Abstract
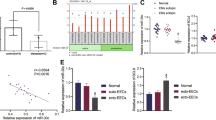
Researchers have reported that miR-124-3p is highly expressed in patients with chronic endometritis. However, the underlying mechanism of miR-124-3p in the development of endometritis remains unclear. This study constructed an in vitro endometrial cell injury model by treating HEECs with 2 μg/mL LPS for 48 h. Then, 1 mg/kg LPS was injected into both sides of the mouse uterus to construct an in vivo endometrial injury model. The expression of miR-124-3p in human endometrial epithelial cells (HEECs) was assessed using RT‒qPCR. Exosomes were separated from bone marrow-derived mesenchymal stem cells (BMSCs) and cocultured with HEECs. A dual-luciferase reporter assay was performed to confirm the relationship between miR-124-3p and DUSP6. The results indicated that LPS inhibited HEEC viability in a time- and dose-dependent manner. The miR-124-3p inhibitor reversed the LPS-induced apoptosis and inhibition of HEEC viability. In addition, miR-124-3p could be transferred from BMSCs to HEECs by exosomes. Exosomes were derived from BMSCs treated with an NC inhibitor (BMSCs/NC Exo) or miR-124-3p inhibitor (BMSCs/anti-miR-124-3p Exo). In addition, BMSCs/anti-miR-124-3p Exo abolished the LPS-induced inhibition of HEEC viability and proliferation by inducing HEEC apoptosis. Moreover, BMSCs/anti-miR-124-3p Exo alleviated the LPS-induced inflammation of HEECs by upregulating DUSP6 and downregulating p-p65 and p-ERK. Furthermore, in an LPS-induced in vivo endometrial injury model, BMSCs/anti-miR-124-3p Exo increased the expression level of DUSP6 and decreased the expression levels of p-p65 and p-ERK. BMSCs/anti-miR-124-3p Exo protected against LPS-induced endometrial damage in vitro and in vivo by upregulating DUSP6 and downregulating p-p65 and p-ERK1/2. This study showed that BMSCs/anti-miR-124-3p Exo might be a potential alternative for the treatment of endometritis.






Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Baker N, Boyette LB, Tuan RS (2015) Characterization of bone marrow-derived mesenchymal stem cells in aging. Bone 70:37–47. https://doi.org/10.1016/j.bone.2014.10.014
Caplan AI (2017) Mesenchymal Stem Cells: Time to Change the Name! Stem Cells Transl Med 6:1445–1451. https://doi.org/10.1002/sctm.17-0051
Di Pietro C, Caruso S, Battaglia R et al (2018) MiR-27a-3p and miR-124-3p, upregulated in endometrium and serum from women affected by Chronic Endometritis, are new potential molecular markers of endometrial receptivity. Am J Reprod Immunol 80:e12858. https://doi.org/10.1111/aji.12858
Dong Y, Yuan Y, Ma Y et al (2023) Corrigendum: Combined intestinal metabolomics and microbiota analysis for acute endometritis induced by lipopolysaccharide in mice. Front Cell Infect Microbiol 13:1223663. https://doi.org/10.3389/fcimb.2023.1223663
Fan MJ, Liang SM, He PJ et al (2019) Dusp6 inhibits epithelial-mesenchymal transition in endometrial adenocarcinoma via ERK signaling pathway. Radiol Oncol 53:307–315. https://doi.org/10.2478/raon-2019-0034
Gao Y, Li H, Han Q et al (2020) Overexpression of DUSP6 enhances chemotherapy-resistance of ovarian epithelial cancer by regulating the ERK signaling pathway. J Cancer 11:3151–3164. https://doi.org/10.7150/jca.37267
Gay C, Hamdaoui N, Pauly V et al (2021) Impact of antibiotic treatment for chronic endometritis on unexplained recurrent pregnancy loss. J Gynecol Obstet Hum Reprod 50:102034. https://doi.org/10.1016/j.jogoh.2020.102034
Huang Y, He B, Wang L et al (2020) Bone marrow mesenchymal stem cell-derived exosomes promote rotator cuff tendon-bone healing by promoting angiogenesis and regulating M1 macrophages in rats. Stem Cell Res Ther 11:496. https://doi.org/10.1186/s13287-020-02005-x
Jang HY, Myoung SM, Choe JM et al (2017) Effects of Autologous Platelet-Rich Plasma on Regeneration of Damaged Endometrium in Female Rats. Yonsei Med J 58:1195–1203. https://doi.org/10.3349/ymj.2017.58.6.1195
Jiang K, Yang J, Yang C et al (2020) miR-148a suppresses inflammation in lipopolysaccharide-induced endometritis. J Cell Mol Med 24:405–417. https://doi.org/10.1111/jcmm.14744
Kimura F, Takebayashi A, Ishida M et al (2019) Review: Chronic endometritis and its effect on reproduction. J Obstet Gynaecol Res 45:951–960. https://doi.org/10.1111/jog.13937
Kitaya K, Takeuchi T, Mizuta S et al (2018) Endometritis: new time, new concepts. Fertil Steril 110:344–350. https://doi.org/10.1016/j.fertnstert.2018.04.012
Margulies SL, Dhingra I, Flores V et al (2021) The Diagnostic Criteria for Chronic Endometritis: A Survey of Pathologists. Int J Gynecol Pathol 40:556–562. https://doi.org/10.1097/PGP.0000000000000737
McDougall S, Aberdein D, Bates A et al (2020) Prevalence of endometritis diagnosed by vaginal discharge scoring or uterine cytology in dairy cows and herds. J Dairy Sci 103:6511–6521. https://doi.org/10.3168/jds.2019-18048
Morrell JM, Rocha A (2021) A Novel Approach to Minimising Acute Equine Endometritis That May Help to Prevent the Development of the Chronic State. Front Vet Sci 8:799619. https://doi.org/10.3389/fvets.2021.799619
Oladejo AO, Li Y, Wu X et al (2020) MicroRNAome: Potential and Veritable Immunomolecular Therapeutic and Diagnostic Baseline for Lingering Bovine Endometritis. Front Vet Sci 7:614054. https://doi.org/10.3389/fvets.2020.614054
Ravel J, Moreno I, Simon C (2021) Bacterial vaginosis and its association with infertility, endometritis, and pelvic inflammatory disease. Am J Obstet Gynecol 224:251–257. https://doi.org/10.1016/j.ajog.2020.10.019
Rupaimoole R, Slack FJ (2017) MicroRNA therapeutics: towards a new era for the management of cancer and other diseases. Nat Rev Drug Discov 16:203–222. https://doi.org/10.1038/nrd.2016.246
Sharma V, Nikolajeff F, Kumar S (2023) Employing nanoparticle tracking analysis of salivary neuronal exosomes for early detection of neurodegenerative diseases. Transl Neurodegener 12:7. https://doi.org/10.1186/s40035-023-00339-z
Shi Q, Wang D, Ding X et al (2021) Exosome-shuttled miR-7162-3p from human umbilical cord derived mesenchymal stem cells repair endometrial stromal cell injury by restricting APOL6. Arch Biochem Biophys 707:108887. https://doi.org/10.1016/j.abb.2021.108887
Umar T, Yin B, Umer S et al (2021) MicroRNA: Could It Play a Role in Bovine Endometritis? Inflammation 44:1683–1695. https://doi.org/10.1007/s10753-021-01458-3
Wang H, Liu D, Sun Y et al (2021a) Upregulation of DUSP6 impairs infectious bronchitis virus replication by negatively regulating ERK pathway and promoting apoptosis. Vet Res 52:7. https://doi.org/10.1186/s13567-020-00866-x
Wang H, Yan Z, Wu X et al (2021b) Using vaginal discharge score (VDS) grading system to evaluate the effect of clinical endometritis on reproductive performance of dairy cows in China. Anim Reprod 18:e20200228. https://doi.org/10.1590/1984-3143-AR2020-0228
Yao Y, Chen R, Wang G et al (2019) Exosomes derived from mesenchymal stem cells reverse EMT via TGF-beta1/Smad pathway and promote repair of damaged endometrium. Stem Cell Res Ther 10:225. https://doi.org/10.1186/s13287-019-1332-8
Yin N, Yang Y, Wang X et al (2019) MiR-19a mediates the negative regulation of the NF-kappaB pathway in lipopolysaccharide-induced endometritis by targeting TBK1. Inflamm Res 68:231–240. https://doi.org/10.1007/s00011-019-01213-3
Yin B, Umar T, Ma X et al (2021) MiR-193a-3p targets LGR4 to promote the inflammatory response in endometritis. Int Immunopharmacol 98:107718. https://doi.org/10.1016/j.intimp.2021.107718
Zhao G, Li R, Cao Y et al (2020a) ΔNp63α-induced DUSP4/GSK3β/SNAI1 pathway in epithelial cells drives endometrial fibrosis. Cell Death Dis 11:449. https://doi.org/10.1038/s41419-020-2666-y
Zhao R, Wang J, Zhang X et al (2020b) MiR-643 inhibits lipopolysaccharide-induced endometritis progression by targeting TRAF6. Cell Biol Int 44:1059–1067. https://doi.org/10.1002/cbin.11306
Zhao Y, Cai H, Ding X, Zhou X (2023) An integrative analysis of the single-cell transcriptome identifies DUSP4 as an exhaustion-associated gene in tumor-infiltrating CD8+ T cells. Funct Integr Genomics 22;23(2):136. https://doi.org/10.1007/s10142-023-01056-6
Funding
This study was supported by the Natural Science Foundation of Fujian Province (No. 2020J01986, and 2021J01236) and Fujian Provincial Finance Project (NO: BPB-CYH2021).
Author information
Authors and Affiliations
Contributions
Conceived and designed the study: Yihong Chen, Shan Zheng, and Juanbing Wei.
Performed the literature search and data extraction: Xiumei Zhao, Yi Zhang, and Suchai Yu.
Analysed the data: Xiumei Zhao and Yi Zhang. Drafted the manuscript: Yihong Chen, and Juanbing Wei.
Corresponding author
Ethics declarations
Ethical approval and consent to participate
This study was reviewed and approved by the Ethics Committee of Fujian Medical University (ethics number: 2023-Y-0524). No patients were involved in this study.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, Y., Zheng, S., Zhao, X. et al. Unveiling the protective effects of BMSCs/anti-miR-124-3p exosomes on LPS-induced endometrial injury. Funct Integr Genomics 24, 32 (2024). https://doi.org/10.1007/s10142-024-01303-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10142-024-01303-4