Abstract
Purpose
To explore whether letrozole improved outcomes in subsequent controlled ovarian hyperstimulation (COH) cycles.
Methods
This was a retrospective repeated measures cohort study examining COH cycles. Patients were included if they underwent two cycles for unexplained infertility, male factor infertility, or planned oocyte/embryo cryopreservation. The first cycles for all patients implemented a non-letrozole, conventional gonadotropin protocol. Second cycles for the study group included letrozole (2.5–7.5 mg for 5 days) with no medication change to second cycles amongst controls.
Our primary objective was to compare oocyte yield. Cohorts were then subdivided by pursuit of oocyte (OC) or embryo (IVF) cryopreservation. Secondary outcome amongst the OC subgroup was oocyte maturation index (metaphase II (MII)/total oocytes). Secondary outcomes amongst the IVF subgroup were normal fertilization rate (2-pronuclear zygotes (2PN)/oocytes exposed to sperm), blastocyst formation rate (blastocysts/2PNs), and embryo ploidy (%euploid and aneuploid).
Results
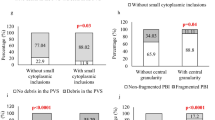
Fifty-four cycles (n = 27) were included in letrozole and 108 cycles (n = 54) were included in control. Oocyte yield was higher in second cycles (p < 0.008) in the letrozole group but similar in second cycles (p = 0.26) amongst controls. Addition of letrozole did not impact MII index (p = 0.90); however, MII index improved in second cycles amongst controls (p < 0.001). Both groups had similar rates of normal fertilization (letrozole: p = 0.52; control: p = 0.61), blast formation (letrozole: p = 0.61; control: p = 0.84), euploid (letrozole: p = 0.29; control: p = 0.47), and aneuploid embryos (letrozole: p = 0.17; control: p = 0.78) between cycles.
Conclusions
Despite improved oocyte yield, letrozole did not yield any difference in oocyte maturation or embryo outcomes.
Similar content being viewed by others
References
Fabian CJ. The what, why and how of aromatase inhibitors: hormonal agents for treatment and prevention of breast cancer. Int J Clin Pract. 2007;61(12):2051–63. https://doi.org/10.1111/j.1742-1241.2007.01587.
Kasuga-Yamashita F, Baba T, Nagao S, Fujibe Y, Morishita M, Kuno Y, Mariya T, Honnma H, Endo T, Kiya T, Saito T. Letrozole increases preantral follicle growth and decreases estradiol production without impairing follicle survival. J Ovarian Res. 2022;15(1):136. https://doi.org/10.1186/s13048-022-01073-2.
Legro RS, Brzyski RG, Diamond MP, Coutifaris C, Schlaff WD, Casson P. Letrozole versus clomiphene for infertility in the polycystic ovary syndrome. N Engl J Med. 2014;371:119–29. https://doi.org/10.1056/NEJMoa1313517.
Pavone ME, Bulun SE. Clinical review: the use of aromatase inhibitors for ovulation induction and superovulation. J Clin Endocrinol Metab. 2013;98:1838–44. https://doi.org/10.1210/jc.2013-1328.
Oktay K, Hourvitz A, Sahin G, Oktem O, Safro B, Cil A, et al. Letrozole reduces estrogen and gonadotropin exposure in women with breast cancer undergoing ovarian stimulation before chemotherapy. J Clin Endocrinol Metab. 2006;91:3885–90. https://doi.org/10.1210/jc.2006-0962.
Keskin U, Ercan CM, Yilmaz A, Babacan A, Korkmaz C, Duru NK, et al. Random-start controlled ovarian hyperstimulation with letrozole for fertility preservation in cancer patients: case series and review of literature. J Pak Med Assoc. 2014;64:830–2.
Khojah M, Khayat S, Dahan MH. Comparison of in vitro fertilization cycles stimulated with 20 mg letrozole daily versus high-dose gonadotropins in Rotterdam Consensus ultra-suboptimal responders: a proof of concept. Int J Gynaecol Obstet. 2022;156(1):102–6. https://doi.org/10.1002/ijgo.13626.
Pajai S, Potdar J, Gopal U, Banait T. A review on the use of letrozole in female and male infertility. Cureus. 2022;14(11): e31291. https://doi.org/10.7759/cureus.31291.
Elizur SE, Tulandi T. Drugs in infertility and fetal safety. Fertil Steril. 2008;89(6):1595–602. https://doi.org/10.1016/j.fertnstert.2008.02.092.
Pereira N, Hancock K, Cordeiro CN, Lekovich JP, Schattman GL, Rosenwaks Z. Comparison of ovarian stimulation response in patients with breast cancer undergoing ovarian stimulation with letrozole and gonadotropins to patients undergoing ovarian stimulation with gonadotropins alone for elective cryopreservation of oocytes. Gynecol Endocrinol. 2016;32(10):823–6. https://doi.org/10.1080/09513590.2016.1177013.
Oktay K, Buyuk E, Libertella N, Akar M, Rosenwaks Z. Fertility preservation in breast cancer patients: a prospective controlled comparison of ovarian stimulation with tamoxifen and letrozole for embryo cryopreservation. J Clin Oncol. 2005;23(19):4347–53. https://doi.org/10.1200/JCO.2005.05.037.
Bülow NS, Warzecha AK, Nielsen MV, Andersen CY, Holt MD, Petersen MR, Sopa N, Zedeler A, Englund AL, Pinborg A, Grøndahl ML, Skouby SO, Macklon NS. Impact of letrozole co-treatment during ovarian stimulation on oocyte yield, embryo development, and live birth rate in women with normal ovarian reserve: secondary outcomes from the RIOT trial. Hum Reprod. 2023 Sep 12:dead182. https://doi.org/10.1093/humrep/dead182.
Turan V, Gayete-Lafuente S, Bang H, Oktay KH. Outcomes of random-start letrozole protocol with PGT-A in women with breast cancer undergoing fertility preservation. J Assist Reprod Genet. 2023;40(10):2401–8. https://doi.org/10.1007/s10815-023-02882-0.
Shapira M, Orvieto R, Lebovitz O, Nahum R, Aizer A, Segev-Zahav A, Haas J. Does daily co administration of gonadotropins and letrozole during the ovarian stimulation improve IVF outcome for suboptimal and sub optimal responders? J Ovarian Res. 2020;13(1):66. https://doi.org/10.1186/s13048-020-00666-z.
Garcia-Velasco JA, Moreno L, Pacheco A, Guillén A, Duque L, Requena A, Pellicer A. The aromatase inhibitor letrozole increases the concentration of intraovarian androgens and improves in vitro fertilization outcome in low responder patients: a pilot study. Fertil Steril. 2005;84(1):82–7. https://doi.org/10.1016/j.fertnstert.2005.01.117.
Goldman KN, Noyes NL, Knopman JM, McCaffrey C, Grifo JA. Oocyte efficiency: does live birth rate differ when analyzing cryopreserved and fresh oocytes on a per-oocyte basis? Fertil Steril. 2013;100(3):712–7. https://doi.org/10.1016/j.fertnstert.2013.04.040.
Grifo JA, Noyes N. Delivery rate using cryopreserved oocytes is comparable to conventional in vitro fertilization using fresh oocytes: potential fertility preservation for female cancer patients. Fertil Steril. 2010;93(2):391–6. https://doi.org/10.1016/j.fertnstert.2009.02.067.
Noyes N, Knopman J, Labella P, McCaffrey C, Clark-Williams M, Grifo J. Oocyte cryopreservation outcomes including pre-cryopreservation and post-thaw meiotic spindle evaluation following slow cooling and vitrification of human oocytes. Fertil Steril. 2010;94(6):2078–82. https://doi.org/10.1016/j.fertnstert.2010.01.019.
Sonigo C, Sermondade N, Calvo J, Benard J, Sifer C, Grynberg M. Impact of letrozole supplementation during ovarian stimulation for fertility preservation in breast cancer patients. Eur J Obstet Gynecol Reprod Biol X. 2019;11(4): 100049. https://doi.org/10.1016/j.eurox.2019.100049.
Goswami SK, Das T, Chattopadhyay R, Sawhney V, Kumar J, Chaudhury K, Chakravarty BN, Kabir SN. A randomized single-blind controlled trial of letrozole as a low-COHt IVF protocol in women with suboptimal ovarian response: a preliminary report. Hum Reprod. 2004;19(9):2031–5. https://doi.org/10.1093/humrep/deh359.
İsrafilova G, Şükür YE, Özkavukcu S, et al. Comparison of oocyte and embryo quality between random start and controlled ovarian stimulation cycles in cancer patients undergoing fertility preservation. Reprod Sci. 2021;28(8):2200–7. https://doi.org/10.1007/s43032-020-00412-2.
Gleicher N, Weghofer A, Barad DH. The role of androgens in follicle maturation and ovulation induction: friend or foe of infertility treatment? Reprod Biol Endocrinol. 2011;17(9):116. https://doi.org/10.1186/1477-7827-9-116.
Lebbe M, Woodruff TK. Involvement of androgens in ovarian health and disease. Mol Hum Reprod. 2013;19(12):828–37. https://doi.org/10.1093/molehr/gat065.
Vendola K, Zhou J, Wang J, Bondy CA. Androgens promote insulin-like growth factor-I and insulin-like growth factor-I receptor gene expression in the primate ovary. Hum Reprod. 1999;14(9):2328–32. https://doi.org/10.1093/humrep/14.9.2328.
Acknowledgements
The authors would like to acknowledge the hard work and mentorship of the clinical and embryology team at the NYU Langone Fertility Center, to make this research possible for trainees.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jain, N.S., Licciardi, F., Kalluru, S. et al. Reassessing the impact of letrozole co-administration in controlled ovarian hyperstimulation: findings from a single-center repeated measures study. J Assist Reprod Genet 41, 979–987 (2024). https://doi.org/10.1007/s10815-024-03067-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-024-03067-z