JJ (1989) Soil and Plant analysis, a series of syllabi. Part 7, Plant analysis procedure. Wageningen Agriculture University
Wang J, Xue Y (1980) Studies on plant phenylalanine ammonia lyase. Acta Physiol Plant 7:374–380. https://doi.org/10.1007/s10886-009-9589-5
Wang Y, Cai QN, Qing-Wen Z, Han Y (2006) Effect of the secondary substances from wheat on the growth and digestive physiology of cotton bollworm Helicoverpa armigera (Lepidoptera: Noctuidae). Eur J Entomol 103(1):255
Weyens N, Van der Lelie D, Taghavi S, Newman L, Vangronsveld J (2009) Exploiting plant–microbe partnerships to improve biomass production and remediation. Trends Biotechnol 27(10):591–598. https://doi.org/10.1016/j.tibtech.2009.07.006
Xiao Y, Liu M, Chen Lu, Ji L, Zhao Z, Wang L, Wei L, Zhang Y (2020) Growth and elemental uptake of Trifolium repens in response to biochar addition, arbuscular mycorrhizal fungi and phosphorus fertilizer applications in low-Cd-polluted soils. Environ Pollut 260:113761
Xue Y (1980) Studies on plant phenylalanine ammonia lyase. Acta Physiol Plant 7:374–380
Yang Y, Liu Q, Han C, Qiao YZ, Yao XQ, Yin HJ (2007) Influence of water stress and low irradiance on morphological and physiological characteristics of Picea asperata seedlings. Photosynthetica 45:613–619. https://doi.org/10.1007/s11099007-0106-1
Yu L, Zhang W, Geng Y, Liu K, Shao X (2022) Cooperation with arbuscular mycorrhizal fungi increases plant nutrient uptake and improves defenses against insects. Front Ecol Evol 157. https://doi.org/10.3389/fevo.2022.8333
Zubek S, Mielcarek S, Turnau (2012) Hypericin and pseudohypericin concentrations of a valuable medicinal plant Hypericum perforatum L. are enhanced by arbuscular mycorrhizal fungi. Mycorrhiza 22(2):149–156. https://doi.org/10.1007/s00572-011-0391-1
Author information
Authors and Affiliations
Contributions
F.S. set up the experiments and analyses the data under guidance and recommendations of S. S.N. and wrote the first draft. S.S.N supervised and deliberated the research. S.S.N. and G.S. contributed to subsequent drafting and editing of the manuscript. E.S. provided the fungi and assisted in some part of the experiments. All authors approved the final draft for submission to the journal.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests. All co-authors have seen and agree with the contents of the manuscript. We certify that the submission is original work and is not under review at any other publication.
Additional information
Edited by Jorge Braz Torres
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shafiei, F., Shahidi-Noghabi, S., Sedaghati, E. et al. Arbuscular Mycorrhizal Fungi Inducing Tomato Plant Resistance and Its Role in Control of Bemisia tabaci Under Greenhouse Conditions. Neotrop Entomol 53, 424–438 (2024). https://doi.org/10.1007/s13744-024-01135-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13744-024-01135-8