Abstract
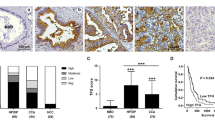
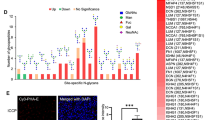
Aberrant glycosylation is an important factor in facilitating tumor progression and therapeutic resistance. In this study, using Wisteria floribunda agglutinin (WFA), we examined the expression of WFA-binding glycans (WFAG) in cholangiocarcinoma (CCA). The results showed that WFAG was highly detected in precancerous and cancerous lesions of human CCA tissues, although it was rarely detected in normal bile ducts. The positive signal of WFAG in the cancerous lesion accounted for 96.2% (50/52) of the cases. Overexpression of WFAG was significantly associated with lymph node and distant metastasis (P < 0.05). The study using the CCA hamster model showed that WFAG is elevated in preneoplastic and neoplastic bile ducts as early as 1 month after being infected with liver fluke and exposed to N-nitrosodimethylamine. Functional analysis was performed to reveal the role of WFAG in CCA. The CCA cell lines KKU-213A and KKU-213B were treated with WFA, followed by migration assay. Our data suggested that WFAG facilitates the migration of CCA cells via the activation of the Akt and ERK signaling pathways. In conclusion, we have demonstrated the association of WFAG with carcinogenesis and metastasis of CCA, suggesting its potential as a target for the treatment of the disease.





Similar content being viewed by others
Reference
Bojar D, Meche L, Meng G, Eng W, Smith DF, Cummings RD, Mahal LK (2022) A useful guide to lectin binding: machine-learning directed annotation of 57 unique lectin specificities. ACS Chem Biol 17(11):2993–3012. https://doi.org/10.1021/acschembio.1c00689
Che MI, Huang J, Hung JS, Lin YC, Huang MJ, Lai HS, Hsu WM, Liang JT, Huang MC (2014) beta1, 4-N-acetylgalactosaminyltransferase III modulates cancer stemness through EGFR signaling pathway in colon cancer cells. Oncotarget 5(11):3673–3684. https://doi.org/10.18632/oncotarget.1981
Detarya M, Sawanyawisuth K, Aphivatanasiri C, Chuangchaiya S, Saranaruk P, Sukprasert L, Silsirivanit A, Araki N, Wongkham S, Wongkham C (2020) The O-GalNAcylating enzyme GALNT5 mediates carcinogenesis and progression of cholangiocarcinoma via activation of AKT/ERK signaling. Glycobiology 30(5):312–324. https://doi.org/10.1093/glycob/cwz098
Detarya M, Lert-Itthiporn W, Mahalapbutr P, Klaewkla M, Sorin S, Sawanyawisuth K, Silsirivanit A, Seubwai W, Wongkham C, Araki N, Wongkham S (2022) Emerging roles of GALNT5 on promoting EGFR activation in cholangiocarcinoma: a mechanistic insight. Am J Cancer Res 12(9):4140–4159
Fedchenko N, Reifenrath J (2014) Different approaches for interpretation and reporting of immunohistochemistry analysis results in the bone tissue - a review. Diagn Pathol 9:221. https://doi.org/10.1186/s13000-014-0221-9
Haji-Ghassemi O, Gilbert M, Spence J, Schur MJ, Parker MJ, Jenkins ML, Burke JE, van Faassen H, Young NM, Evans SV (2016) Molecular basis for recognition of the cancer glycobiomarker, LacdiNAc (GalNAc[beta1–>4]GlcNAc), by Wisteria floribunda Agglutinin. J Biol Chem 291(46):24085–24095. https://doi.org/10.1074/jbc.M116.750463
Hirano K, Furukawa K (2022) Biosynthesis and biological significances of LacdiNAc group on N- and O-Glycans in human cancer cells. Biomolecules. https://doi.org/10.3390/biom12020195
Hsu WM, Che MI, Liao YF, Chang HH, Chen CH, Huang YM, Jeng YM, Huang J, Quon MJ, Lee H, Huang HC, Huang MC (2011) B4GALNT3 expression predicts a favorable prognosis and suppresses cell migration and invasion via beta(1) integrin signaling in neuroblastoma. Am J Pathol 179(3):1394–1404. https://doi.org/10.1016/j.ajpath.2011.05.025
Huang J, Liang JT, Huang HC, Shen TL, Chen HY, Lin NY, Che MI, Lin WC, Huang MC (2007) Beta 1,4-N-acetylgalactosaminyltransferase III enhances malignant phenotypes of colon cancer cells. Mol Cancer Res 5(6):543–552. https://doi.org/10.1158/1541-7786.MCR-06-0431
Indramanee S, Silsirivanit A, Pairojkul C, Wongkham C, Wongkham S (2012) Aberrant glycosylation in cholangiocarcinoma demonstrated by lectin-histochemistry. Asian Pac J Cancer Prev 13(Suppl):119–124
Indramanee S, Sawanyawisuth K, Silsirivanit A, Dana P, Phoomak C, Kariya R, Klinhom-On N, Sorin S, Wongkham C, Okada S, Wongkham S (2019) Terminal fucose mediates progression of human cholangiocarcinoma through EGF/EGFR activation and the Akt/Erk signaling pathway. Sci Rep 9(1):17266. https://doi.org/10.1038/s41598-019-53601-8
Kailemia MJ, Park D, Lebrilla CB (2017) Glycans and glycoproteins as specific biomarkers for cancer. Anal Bioanal Chem 409(2):395–410. https://doi.org/10.1007/s00216-016-9880-6
Kuno A, Ikehara Y, Tanaka Y, Ito K, Matsuda A, Sekiya S, Hige S, Sakamoto M, Kage M, Mizokami M, Narimatsu H (2013) A serum “sweet-doughnut” protein facilitates fibrosis evaluation and therapy assessment in patients with viral hepatitis. Sci Rep 3(1):1065. https://doi.org/10.1038/srep01065
Li J, Zhao T, Li J, Shen J, Jia L, Zhu B, Dang L, Ma C, Liu D, Mu F, Hu L, Sun S (2022) Precision N-glycoproteomics reveals elevated LacdiNAc as a novel signature of intrahepatic cholangiocarcinoma. Mol Oncol 16(11):2135–2152. https://doi.org/10.1002/1878-0261.13147
Maruyama M, Kobayashi N, Westerman KA, Sakaguchi M, Allain JE, Totsugawa T, Okitsu T, Fukazawa T, Weber A, Stolz DB, Leboulch P, Tanaka N (2004) Establishment of a highly differentiated immortalized human cholangiocyte cell line with SV40T and hTERT. Transplantation 77(3):446–451. https://doi.org/10.1097/01.Tp.0000110292.73873.25
Matsuda A, Kuno A, Kawamoto T, Matsuzaki H, Irimura T, Ikehara Y, Zen Y, Nakanuma Y, Yamamoto M, Ohkohchi N, Shoda J, Hirabayashi J, Narimatsu H (2010) Wisteria floribunda agglutinin-positive mucin 1 is a sensitive biliary marker for human cholangiocarcinoma. Hepatology 52(1):174–182. https://doi.org/10.1002/hep.23654
Matsuda A, Kuno A, Nakagawa T, Ikehara Y, Irimura T, Yamamoto M, Nakanuma Y, Miyoshi E, Nakamori S, Nakanishi H, Viwatthanasittiphong C, Srivatanakul P, Miwa M, Shoda J, Narimatsu H (2015) Lectin microarray-based sero-Biomarker verification targeting aberrant O-linked glycosylation on mucin 1. Anal Chem 87(14):7274–7281. https://doi.org/10.1021/acs.analchem.5b01329
Matsuda A, Higashi M, Nakagawa T, Yokoyama S, Kuno A, Yonezawa S, Narimatsu H (2017) Assessment of tumor characteristics based on glycoform analysis of membrane-tethered MUC1. Lab Invest 97(9):1103–1113. https://doi.org/10.1038/labinvest.2017.53
Munkley J, Elliott DJ (2016) Hallmarks of glycosylation in cancer. Oncotarget 7(23):35478–35489. https://doi.org/10.18632/oncotarget.8155
Narimatsu H, Sato T (2018) Wisteria floribunda agglutinin positive glycobiomarkers: a unique lectin as a serum biomarker probe in various diseases. Expert Rev Proteomics 15(2):183–190. https://doi.org/10.1080/14789450.2018.1419066
Narimatsu Y, Kuno A, Ito H, Kaji H, Kaneko S, Usui J, Yamagata K, Narimatsu H (2014) IgA nephropathy caused by unusual polymerization of IgA1 with aberrant N-Glycosylation in a patient with monoclonal immunoglobulin deposition disease. PloS One 9(3):e91079. https://doi.org/10.1371/journal.pone.0091079
Ohtsubo K, Marth JD (2006) Glycosylation in cellular mechanisms of health and disease. Cell 126(5):855–867. https://doi.org/10.1016/j.cell.2006.08.019
Panawan O, Silsirivanit A, Chang CH, Putthisen S, Boonnate P, Yokota T, Nishisyama-Ikeda Y, Detarya M, Sawanyawisuth K, Kaewkong W, Muisuk K, Luang S, Vaeteewoottacharn K, Kariya R, Yano H, Komohara Y, Ohta K, Okada S, Wongkham S, Araki N (2023) Establishment and characterization of a novel cancer stem-like cell of cholangiocarcinoma. Cancer Sci. https://doi.org/10.1111/cas.15812
Poiroux G, Barre A, van Damme EJM, Benoist H, Rouge P (2017) Plant lectins targeting o-glycans at the cell surface as tools for cancer diagnosis, prognosis and therapy. Int J Mol Sci. https://doi.org/10.3390/ijms18061232
Prakobwong S, Yongvanit P, Hiraku Y, Pairojkul C, Sithithaworn P, Pinlaor P, Pinlaor S (2010) Involvement of MMP-9 in peribiliary fibrosis and cholangiocarcinogenesis via Rac1-dependent DNA damage in a hamster model. Int J Cancer 127(11):2576–2587. https://doi.org/10.1002/ijc.25266
Saentaweesuk W, Silsirivanit A, Vaeteewoottacharn K, Sawanyawisuth K, Pairojkul C, Cha’on U, Indramanee S, Pinlaor S, Boonmars T, Araki N, Wongkham C (2018) Clinical significance of GalNAcylated glycans in cholangiocarcinoma: Values for diagnosis and prognosis. Clin Chim Acta 477:66–71. https://doi.org/10.1016/j.cca.2017.12.005
Sato T, Tateno H, Kaji H, Chiba Y, Kubota T, Hirabayashi J, Narimatsu H (2017) Engineering of recombinant Wisteria floribunda agglutinin specifically binding to GalNAcbeta1,4GlcNAc (LacdiNAc). Glycobiology 27(8):743–754. https://doi.org/10.1093/glycob/cwx038
Shin HR, Oh JK, Masuyer E, Curado MP, Bouvard V, Fang YY, Wiangnon S, Sripa B, Hong ST (2010) Epidemiology of cholangiocarcinoma: an update focusing on risk factors. Cancer Sci 101(3):579–585. https://doi.org/10.1111/j.1349-7006.2009.01458.x
Shoda J, Matsuda A, Shida T, Yamamoto M, Nagino M, Tsuyuguchi T, Yasaka T, Tazuma S, Uchiyama K, Unno M, Ohkohchi N, Nakanuma Y, Kuno A, Narimatsu H (2017) Wisteria floribunda agglutinin-sialylated mucin core polypeptide 1 is a sensitive biomarker for biliary tract carcinoma and intrahepatic cholangiocarcinoma: a multicenter study. J Gastroenterol 52(2):218–228. https://doi.org/10.1007/s00535-016-1230-0
Silsirivanit A (2019) Glycosylation markers in cancer. Adv Clin Chem 89:189–213. https://doi.org/10.1016/bs.acc.2018.12.005
Silsirivanit A (2021) Glycans: potential therapeutic targets for cholangiocarcinoma and their therapeutic and diagnostic implications. Expert Opin Ther Targets 25(1):1–4. https://doi.org/10.1080/14728222.2021.1861250
Silsirivanit A, Araki N, Wongkham C, Pairojkul C, Narimatsu Y, Kuwahara K, Narimatsu H, Wongkham S, Sakaguchi N (2011) A novel serum carbohydrate marker on mucin 5AC: values for diagnostic and prognostic indicators for cholangiocarcinoma. Cancer 117(15):3393–3403. https://doi.org/10.1002/cncr.25912
Silsirivanit A, Phoomak C, Wongkham S (2021) Glycosylation in cholangiocarcinoma development and metastasis: diagnostic and therapeutic considerations. In: Tabibian, J.H. (eds) Diagnosis and management of cholangiocarcinoma: a multidisciplinary approach. Springer, Cham, pp 527–553. https://doi.org/10.1007/978-3-030-70936-5_25
Sperandio M, Gleissner CA, Ley K (2009) Glycosylation in immune cell trafficking. Immunol Rev 230(1):97–113. https://doi.org/10.1111/j.1600-065X.2009.00795.x
Sripa B, Pairojkul C (2008) Cholangiocarcinoma: lessons from Thailand. Curr Opin Gastroenterol 24(3):349–356. https://doi.org/10.1097/MOG.0b013e3282fbf9b3
Sripa B, Seubwai W, Vaeteewoottacharn K, Sawanyawisuth K, Silsirivanit A, Kaewkong W, Muisuk K, Dana P, Phoomak C, Lert-Itthiporn W, Luvira V, Pairojkul C, Teh BT, Wongkham S, Okada S, Chamgramol Y (2020) Functional and genetic characterization of three cell lines derived from a single tumor of an Opisthorchis viverrini-associated cholangiocarcinoma patient. Hum Cell 33(3):695–708. https://doi.org/10.1007/s13577-020-00334-w
Teeravirote K, Sutthanut K, Thonsri U, Mahalapbutr P, Seubwai W, Luang S, Tippayawat P, Kanthawong S, Pipattanaboon C, Duangjinda M, Chankitisakul V, Silsirivanit A (2022) Anserine/Carnosine-rich extract from Thai Native chicken suppresses melanogenesis via activation of ERK signaling pathway. Molecules. https://doi.org/10.3390/molecules27217440
Titapun A, Pugkhem A, Luvira V, Srisuk T, Somintara O, Saeseow OT, Sripanuskul A, Nimboriboonporn A, Thinkhamrop B, Khuntikeo N (2015) Outcome of curative resection for perihilar cholangiocarcinoma in Northeast Thailand. World J Gastrointest Oncol 7(12):503–512. https://doi.org/10.4251/wjgo.v7.i12.503
Wang Y, Li K, Zhao W, Liu Z, Liu J, Shi A, Chen T, Mu W, Xu Y, Pan C, Zhang Z (2021) Aldehyde dehydrogenase 3B2 promotes the proliferation and invasion of cholangiocarcinoma by increasing Integrin Beta 1 expression. Cell Death Dis 12(12):1158. https://doi.org/10.1038/s41419-021-04451-8
Acknowledgments
This project was funded by Kasetsart University through the Graduate School Fellowship Program and Faculty of Public Health, Kasetsart University, Thailand, to W.P. and S.C. and by the Fundamental Fund of Khon Kaen University (FF65) to A.S. We thank Prof. Yukifumi Nawa for English editing via the KKU publication clinic.
Funding
Fundamental Fund of Khon Kaen University (FF65). Kasetsart University through the Graduate School Fellowship Program
Author information
Authors and Affiliations
Contributions
W.P., S.P., O.P., P.M., K.T., and M.D. performed experiments and analyzed data; W.P., S.P, and P.M. prepared Figs. 1–2; W.P., O.P., P.S., and S.U. prepared Figs. 3–4; T.S., A.K., P.M., and S.L. supervised experiment handling and data analysis; S.C. and A.S. designed the study and funding acquisition; and W.P., S.P., S.C., and A.S. wrote and edited manuscript.
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Phuyathip, W., Putthisen, S., Panawan, O. et al. Role of Wisteria floribunda agglutinin binding glycans in carcinogenesis and metastasis of cholangiocarcinoma. Histochem Cell Biol 161, 423–434 (2024). https://doi.org/10.1007/s00418-024-02270-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-024-02270-4