Abstract
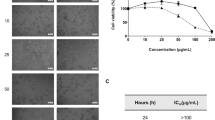
We previously reported that the bismuth(III) dithiocarbamate derivative, bismuth diethyldithiocarbamate (1) exhibited greater cytotoxicity while inducing apoptosis via the intrinsic pathway in MCF-7 cells. We further evaluated the other bismuth(III) dithiocarbamate derivatives, Bi[S2CNR]3, with R = (CH2CH2OH)(iPr), (CH2)4, and (CH2CH2OH)(CH3), denoted as 2, 3, and 4, respectively, in the same MCF-7 cell line. 2–4 were found to exhibit IC50 values of 10.33 ± 0.06 µM, 1.07 ± 0.01 µM and 25.37 ± 0.12 µM, respectively, compared to that of cisplatin at 30.53 ± 0.23 µM. Apoptotic promotion via the mitochondrial-dependent pathway was due to the elevation of intracellular reactive oxygen species (ROS), promotion of caspases, release of cytochrome c, fragmentation of DNA, and results of staining assay observed in all compound-treated cells. 2–4 are also capable of suppressing MCF-7 cell invasion and modulate Lys-48 also Lys-63 linked polyubiquitination, leading to proteasomal degradation. Analysis of gene expression via qRT-PCR revealed their modulation, which supported all activities conducted upon treatment with 2–4. Altogether, bismuth dithiocarbamate derivatives, with bismuth(III) as the metal center bound to ligands, isopropyl ethanol, pyrrolidine, and methyl ethanol dithiocarbamate, are potential anti-breast cancer agents that induce apoptosis and suppress metastasis. Further studies using other breast cancer cell lines and in vivo studies are recommended to clarify the anticancer effects of these compounds.
Graphical abstract










Similar content being viewed by others
Data availability
The data are available from the corresponding author upon reasonable request.
References
Abbas R, Larisch S (2021) Killing by degradation: regulation of apoptosis by the ubiquitin-proteasome-system. Cells 10(12):3465. https://doi.org/10.3390/cells10123465
Adcock AK, Ayscue RL, Breuer LM, Verwiel CP, Marwitz AC, Bertke JA, Vallet V, Réal F, Knope KE (2020) Synthesis and photoluminescence of three bismuth(III)organic compounds bearing heterocyclic n-donor ligands. Dalton Trans 49:11756–11771. https://doi.org/10.1039/D0DT02360D
Adeyemi JO, Onwudiwe DC (2020) The mechanisms of action involving dithiocarbamate complexes in biological systems. Inorg Chim Acta 511:119809. https://doi.org/10.1016/j.ica.2020.119809
Aggarwal V, Tuli HS, Varol A, Thakral F, Yerer MB, Sak K, Varol M, Jain A, Khan MA, Sethi G (2019) Role of reactive oxygen species in cancer progression: molecular mechanisms and recent advancements. Biomolecules 9(11):735. https://doi.org/10.3390/biom9110735
Ajiboye TO, Ajiboye TT, Marzouki R, Onwudiwe DC (2022) The versatility of dithiocarbamates. Int J Mol Sci 23(3):1317. https://doi.org/10.3390/ijms23031317
Akahane M, Akahane T, Shah A, Okajima E, Thorgeirsson UP (2005) A potential role for vascular endothelial growth factor-D as an autocrine growth factor for human breast carcinoma cells. Anticancer Res 25(2A):701–707
Algeciras-Schimnich A, Barnhart BC, Peter ME (2013) Apoptosis-dependent and independent functions of caspases. In: Madame Curie Bioscience Database [Internet]. Landes Bioscience, Austin (TX), 2000–2013. https://www.ncbi.nlm.nih.gov/books/NBK6198/. Accessed 30 June 2023
Al-Janabi ASM, Saleh AM, Hatshan MR (2021) Cytotoxicity, antimicrobial studies of M(II)-dithiocarbamate complexes, and molecular docking study against SARS COV2 RNA-dependent RNA polymerase. J Chin Chem Soc 68(6):1104–1115. https://doi.org/10.1002/jccs.202000504
Andrew FP, Ajibade PA (2018) Metal complexes of alkyl-aryl dithiocarbamates: structural studies, anticancer potentials and applications as precursors for semiconductor nanocrystals. J Mol Str 1155:843–855. https://doi.org/10.1016/j.molstruc.2017.10.106
Bao R, Christova T, Song S, Angers S, Yan X, Attisano L (2012) Inhibition of tankyrases induces Axin stabilization and blocks Wnt signaling in breast cancer cells. PLoS ONE 7(11):e48670. https://doi.org/10.1371/journal.pone.0048670
Baritaki S, Chapman A, Yeung K, Spandidos DA, Palladino M, Bonavida B (2009) Inhibition of epithelial to mesenchymal transition in metastatic prostate cancer cells by the novel proteasome inhibitor, NPI-0052: pivotal roles of Snail repression and RKIP induction. Oncogene 28(40):3573–3585. https://doi.org/10.1038/onc.2009.214
Baskaran Nair HB, Krishnegowda N, Nair B, Ganapathy N, Olga S et al (2010) Novel role of PARP4 in hormone-dependent breast cancer progression and metastasis. Cancer Res. https://doi.org/10.1158/1538-7445.AM10-4607
Biersack B, Schobert R (2019) Current state of platinum complexes for the treatment of advanced and drug-resistant breast cancers. Adv Exp Med Biol 1152:253–270. https://doi.org/10.1007/978-3-030-20301-6_13
Bratton SB, Salvesen GS (2010) Regulation of the Apaf-1-caspase-9 apoptosome. J Cell Sci 123(Pt 19):3209–3214. https://doi.org/10.1242/jcs.073643
Chan PF, Ang KP, Hamid RA (2021) A bismuth diethyldithiocarbamate compound induced apoptosis via mitochondria-dependent pathway and suppressed invasion in MCF-7 breast cancer cells. Biometals 34:365–391. https://doi.org/10.1007/s10534-021-00286-0
Chen X, Yang Q, Xiao L, Tang D, Dou QP, Liu J (2017) Metal-based proteasomal deubiquitinase inhibitors as potential anticancer agents. Cancer Metastasis Rev 36(4):655–668. https://doi.org/10.1007/s10555-017-9701-1
Chen D, Frezza M, Schmitt S, Kanwar J, Dou QP (2011) Bortezomib as the first proteasome inhibitor anticancer drug: current status and future perspectives. Curr Cancer Drug Targets 11(3):239–253. https://doi.org/10.2174/156800911794519752
Cho J, Park J, Kim EE, Song EJ (2020) Assay systems for profiling deubiquitination activity. Int J Mol Sci 21(16):5638. https://doi.org/10.3390/ijms21165638
Comşa Ş, Cîmpean AM, Raica M (2015) The story of MCF-7 breast cancer cell line: 40 years of experience in research. Anticancer Res 35(6):3147–3154
Cvek B, Dvorak Z (2007) Targeting of nuclear factor-kappa B and proteasome by dithiocarbamate complexes with metals. Curr Pharm Des 13(30):3155–3167. https://doi.org/10.2174/138161207782110390
Ebright RY, Zachariah MA, Micalizzi DS, Wittner BS, Niederhoffer KL, Nieman LT, Chirn B, Wiley DF, Wesley B, Shaw B, Nieblas-Bedolla E, Atlas L, Szabolcs A, Iafrate AJ, Toner M, Ting DT, Brastianos PK, Haber DA, Maheswaran S (2020) HIF1A signaling selectively supports proliferation of breast cancer in the brain. Nat Commun 11(1):6311. https://doi.org/10.1038/s41467-020-20144-w
Ejelonu BC, Olagboye SA, Oyeneyin OE, Ebiesuwa OA, Bada OE (2018) Synthesis, characterization and antimicrobial activities of sulfadiazine Schiff base and phenyl dithiocarbamate mixed ligand metal complexes. Open J Appl Sci 8:346. https://doi.org/10.4236/ojapps.2018.88026
Fang Y, Wang Y, Zhao M, Lu Y, Li M, Zhang Y (2018) Bismuth(III) and diorganotin(IV) complexes of bis(2-acetylpyridine) thiocarbonohydrazone: synthesis, characterization, and apoptosis mechanism of action in vitro. Polyhedron 155:254–260. https://doi.org/10.1016/j.poly.2018.08.049
Fasching PA, Weihbrecht S, Haeberle L, Gasparyan A, Villalobos IE, Ma Y, Ekici AB, Wachter DL, Hartmann A, Beckmann MW, Slamon DJ, Press MF (2014) HER2 and TOP2A amplification in a hospital-based cohort of breast cancer patients: associations with patient and tumor characteristics. Breast Cancer Res Treat 145(1):193–203. https://doi.org/10.1007/s10549-014-2922-x
Frezza M, Schmitt S, Dou QP (2011) Targeting the ubiquitin-proteasome pathway: an emerging concept in cancer therapy. Curr Top Med Chem 11(23):2888–2905. https://doi.org/10.2174/156802611798281311
Gowda V, Sarma B, Laitinen RS, Larsson AC, Ivanov AV et al (2017) Structural insights into the polymorphism of bismuth(III) di-n-butyldithiocarbamate by X-ray diffraction, solid-state (13C/15N) CP-MAS NMR and DFT calculations. Polyhedron 129:123–132. https://doi.org/10.1016/j.poly.2017.03.018
Han J, Liu L, Yue X, Chang J, Shi W, Hua Y (2013) A binuclear complex constituted by diethyldithiocarbamate and copper(I) functions as a proteasome activity inhibitor in pancreatic cancer cultures and xenografts. Toxicol Appl Pharmacol 273(3):477–483. https://doi.org/10.1016/j.taap.2013.09.009
Honorat M, Mesnier A, Vendrell J, Guitton J, Bieche I, Lidereau R, Kruh GD, Dumontet C, Cohen P, Payen L (2008) ABCC11 expression is regulated by estrogen in MCF7 cells, correlated with estrogen receptor alpha expression in postmenopausal breast tumors and overexpressed in tamoxifen-resistant breast cancer cells. Endocr Relat Cancer 15(1):125–138. https://doi.org/10.1677/ERC-07-0189
Huang XC, Wang M, Wang HS et al (2014) Synthesis and antitumor activity of novel dipeptide derivatives derived from dehydroabietic acid. Bioorg Med Chem Lett 24(6):1511–1518. https://doi.org/10.1016/j.bmcl.2014.02.001
Ishak DHA (2017) Synthesis, characterization and biological activity of bismuth and antimony 1,1-dithiolates. MSc thesis. Universiti Malaya, Kuala Lumpur, Malaysia, 2017
Ishak DH, Ooi KK, Ang KP, Akim AM, Cheah YK, Nordin N, Halim SN, Seng HL, Tiekink ER (2014) A bismuth diethyldithiocarbamate compound promotes apoptosis in HepG2 carcinoma, cell cycle arrest and inhibits cell invasion through modulation of the NF-κB activation pathway. J Inorg Biochem 130:38–51. https://doi.org/10.1016/j.jinorgbio.2013.09.018
Jacobson AD, Zhang NY, Xu P, Han KJ, Noone S, Peng J, Liu CW (2009) The lysine 48 and lysine 63 ubiquitin conjugates are processed differently by the 26 s proteasome. J Biol Chem 284(51):35485–35494. https://doi.org/10.1074/jbc.M109.052928
Jänicke RU (2009) MCF-7 breast carcinoma cells do not express caspase-3. Breast Cancer Res Treat 117(1):219–221. https://doi.org/10.1007/s10549-008-0217-9
Jin J, Zhang W, Ji W, Yang F, Guan X (2017) Predictive biomarkers for triple negative breast cancer treated with platinum-based chemotherapy. Cancer Biol Ther 18(6):369–378. https://doi.org/10.1080/15384047.2017.1323582
Jones MD, Liu JC, Barthel TK, Hussain S, Lovria E, Cheng D, Schoonmaker JA, Mulay S, Ayers DC, Bouxsein ML, Stein GS, Mukherjee S, Lian JB (2010) A proteasome inhibitor, bortezomib, inhibits breast cancer growth and reduces osteolysis by downregulating metastatic genes. Clin Cancer Res 16(20):4978–4989. https://doi.org/10.1158/1078-0432.CCR-09-3293
Joshi S, Chauhan H, Carpenter N (2017) Preparation, spectroscopic characterization, and antimicrobial activities of mixed-metal (Sb and Bi) bridged derivatives with mixed sulfur donor ligands. J Mol Str 1128:221–229. https://doi.org/10.1016/j.molstruc.2016.08.063
Kadivar A, Kamalidehghan B, Akbari Javar H, Karimi B, Sedghi R, Noordin MI (2017) Antiproliferation effect of imatinib mesylate on MCF7, T-47D tumorigenic and MCF 10A nontumorigenic breast cell lines via PDGFR-β, PDGF-BB, c-Kit and SCF genes. Drug Des Devel Ther 11:469–481. https://doi.org/10.2147/DDDT.S124102
Kanchi S, Singh P, Bisetty K (2014) Dithiocarbamates as hazardous remediation agent: a critical review on progress in environmental chemistry for inorganic species studies of 20th century. Arab J Chem 7:11–25. https://doi.org/10.1016/j.arabjc.2013.04.026
Keogan DM, Griffith DM (2014) Current and potential applications of bismuth-based drugs. Molecules 19(9):15258–15297. https://doi.org/10.3390/molecules190915258
Kehrer JP (2000) The Haber-Weiss reaction and mechanisms of toxicity. Toxicology 149(1):43–50. https://doi.org/10.1016/s0300-483x(00)00231-6
Kumar R, Saneja A, Panda AK (2021) An annexin V-FITC—propidium iodide-based method for detecting apoptosis in a non-small cell lung cancer cell line. In: Santiago-Cardona PG (ed) Lung cancer: methods in molecular biology, vol 2279. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1278-1_17
Lai C, Tiekink E (2007) Prevalence of intermolecular Bi S interactions in bismuth dithiocarbamate compounds: Bi(S2CNR2)3. Z Kristallogr 222(10):532–538. https://doi.org/10.1524/zkri.2007.222.10.532
Li P, Nijhawan D, Budihardjo I, Srinivasula SM, Ahmad M, Alnemri ES, Wang X (1997) Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 91(4):479–489. https://doi.org/10.1016/s00928674(00)80434-1
Li Y, Seto E (2016) HDACs and HDAC inhibitors in cancer development and therapy. Cold Spring Harb Perspect Med 6(10):a026831. https://doi.org/10.1101/cshperspect.a026831
Li H, Lai CS, Wu J, Ho PC, de Vos D, Tiekink ER (2007) Cytotoxicity, qualitative structure-activity relationship (QSAR), and antitumor activity of bismuth dithiocarbamate complexes. J Inorg Biochem 101(5):809–816. https://doi.org/10.1016/j.jinorgbio.2007.01.010
Liu B, Ordonez-Ercan D, Fan Z, Edgerton SM, Yang X, Thor AD (2008) Downregulation of erbB3 abrogates erbB2-mediated tamoxifen resistance in breast cancer cells. Int J Cancer 120(9):1874–1882. https://doi.org/10.1002/ijc.22423
Liu L, Kawashima M, Toi M (2020) Breast cancer in global health: beyond diversity and inequality. Int J Surg: Global Health 3(6):e32. https://doi.org/10.1097/GH9.0000000000000032
Lizarazo-Jaimes EH, Monte-Neto RL, Reis PG, Fernandes NG, Speziali NL, Melo MN, Frézard F, Demicheli C (2012) Improved antileishmanial activity of Dppz through complexation with antimony(III) and bismuth(III): investigation of the role of the metal. Molecules 17(11):12622–12635. https://doi.org/10.3390/molecules171112622
López-Cardoso M, Tlahuext H, Pérez-Salgado M, Vargas-Pineda DG, Román-Bravo P et al (2020) Synthesis, crystal structure, antibacterial, antiproliferative, and QSAR studies of new bismuth(III) complexes of pyrrolidineditiocarbamates of dithia-bismolane and bismane, oxodithia-, and trithia-bismocane. J Mol Str 1217:128456. https://doi.org/10.1016/j.molstruc.2020.128456
Makeen HA, Mohan S, Al-Kasim MA, Sultan MH, Albarraq AA, Ahmed RA, Alhazmi HA, Alam MI (2021) Preparation, characterization, and anticancer activity of nanostructured lipid carriers containing imatinib. Pharmaceutics 13(7):1086. https://doi.org/10.3390/pharmaceutics13071086
Matsuno T, Kariya R, Yano S, Morino-Koga S, Taura M, Suico MA, Shimauchi Y, Matsuyama S, Okamoto Y, Shuto T, Kai H, Okada S (2011) Diethyldithiocarbamate induces apoptosis in HHV-8-infected primary effusion lymphoma cells via inhibition of the NF-κB pathway. Int J Oncol 40(4):1071–1078. https://doi.org/10.3892/ijo.2011.1313
McComb S et al (2019) Efficient apoptosis requires feedback amplification of upstream apoptotic signals by effector caspase-3 or -7. Sci Adv. https://doi.org/10.1126/sciadv.aau9433
Mc Gee MM, Hyland E, Campiani G, Ramunno A, Nacci V, Zisterer DM (2002) Caspase-3 is not essential for DNA fragmentation in MCF-7 cells during apoptosis induced by the pyrrolo-1,5-benzoxazepine, PBOX-6. FEBS Lett 515(1–3):66–70. https://doi.org/10.1016/s0014-5793(02)02440-7
Médale-Giamarchi C, Lajoie-Mazenc I, Malissein E, Meunier E, Couderc B, Bergé Y, Filleron T, Keller L, Marty C, Lacroix-Triki M, Dalenc F, Doisneau-Sixou SF, Favre G (2013) RhoB modifies estrogen responses in breast cancer cells by influencing expression of the estrogen receptor. Breast Cancer Res 15(1):R6. https://doi.org/10.1186/bcr3377
Morais C, Pat B, Gobe G, Johnson DW, Healy H (2006) Pyrrolidine dithiocarbamate exerts antiproliferative and pro-apoptotic effects in renal cell carcinoma cell lines. Nephrol Dial Transplant 21(12):3377–3388. https://doi.org/10.1093/ndt/gfl543
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65(1–2):55–63. https://doi.org/10.1016/0022-1759(83)90303-4
Müller BM, Jana L, Kasajima A, Lehmann A, Prinzler J, Budczies J, Winzer J, Dietel M, Weichert W, Denkert C (2013) Differential expression of histone deacetylases HDAC1, 2 and 3 in human breast cancer - overexpression of HDAC2 and HDAC3 is associated with clinicopathological indicators of disease progression. BMC Cancer 13:215. https://doi.org/10.1186/1471-2407-13-215
O’Connor L, Harris AW, Strasser A (2000) CD95 (Fas/APO-1) and p53 signal apoptosis independently in diverse cell types. Cancer Res 60(5):1217–1220
Ohtake F, Saeki Y, Ishido S, Kanno J, Tanaka K (2016) The K48–K63 branched ubiquitin chain regulates NF-kappaB signaling. Mol Cell 64:251–266. https://doi.org/10.1016/j.molcel.2016.09.014
Ouyang R, Yang Y, Tong X, Feng K, Yang Y, Tao H, Zhang X, Zong T, Cao P, Xiong F, Guo N, Li Y, Miao Y, Zhou S (2017) Potent anticancer activity of a new bismuth(III) complex against human lung cancer cells. J Inorg Biochem 168:18–26. https://doi.org/10.1016/j.jinorgbio.2016.12.006
Ozturk II, Banti CN, Kourkoumelis N, Manos MJ, Tasiopoulos AJ et al (2014) Synthesis, characterization and biological activity of antimony(III) or bismuth(III) chloride complexes with dithiocarbamate ligands derived from thiuram degradation. Polyhedron 67:89–103. https://doi.org/10.1016/j.poly.2013.08.052
Paca AM, Ajibade PA, Andrew FP, Nundkumar N, Singh M (2021) Synthesis, X-ray crystal structures and anticancer studies of four Pd(II) dithiocarbamate complexes. Arab J Chem 14(9):103326. https://doi.org/10.1016/j.arabjc.2021.103326
Pandey P, Saleh A, Nakazawa A, Kumar S, Srinivasula SM, Kumar V, Weichselbaum R, Nalin C, Alnemri ES, Kufe D, Kharbanda S (2000) Negative regulation of cytochrome c-mediated oligomerization of Apaf-1 and activation of procaspase-9 by heat shock protein 90. EMBO J 19:4310–4322. https://doi.org/10.1093/emboj/19.16.4310
Park M, Kim D, Ko S, Kim A, Mo K, Yoon H (2022) Breast cancer metastasis: mechanisms and therapeutic implications. Int J Mol Sci 23(12):6806. https://doi.org/10.3390/ijms23126806
Rajkumar SV, Richardson PG, Hideshima T, Anderson KC (2005) Proteasome inhibition as a novel therapeutic target in human cancer. J Clin Oncol 23(3):630–639. https://doi.org/10.1200/JCO.2005.11.030
Richard IA, Burgess JT, O’Byrne KJ, Bolderson E (2022) Beyond PARP1: the potential of other members of the poly (ADP-Ribose) polymerase family in DNA repair and cancer therapeutics. Front Cell Dev Biol 9:801200. https://doi.org/10.3389/fcell.2021.801200
Rosário JDS, Moreira FH, Rosa LHF, Guerra W, Silva-Caldeira PP (2023) Biological activities of bismuth compounds: an overview of the new findings and the old challenges not yet overcome. Molecules 28(15):5921. https://doi.org/10.3390/molecules28155921
Ross JS, Fletcher JA (1998) The HER-2/neu oncogene in breast cancer: prognostic factor, predictive factor, and target for therapy. Stem Cells 16(6):413–428. https://doi.org/10.1002/stem.160413
Roussidis AE, Theocharis AD, Tzanakakis GN, Karamanos NK (2007) The importance of c-Kit and PDGF receptors as potential targets for molecular therapy in breast cancer. Curr Med Chem 14(7):735–743. https://doi.org/10.2174/092986707780090963
Samali A, Cai J, Zhivotovsky B, Jones DP, Orrenius S (1999) Presence of a pre-apoptotic complex of procaspase-3, Hsp60 and Hsp10 in the mitochondrial fraction of jurkat cells. EMBO J 18(8):2040–2048. https://doi.org/10.1093/emboj/18.8.2040
Sato S, Fujita N, Tsuruo T (2000) Modulation of Akt kinase activity by binding to Hsp90. Proc Natl Acad Sci USA 97(20):10832–10837. https://doi.org/10.1073/pnas.170276797
Saraste A, Pulkki K (2000) Morphologic and biochemical hallmarks of apoptosis. Cardiovasc Res 45(3):528–537. https://doi.org/10.1016/s0008-6363(99)00384-3
Sedlacek J, Martins LDMRS, Danek P, Pombeiro AJL, Cvek B (2014) Diethyldithiocarbamate complexes with metals used as food supplements show different effects in cancer cells. J Appl Biomed 12:301–304. https://doi.org/10.1016/j.jab.2014.04.002
Seo J, Min SK, Park R, Kim DH, Kwon MJ, Kim LS, Ju S (2014) Expression of HDAC1, HDAC2, HDAC3, and HDAC6 in invasive ductal carcinomas of the breast. J Breast Cancer 17(4):323–331. https://doi.org/10.4048/jbc.2014.17.4.323
Shakibaie M, Forootanfar H, Ameri A, Jafari M, Rahimi HR (2018) Cytotoxicity of biologically synthesised bismuth nanoparticles against HT-29 cell line. IET Nanobiotechnol 12(5):653–657. https://doi.org/10.1049/iet-nbt.2017.0295
Shamseddine AI, Farhat FS (2011) Platinum-based compounds for metastatic breast cancer treatment. Chemotherapy 57(6):468–487. https://doi.org/10.1159/000334093
Simpson P, Desai NM, Casari I, Massi M, Falasca M (2019) Metal-based antitumor compounds: beyond cisplatin. Future Med Chem 11(2):119–135. https://doi.org/10.4155/fmc-2018-0248
Singh S, Bharti N, Mohapatra PP (2009) Chemistry and biology of synthetic and naturally occurring antiamoebic agents. Chem Rev 109(5):1900–1947. https://doi.org/10.1021/cr068217k
Singh L, Aldosary S, Saeedan AS, Ansari MN, Kaithwas G (2018) Prolyl hydroxylase 2: a promising target to inhibit hypoxia-induced cellular metabolism in cancer cells. Drug Discov Today 23(11):1873–1882. https://doi.org/10.1016/j.drudis.2018.05.016
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021) Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71:209–249. https://doi.org/10.3322/caac.21660
Tahata S, Yuan B, Kikuchi H, Takagi N, Hirano T, Toyoda H (2014) Cytotoxic effects of pyrrolidine dithiocarbamate in small-cell lung cancer cells, alone and in combination with cisplatin. Int J Oncol 45(4):1749–1759. https://doi.org/10.3892/ijo.2014.2564
Takayama S, Reed JC, Homma S (2003) Heat-shock proteins as regulators of apoptosis. Oncogene 22(56):9041–9047. https://doi.org/10.1038/sj.onc.1207114
Tang D, Kidd VJ (1998) Cleavage of DFF-45/ICAD by multiple caspases is essential for its function during apoptosis. J Biol Chem 273(44):28549–28552. https://doi.org/10.1074/jbc.273.44.28549
Tomblin JK, Salisbury TB (2014) Insulin-like growth factor 2 regulation of aryl hydrocarbon receptor in MCF-7 breast cancer cells. Biochem Biophys Res Comm 443(3):1092–1096. https://doi.org/10.1016/j.bbrc.2013.12.112
Tiekink ERT (2002) Antimony and bismuth compounds in oncology. Crit Rev Oncol Hemat 42(3):217–224. https://doi.org/10.1016/s1040-8428(01)00217-7
Tu G, Yao ZX (2004) Pyrrolidine dithiocarbamate inhibits NF-κB activation and enhances TNF-induced apoptosis in human breast cancer cells. Chin J Cancer Res 16:99–104. https://doi.org/10.1007/s11670-004-0005-y
Turk K, Grześkiewicz AM, Banti CN, Hadjikakou SK, Kubicki M, Ozturk II (2022) Synthesis, characterization, and biological properties of mono-, di- and poly-nuclear bismuth (III) halide complexes containing thiophene-2-carbaldehyde thiosemicarbazones. J Inorg Biochem 237:111987. https://doi.org/10.1016/j.jinorgbio.2022.111987
Viola-Rhenals M, Patel KR, Jaimes-Santamaria L, Wu G, Liu J, Dou QP (2018) Recent advances in antabuse (disulfiram): the importance of its metal-binding ability to its anticancer activity. Curr Med Chem 25(4):506–524. https://doi.org/10.2174/0929867324666171023161121
Wang R, Li H, Sun H (2019) Bismuth: environmental pollution and health effects. Encyclopedia of environmental health, 2nd edn. Elsevier, Amsterdam, pp 415–423. https://doi.org/10.1016/B978-0-12-409548-9.11870-6
Welsh SJ, Bellamy WT, Briehl MM, Powis G (2002) The redox protein thioredoxin-1 (Trx-1) increases hypoxia-inducible factor 1 protein expression: Trx-1 overexpression results in increased vascular endothelial growth factor production and enhanced tumor angiogenesis. Cancer Res 62(17):5089–5095
Yang C, Zhang H, Huang W, Lin Q, Wei H (2010) Effect of combined use of PDTC and paclitaxel on proliferation and invasion of human breast cancer cell line MCF-7. Sheng Wu Yi Xue Gong Cheng Xue Za Zhi 27(5):1105
Yarar S, Ozturk I, Banti C, Panagiotou N, Papatriantafyllopoulou C, Manoli M, Manos M, Tasiopoulos A, Hadjikakou S (2018) Synthesis, characterization and cytotoxic properties of bismuth(III) chloride complexes with heterocyclic thioamides. Inorg Chim Acta 471:23–33. https://doi.org/10.1016/j.ica.2017.10.026
Yu S, Cai X, Wu C, Liu Y, Zhang J, Gong X, Wang X, Wu X, Zhu T, Mo L, Gu J, Yu Z, Chen J, Thiery JP, Chai R, Chen L (2017) Targeting HSP90-HDAC6 regulating network implicates precision treatment of breast cancer. Int J Biol Sci 13(4):505–517. https://doi.org/10.7150/ijbs.18834
Zhang J, Xu M (2000) DNA fragmentation in apoptosis. Cell Res 10:205–211. https://doi.org/10.1038/sj.cr.7290049
Zhang H, Wu J-S, Peng F (2008) Potent anticancer activity of pyrrolidine dithiocarbamate–copper complexes against cisplatin-resistant neuroblastoma cells. Anticancer Drugs 19(2):125–132. https://doi.org/10.1097/CAD.0b013e3282f2bdff
Zhang L, Lei J, Liu J, Ma F, Ju H (2015) In situ activation and monitoring of the evolution of the intracellular caspase family. Chem Sci 6(6):3365–3372. https://doi.org/10.1039/c5sc00471c
Zhang Y, Yan J, Wang L, Dai H, Li N, Hu W, Cai H (2017) HIF-1α promotes breast cancer cell MCF-7 proliferation and invasion through regulating miR-210. Cancer Biother Radiopharm 32(8):297–301. https://doi.org/10.1089/cbr.2017.2270
Zhang T, Ma C, Zhang Z, Zhang H, Hu H (2021) NF-κB signaling in inflammation and cancer. MedComm 2(4):618–653. https://doi.org/10.1002/mco2.104
Zhu X, Wang K, Zhang K, Xu F, Yin Y, Zhu L, Zhou F (2016) Galectin-1 knockdown in carcinoma-associated fibroblasts inhibits migration and invasion of human MDA-MB-231 breast cancer cells by modulating MMP-9 expression. Acta Biochim Biophys Sin (Shanghai) 48(5):462–467. https://doi.org/10.1093/abbs/gmw019
Acknowledgements
The authors thanked Prof Edward R.T. Tiekink for providing the sample and Universiti Malaya for providing the grants under High Impact Research Schemes and collaborated with us by providing the compounds and the funding. The authors also thanked Prof Dr Noor Saadah Abd Rahman, the Deputy Vice Chancellor (Research and Innovation) at the University of Malaya for giving us the consent to publish the data
Funding
This research was funded by the Ministry of Higher Education, Malaysia (grant numbers UM.C/HIR-MOHE/SC/03 and UM.C/HIR-MOHE/SC/12).
Author information
Authors and Affiliations
Contributions
Conceptualization, RAH; methodology, RAH; validation, CPF and RAH; formal analysis, CPF and AKP; investigation, CPF and AKP; data curation, CPF and AKP; writing—original draft preparation, CPF; writing—review and editing, RAH; visualization, CPF and AKP; supervision, RAH; project administration, RAH All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chan, P.F., Ang, K.P. & Hamid, R.A. Cytotoxicity of bismuth(III) dithiocarbamate derivatives by promoting a mitochondrial-dependent apoptotic pathway and suppressing MCF-7 breast adenocarcinoma cell invasion. J Biol Inorg Chem (2024). https://doi.org/10.1007/s00775-023-02041-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00775-023-02041-x