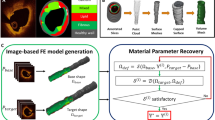
Abstract
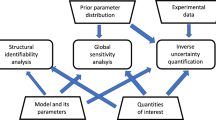
Central to the clinical adoption of patient-specific modeling strategies is demonstrating that simulation results are reliable and safe. Indeed, simulation frameworks must be robust to uncertainty in model input(s), and levels of confidence should accompany results. In this study, we applied a coupled uncertainty quantification–finite element (FE) framework to understand the impact of uncertainty in vascular material properties on variability in predicted stresses. Univariate probability distributions were fit to material parameters derived from layer-specific mechanical behavior testing of human coronary tissue. Parameters were assumed to be probabilistically independent, allowing for efficient parameter ensemble sampling. In an idealized coronary artery geometry, a forward FE model for each parameter ensemble was created to predict tissue stresses under physiologic loading. An emulator was constructed within the UncertainSCI software using polynomial chaos techniques, and statistics and sensitivities were directly computed. Results demonstrated that material parameter uncertainty propagates to variability in predicted stresses across the vessel wall, with the largest dispersions in stress within the adventitial layer. Variability in stress was most sensitive to uncertainties in the anisotropic component of the strain energy function. Moreover, unary and binary interactions within the adventitial layer were the main contributors to stress variance, and the leading factor in stress variability was uncertainty in the stress-like material parameter that describes the contribution of the embedded fibers to the overall artery stiffness. Results from a patient-specific coronary model confirmed many of these findings. Collectively, these data highlight the impact of material property variation on uncertainty in predicted artery stresses and present a pipeline to explore and characterize forward model uncertainty in computational biomechanics.









Similar content being viewed by others
Notes
Pilot studies demonstrated that 2 × oversampling notably reduced fluctuations in the Sobol indices and was thus sufficient to ensure stability in PCE results and conclusions drawn (Supp. Figure 2).
References
Ahmed KBR, Pathmanathan P, Kabadi SV et al (2023) Editorial on the FDA report on “Successes and opportunities in modeling & simulation for FDA.” Ann Biomed Eng 51:6–9. https://doi.org/10.1007/s10439-022-03112-x
Bracamonte JH, Wilson JS, Soares JS (2020) Assessing patient-specific mechanical properties of aortic wall and peri-aortic structures From in vivo DENSE magnetic resonance imaging using an inverse finite element method and elastic foundation boundary conditions. J Biomech Eng 142:121011. https://doi.org/10.1115/1.4047721
Brown AJ, Teng Z, Evans PC et al (2016) Role of biomechanical forces in the natural history of coronary atherosclerosis. Nat Rev Cardiol 13:210–220. https://doi.org/10.1038/nrcardio.2015.203
Burk KM, Narayan A, Orr JA (2020) Efficient sampling for polynomial chaos-based uncertainty quantification and sensitivity analysis using weighted approximate Fekete points. Int J Numer Method Biomed Eng 36:e3395. https://doi.org/10.1002/cnm.3395
Burton AC (1954) Relation of structure to function of the tissues of the wall of blood vessels. Physiol Rev 34:619–642. https://doi.org/10.1152/physrev.1954.34.4.619
Cheng GC, Loree HM, Kamm RD et al (1993) Distribution of circumferential stress in ruptured and stable atherosclerotic lesions. A structural analysis with histopathological correlation. Circulation 87:1179–1187. https://doi.org/10.1161/01.cir.87.4.1179
Chuong CJ, Fung YC (1986) On residual stresses in arteries. J Biomech Eng 108:189–192. https://doi.org/10.1115/1.3138600
Crestaux T, Le Maıˆtre O, Martinez J-M (2009) Polynomial chaos expansion for sensitivity analysis. Reliab Eng Syst Saf 94:1161–1172. https://doi.org/10.1016/j.ress.2008.10.008
Douglas PS, Pontone G, Hlatky MA et al (2015) Clinical outcomes of fractional flow reserve by computed tomographic angiography-guided diagnostic strategies vs. usual care in patients with suspected coronary artery disease: the prospective longitudinal trial of FFR(CT): outcome and resource impacts study. Eur Heart J 36:3359–3367. https://doi.org/10.1093/eurheartj/ehv444
Driessen RS, Danad I, Stuijfzand WJ et al (2019) Comparison of coronary computed tomography angiography, fractional flow reserve, and perfusion imaging for ischemia diagnosis. J Am Coll Cardiol 73:161–173. https://doi.org/10.1016/j.jacc.2018.10.056
Eck VG, Donders WP, Sturdy J et al (2016) A guide to uncertainty quantification and sensitivity analysis for cardiovascular applications. Int J Numer Method Biomed Eng 32:e02755. https://doi.org/10.1002/cnm.2755
Ferruzzi J, Bersi MR, Humphrey JD (2013) Biomechanical phenotyping of central arteries in health and disease: advantages of and methods for murine models. Ann Biomed Eng 41:1311–1330. https://doi.org/10.1007/s10439-013-0799-1
Fleeter CM, Geraci G, Schiavazzi DE et al (2020) Multilevel and multifidelity uncertainty quantification for cardiovascular hemodynamics. Comput Methods Appl Mech Eng 365:113030. https://doi.org/10.1016/j.cma.2020.113030
Food and Drug Administration: Center for Devices and Radiological Health (2023) FDA guidance: assessing the credibility of computational modeling and simulation in medical device submissions. U.S. Food and Drug Administration
García-García H, Mintz G, Lerman A et al (2009) Tissue characterisation using intravascular radiofrequency data analysis: recommendations for acquisition, analysis, interpretation and reporting. EuroIntervention 5:177–189. https://doi.org/10.4244/eijv5i2a29PMID-20449928
Guo L, Narayan A, Yan L, Zhou T (2018) Weighted approximate Fekete points: sampling for least-squares polynomial approximation. SIAM J Sci Comput 40:A366–A387. https://doi.org/10.1137/17M1140960
Holzapfel GA, Gasser TC, Ogden RW (2000) A new constitutive framework for arterial wall mechanics and a comparative study of material models. J Elasticity Phys Sci Solids 61:1–48. https://doi.org/10.1023/a:1010835316564
Holzapfel GA, Sommer G, Gasser CT, Regitnig P (2005) Determination of layer-specific mechanical properties of human coronary arteries with nonatherosclerotic intimal thickening and related constitutive modeling. Amer J Physiol-Heart Circulat Phys 289:H2048–H2058. https://doi.org/10.1152/ajpheart.00934.2004
Humphrey JD (2002) Cardiovascular Solid Mechanics, 1st edn. Springer-Verlag, New York
Humphrey JD (2021) Constrained mixture models of soft tissue growth and remodelling—twenty years After. J Elast 145:49–75. https://doi.org/10.1007/s10659-020-09809-1
Humphrey JD, Schwartz MA (2021) Vascular mechanobiology: homeostasis, adaptation, and disease. Annu Rev Biomed Eng 23:1–27. https://doi.org/10.1146/annurev-bioeng-092419-060810
Jadidi M, Razian SA, Anttila E et al (2021) Comparison of morphometric, structural, mechanical, and physiologic characteristics of human superficial femoral and popliteal arteries. Acta Biomater 121:431–443. https://doi.org/10.1016/j.actbio.2020.11.025
Maas SA, Ellis BJ, Ateshian GA, Weiss JA (2012) FEBio: finite elements for biomechanics. J Biomech Eng 134:011005. https://doi.org/10.1115/1.4005694PMID-22482660
Maas SA, Ellis BJ, Rawlins DS, Weiss JA (2016) Finite element simulation of articular contact mechanics with quadratic tetrahedral elements. J Biomech 49:659–667. https://doi.org/10.1016/j.jbiomech.2016.01.024PMID-26900037
Maas SA, Ateshian GA, Weiss JA (2017) FEBio: history and advances. Annu Rev Biomed Eng 19:279–299. https://doi.org/10.1146/annurev-bioeng-071516-044738PMID-28633565
Moerman KM (2018) GIBBON: the geometry and image-based bioengineering add-on. J Open Source Softw 3:506. https://doi.org/10.21105/joss.00506
Morrison TM, Dreher ML, Nagaraja S et al (2017) The role of computational modeling and simulation in the total product life cycle of peripheral vascular devices. J Med Device 11:024503. https://doi.org/10.1115/1.4035866
Morrison TM, Pathmanathan P, Adwan M, Margerrison E (2018) Advancing regulatory science with computational modeling for medical devices at the FDA’s office of science and engineering laboratories. Front Med 5:241. https://doi.org/10.3389/fmed.2018.00241
Najm HN (2009) Uncertainty quantification and polynomial chaos techniques in computational fluid dynamics. Annu Rev Fluid Mech 41:35–52. https://doi.org/10.1146/annurev.fluid.010908.165248
Narayan A, Liu Z, Bergquist JA et al (2022) UncertainSCI: uncertainty quantification for computational models in biomedicine and bioengineering. Comput Biol Med 152:106407. https://doi.org/10.1016/j.compbiomed.2022.106407
Pathmanathan P, Gray RA, Romero VJ, Morrison TM (2017) Applicability analysis of validation evidence for biomedical computational models. J Verif Valid Uncert 2:021005. https://doi.org/10.1115/1.4037671
Polzer S, Gasser TC, Vlachovský R et al (2020) Biomechanical indices are more sensitive than diameter in predicting rupture of asymptomatic abdominal aortic aneurysms. J Vasc Surg 71:617-626.e6. https://doi.org/10.1016/j.jvs.2019.03.051
Richardson PD, Davies MJ, Born GVR (1989) Influence of plaque configuration and stress distribution on fissuring of coronary atherosclerotic plaques. Lancet 334:941–944. https://doi.org/10.1016/s0140-6736(89)90953-7PMID-2571862
Robertson D, Cook D (2014) Unrealistic statistics: how average constitutive coefficients can produce non-physical results. J Mech Behav Biomed Mater 40:234–239. https://doi.org/10.1016/j.jmbbm.2014.09.006
Samady H, Eshtehardi P, McDaniel MC et al (2011) Coronary artery wall shear stress is associated with progression and transformation of atherosclerotic plaque and arterial remodeling in patients with coronary artery disease. Circulation 124:779–788. https://doi.org/10.1161/circulationaha.111.021824
Sankaran S, Marsden AL (2011) A stochastic collocation method for uncertainty quantification and propagation in cardiovascular simulations. J Biomech Eng 133:031001. https://doi.org/10.1115/1.4003259
Si H (2015) TetGen, a delaunay-based quality tetrahedral mesh generator. ACM Trans Math Softw 41:1–36. https://doi.org/10.1145/2629697
Sobol′ IM (2001) Global sensitivity indices for nonlinear mathematical models and their Monte Carlo estimates. Math Comput Simul 55:271–280. https://doi.org/10.1016/S0378-4754(00)00270-6
Taylor CA, Figueroa CA (2009) Patient-specific modeling of cardiovascular mechanics. Annu Rev Biomed Eng 11:109–134. https://doi.org/10.1146/annurev.bioeng.10.061807.160521
Taylor CA, Humphrey JD (2009) Open problems in computational vascular biomechanics: hemodynamics and arterial wall mechanics. Comput Methods Appl Mech Eng 198:3514–3523. https://doi.org/10.1016/j.cma.2009.02.004
Teng Z, Tang D, Zheng J et al (2009) An experimental study on the ultimate strength of the adventitia and media of human atherosclerotic carotid arteries in circumferential and axial directions. J Biomech 42:2535–2539. https://doi.org/10.1016/j.jbiomech.2009.07.009
Teng Z, Brown AJ, Calvert PA et al (2014) Coronary plaque structural stress is associated with plaque composition and subtype and higher in acute coronary syndrome. Circ Cardiovasc Imaging 7:461–470. https://doi.org/10.1161/circimaging.113.001526
Timmins LH, Miller MW, Clubb FJ Jr, Moore JE Jr (2011) Increased artery wall stress post-stenting leads to greater intimal thickening. Lab Invest 91:955–967. https://doi.org/10.1038/labinvest.2011.57
Timmins LH, Molony DS, Eshtehardi P et al (2015) Focal association between wall shear stress and clinical coronary artery disease progression. Ann Biomed Eng 43:94–106. https://doi.org/10.1007/s10439-014-1155-9PMID-25316593
Tran JS, Schiavazzi DE, Kahn AM, Marsden AL (2019) Uncertainty quantification of simulated biomechanical stimuli in coronary artery bypass grafts. Comput Methods Appl Mech Eng 345:402–428. https://doi.org/10.1016/j.cma.2018.10.024
Trusty PM, Wei ZA, Slesnick TC et al (2019) The first cohort of prospective Fontan surgical planning patients with follow-up data: how accurate is surgical planning? J Thorac Cardiovasc Surg 157:1146–1155. https://doi.org/10.1016/j.jtcvs.2018.11.102
Tsao CW, Aday AW, Almarzooq ZI et al (2022) Heart disease and stroke statistics-2022 update: a report from the american heart association. Circulation 145:e153–e639. https://doi.org/10.1161/CIR.0000000000001052
Vande Geest JP, Sacks MS, Vorp DA (2004) Age dependency of the biaxial biomechanical behavior of human abdominal aorta. J Biomech Eng 126:815–822. https://doi.org/10.1115/1.1824121
Wahle A, Prause GPM, DeJong SC, Sonka M (1999) Geometrically correct 3-D reconstruction of intravascular ultrasound images by fusion with biplane angiography-methods and validation. IEEE Trans Med Imaging 18:686–699. https://doi.org/10.1109/42.796282PMID-10534051
Waller BF, Orr CM, Slack JD et al (1992) Anatomy, histology, and pathology of coronary arteries: a review relevant to new interventional and imaging techniques–Part I. Clin Cardiol 15:451–457. https://doi.org/10.1002/clc.4960150613
Walsh MT, Cunnane EM, Mulvihill JJ et al (2014) Uniaxial tensile testing approaches for characterisation of atherosclerotic plaques. J Biomech 47:793–804. https://doi.org/10.1016/j.jbiomech.2014.01.017PMID-24508324
Xiu D, Hesthaven JS (2005) High-order collocation methods for differential equations with random inputs. SIAM J Sci Comput 27:1118–1139. https://doi.org/10.1137/040615201
Acknowledgements
The authors thank Prof. Jeff Weiss and Dr. Steve Maas for their insightful discussions and assistance with the FEBio Software Suite, and Drs. Habib Samady and David Molony for access to the clinical data.
Funding
This work was supported, in part, by the National Institutes of Health grants R01 HL-150608 (L.H.T.), U24 EB-029012 (R.S.M., A.N.), P41 GM-103545 (R.S.M.), R24 GM-136986 (R.S.M.), American Heart Association grant 23PRE1019455 (C.C.B.), National Science Foundation GRFP (L.C.R.), and the Nora Eccles Treadwell Foundation for Cardiovascular Research (R.S.M.).
Author information
Authors and Affiliations
Contributions
Conceptualization was performed by R.S.M., A.N., L.H.T; methodology by all authors; formal analysis and investigation by all authors; writing—original draft preparation by C.C.B., D.J., Y.F.J.W., L.H.T; writing—review and editing by all authors; funding acquisition by R.S.M., A.N., L.H.T.
Corresponding author
Ethics declarations
Conflict of interests
The authors have no competing financial or non-financial interests to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Berggren, C.C., Jiang, D., Jack Wang, Y.F. et al. Influence of material parameter variability on the predicted coronary artery biomechanical environment via uncertainty quantification. Biomech Model Mechanobiol (2024). https://doi.org/10.1007/s10237-023-01814-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10237-023-01814-2