Abstract
Purpose
Diffuse gliomas are managed with radiation and temozolomide; however, this therapy often results in hematologic toxicities. Patients undergoing chemoradiation also risk contracting Pneumocystis jirovecii pneumonia (PJP), and frequently receive prophylaxis against PJP during treatment. Independent of chemoradiation, some PJP prophylaxis drugs have the potential to cause myelosuppression, which could require cessation of chemotherapy. Here, we evaluate differences in the frequency of hematologic toxicities during chemoradiation when patients receive PJP prophylaxis.
Methods
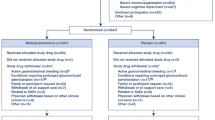
This retrospective chart review evaluated patients with primary brain tumors treated with radiation and concurrent temozolomide. Analyses were performed to assess the effect of the type of PJP prophylaxis on risk for neutropenia, lymphopenia, or thrombocytopenia and the severity of these adverse effects as defined using the Common Terminology Criteria for Adverse Events.
Results
Of the 217 patients included in this analysis, 144 received trimethoprim-sulfamethoxazole (TMP/SMX) and 69 received pentamidine. Of the patients who received TMP/SMX, 15.3% developed an absolute neutrophil count < 1500 cells/µL compared with 7.2% of patients receiving pentamidine (p = 0.10). Platelet count < 100,000/µL occurred in 18.1% of patients who received TMP/SMX and 20.3% of patients who received pentamidine (p = 0.70). No significant differences in lymphocyte counts between therapies were seen. Severity of hematologic toxicities were similar between PJP prophylaxis groups.
Conclusion
These findings suggest that the type of PJP prophylaxis does not significantly affect the risk for hematologic toxicity in brain tumor patients receiving radiation and temozolomide. Additional studies are merited to evaluate the higher rate of neutropenia in patients on TMP/SMX observed in this study.

Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO, National Cancer Institute of Canada Clinical Trials Group (2005) European Organisation for Research and Treatment of Cancer Brain Tumor Radiotherapy Groups, Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352: 987–996 https://doi.org/10.1056/NEJMoa043330
van den Bent MJ, Tesileanu CMS, Wick W, Sanson M, Brandes AA, Clement PM, Erridge S, Vogelbaum MA, Nowak AK, Baurain JF, Mason WP, Wheeler H, Chinot OL, Gill S, Griffin M, Rogers L, Taal W, Ruda R, Weller M, McBain C, Reijneveld J, Enting RH, Caparrotti F, Lesimple T, Clenton S, Gijtenbeek A, Lim E, Herrlinger U, Hau P, Dhermain F, de Heer I, Aldape K, Jenkins RB, Dubbink HJ, Kros JM, Wesseling P, Nuyens S, Golfinopoulos V, Gorlia T, French P, Baumert BG (2021) Adjuvant and concurrent temozolomide for 1p/19q non-co-deleted anaplastic glioma (CATNON; EORTC study 26053– 22054): second interim analysis of a randomised, open-label, phase 3 study. Lancet Oncol 22:813–823. https://doi.org/10.1016/S1470-2045(21)00090-5
Grossman SA, Desideri S, Ye X, Szajna K, Chamberlain M, Lesser G (2007) Iatrogenic immunosuppression in patients with newly diagnosed high-grade gliomas. J Clin Oncol 25:2012–2012. https://doi.org/10.1200/jco.2007.25.18_suppl.2012
Grossman SA, Ye X, Lesser G, Sloan A, Carraway H, Desideri S, Piantadosi S, Consortium NC (2011) Immunosuppression in patients with high-grade gliomas treated with radiation and temozolomide. Clin Cancer Res 17:5473–5480. https://doi.org/10.1158/1078-0432.CCR-11-0774
Mahindra AK, Grossman SA (2003) Pneumocystis carinii pneumonia in HIV negative patients with primary brain tumors. J Neurooncol 63:263–270. https://doi.org/10.1023/a:1024217527650
Merck & Co (2016) Temozolomide [package insert]. Whitehouse Station, NJ
Cohen MH, Johnson JR, Pazdur R (2005) Food and Drug Administration Drug approval summary: temozolomide plus radiation therapy for the treatment of newly diagnosed glioblastoma multiforme. Clin Cancer Res 11:6767–6771. https://doi.org/10.1158/1078-0432.CCR-05-0722
Hirschel B, Lazzarin A, Chopard P, Opravil M, Furrer HJ, Ruttimann S, Vernazza P, Chave JP, Ancarani F, Gabriel V et al (1991) A controlled study of inhaled pentamidine for primary prevention of Pneumocystis carinii pneumonia. N Engl J Med 324:1079–1083. https://doi.org/10.1056/NEJM199104183241602
Schneider MM, Hoepelman AI, Eeftinck Schattenkerk JK, Nielsen TL, van der Graaf Y, Frissen JP, van der Ende IM, Kolsters AF, Borleffs JC (1992) A controlled trial of aerosolized pentamidine or trimethoprim-sulfamethoxazole as primary prophylaxis against Pneumocystis carinii pneumonia in patients with human immunodeficiency virus infection. The Dutch AIDS Treatment Group. N Engl J Med 327:1836–1841. https://doi.org/10.1056/NEJM199212243272603
Gerber DE, Grossman SA, Zeltzman M, Parisi MA, Kleinberg L (2007) The impact of thrombocytopenia from temozolomide and radiation in newly diagnosed adults with high-grade gliomas. Neuro Oncol 9:47–52. https://doi.org/10.1215/15228517-2006-024
Climans SA, Grunfeld E, Mason WP, Chan KKW (2022) Effectiveness and safety of pneumocystis pneumonia prophylaxis for patients receiving temozolomide chemoradiotherapy. Neuro Oncol 24:1738–1748. https://doi.org/10.1093/neuonc/noac072
Singhal N, Selva-Nayagam S, Brown MP (2007) Prolonged and severe myelosuppression in two patients after low-dose temozolomide treatment- case study and review of literature. J Neurooncol 85:229–230. https://doi.org/10.1007/s11060-007-9403-6
Hoffmann-LaRoche (2013) Bactrim™: sulfamethoxazole and trimethoprim DS (double-strength) tablets and tablets USP. https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/017377s074lbl.pdf
Elajez R, Adeli M (2018) Myelosuppression effect of trimethoprim-sulfamethoxazole prophylaxis in innate immune defect patients: retrospective comparative study. J Allergy Clin Immunol 141:AB25. https://doi.org/10.1016/j.jaci.2017.12.078
Lexicomp (n.d.) Pentamidine (oral inhalation): drug information. https://www.uptodate.com/contents/pentamidine-oral-inhalation-drug-information?search=pentamidine&source=panel_search_result&selectedTitle=1~58&usage_type=panel&showDrugLabel=true&display_rank=1#F44486498. Accessed January 13 2024
Walker JG, Kadia T, Brown L, Juneja HS, de Groot JF (2009) Dapsone induced methemoglobinemia in a patient with glioblastoma. J Neurooncol 94:149–152. https://doi.org/10.1007/s11060-009-9813-8
Mitta A, Curtis BR, Reese JA, George JN (2019) Drug-induced thrombocytopenia: 2019 update of clinical and laboratory data. Am J Hematol 94:E76–E78. https://doi.org/10.1002/ajh.25379
Armstrong TS, Cao Y, Scheurer ME, Vera-Bolanos E, Manning R, Okcu MF, Bondy M, Zhou R, Gilbert MR (2009) Risk analysis of severe myelotoxicity with temozolomide: the effects of clinical and genetic factors. Neuro Oncol 11:825–832. https://doi.org/10.1215/15228517-2008-120
Henson JW, Jalaj JK, Walker RW, Stover DE, Fels AO (1991) Pneumocystis carinii pneumonia in patients with primary brain tumors. Arch Neurol 48:406–409. https://doi.org/10.1001/archneur.1991.00530160074017
Neuwelt AJ, Nguyen TM, Fu R, Bubalo J, Tyson RM, Lacy C, Gahramanov S, Nasseri M, Barnes PD, Neuwelt EA (2014) Incidence of Pneumocystis Jirovecii pneumonia after temozolomide for CNS malignancies without prophylaxis. CNS Oncol 3:267–273. https://doi.org/10.2217/cns.14.24
Rifino N, Rigamonti A, Guida FM, De Nobili G, Spena G, Ferrarese C, Salmaggi A (2019) Lack of development of Pneumocystis Jirovecii Pneumonia in a cohort of 103 Italian glioblastoma patients not receiving prophylaxis during post-surgical chemoradiotherapy. J Neurol Sci 405:116431. https://doi.org/10.1016/j.jns.2019.116431
Skorupan N, Ranjan S, Mehta S, Yankulina O, Nenortas N, Grossman S, Ye X, Holdhoff M (2019) Pneumocystis jirovecii prophylaxis in patients treated for high-grade gliomas: a survey among neuro-oncologists. Neurooncol Pract 6:321–326. https://doi.org/10.1093/nop/npy049
Acknowledgements
We thank Kristin Kraus and Cortlynd Olsen for their editorial expertise.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
LMA, HC, and JM conceived and designed the study. LMA wrote the manuscript. LMA and YH performed the chart review. HL conducted all statistical analyses. All authors reviewed the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This project was approved by the Institutional Review Board at the University of Utah.
Consent to participate
The study was performed with a waiver of informed consent from the Institutional Review Board.
Consent to publish
This study was performed with a waiver of informed consent from the Institutional Review Board.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Arnold, L.M., Hoshina, Y., Lee, H. et al. Effect of Pneumocystis jirovecii pneumonia prophylaxis on hematologic toxicity in patients receiving chemoradiation for primary brain tumors. J Neurooncol 167, 211–217 (2024). https://doi.org/10.1007/s11060-024-04588-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-024-04588-4