Abstract
Background
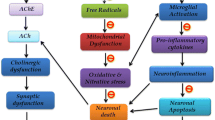
Despite extensive global efforts, there is currently no effective remedy for Alzheimer’s disease (AD).
Objectives
This investigation aimed to examine the impact of vitamin D/vitamin D receptor (VDR)/ATG16L1 signaling on memory in AD mice, as well as the levels of autophagy and inflammation in the hippocampus.
Results
Administering cholecalciferol cholesterol emulsion (CCE), which serves as a precursor to 1,25-OH vitamin D3, through water feeding for a duration of 12 weeks demonstrated the ability to mitigate both short-term and long-term memory impairments in AD mice. CCE treatment reduced the deposition of TAU protein in the hippocampus, suppressed the phosphorylation of TAU Thr181 and 212, and significantly decreased Aβ (1–40) levels. Moreover, CCE heightened hippocampal autophagy in AD mice, upregulated the expression of ATG16L1 and LC3B, and inhibited P62 expression. In addition, CCE supplementation attenuated hippocampal inflammation in AD mice by decreasing the levels of IL1β and TNFα and restoring the TOM1/IL1R1 pathway. The Luciferase reporter assay verified that the knockdown of VDR could impede the activation of the ATG16L1 promoter by vitamin D, and ChIP analysis confirmed that VDR could bind to the ATG16L1 promoter and exert its function.
Conclusion
CCE exhibits potential in safeguarding hippocampal neurons against AD-induced harm, stimulating autophagy, and suppressing inflammation. These effects may be regulated by VD/VDR/ATG16L1 signaling.





Similar content being viewed by others
Data availability
The original contributions presented in the study are included in the article, further inquiries can be directed to the corresponding authors.
References
Akiyama H, Barger S, Barnum S, Bradt B, Bauer J, Cole GM, Cooper NR, Eikelenboom P, Emmerling M, Fiebich BL et al (2000) Inflammation and Alzheimer’s disease. Neurobiol Aging 21(3):383–421
Arévalo NB, Castillo-Godoy DP, Espinoza-Fuenzalida I, Rogers NK, Farias G, Delgado C, Henriquez M, Herrera L, Behrens MI, SanMartín CD (2021) Association of vitamin D receptor polymorphisms with amyloid-Β transporters expression and risk of mild cognitive impairment in a Chilean cohort. J Alzheimers Dis 82(s1):S283–S297
Benilova I, Karran E, De Strooper B (2012) The toxic Aβ oligomer and Alzheimer’s disease: an emperor in need of clothes. Nat Neurosci 15(3):349–357
Berridge MJ (2017) Vitamin D deficiency accelerates ageing and age-related diseases: a novel hypothesis. J Physiol 595(22):6825–6836
Bhutia SK (2022) Vitamin D in autophagy signaling for health and diseases: insights on potential mechanisms and future perspectives. J Nutr Biochem 99:108841
Bishop NA, Lu T, Yankner BA (2010) Neural mechanisms of ageing and cognitive decline. Nature 464(7288):529–535
Boya P, Reggiori F, Codogno P (2013) Emerging regulation and functions of autophagy. Nat Cell Biol 15(7):713–720
Christakos S, Dhawan P, Liu Y, Peng X, Porta A (2003) New insights into the mechanisms of vitamin D action. J Cell Biochem 88(4):695–705
Christakos S, Dhawan P, Verstuyf A, Verlinden L, Carmeliet G (2016) Vitamin D: metabolism, molecular mechanism of action, and pleiotropic effects. Physiol Rev 96(1):365–408
Glatigny M, Moriceau S, Rivagorda M, Ramos-Brossier M, Nascimbeni AC, Lante F, Shanley MR, Boudarene N, Rousseaud A, Friedman AK et al (2019) Autophagy is required for memory formation and reverses age-related memory decline. Curr Biol 29(3):435–48.e8
Gulen MF, Samson N, Keller A, Schwabenland M, Liu C, Glück S, Thacker VV, Favre L, Mangeat B, Kroese LJ et al (2023) Cgas-sting drives ageing-related inflammation and neurodegeneration. Nature 620(7973):374–380
Janelidze S, Mattsson N, Palmqvist S, Smith R, Beach TG, Serrano GE, Chai X, Proctor NK, Eichenlaub U, Zetterberg H et al (2020) Plasma P-Tau181 in Alzheimer’s disease: relationship to other biomarkers, differential diagnosis, neuropathology and longitudinal progression to Alzheimer’s dementia. Nat Med 26(3):379–386
Jia J, Hu J, Huo X, Miao R, Zhang Y, Ma F (2019) Effects of vitamin D supplementation on cognitive function and blood Aβ-related biomarkers in older adults with Alzheimer’s disease: a randomised, double-blind, placebo-controlled trial. J Neurol Neurosurg Psychiatry 90(12):1347–1352
Kaushal A, Wani WY, Bal A, Gill KD, Kaur J (2019) Okadaic acid and hypoxia induced dementia model of Alzheimer’s type in rats. Neurotox Res 35(3):621–634
Khorram Khorshid HR, Gozalpour E, Saliminejad K, Karimloo M, Ohadi M, Kamali K (2013) Vitamin D receptor (Vdr) polymorphisms and late-onset Alzheimer’s disease: an association study. Iran J Public Health 42(11):1253–1258
Kitazawa M, Oddo S, Yamasaki TR, Green KN, LaFerla FM (2005) Lipopolysaccharide-induced inflammation exacerbates tau pathology by a cyclin-dependent kinase 5-mediated pathway in a transgenic model of Alzheimer’s disease. J Neurosci 25(39):8843–8853
Kitazawa M, Cheng D, Tsukamoto MR, Koike MA, Wes PD, Vasilevko V, Cribbs DH, LaFerla FM (2011) Blocking Il-1 signaling rescues cognition, attenuates tau pathology, and restores neuronal β-catenin pathway function in an Alzheimer’s disease model. J Immunol 187(12):6539–6549
Ktistakis NT, Tooze SA (2016) Digesting the expanding mechanisms of autophagy. Trends Cell Biol 26(8):624–635
Kulkarni A, Chen J, Maday S (2018) Neuronal autophagy and intercellular regulation of homeostasis in the brain. Curr Opin Neurobiol 51:29–36
Leuner B, Gould E (2010) Structural plasticity and hippocampal function. Annu Rev Psychol 61(111–40):c1-3
Li A, Yi B, Han H, Yang S, Hu Z, Zheng L, Wang J, Liao Q, Zhang H (2022) Vitamin D-Vdr (vitamin D receptor) regulates defective autophagy in renal tubular epithelial cell in streptozotocin-induced diabetic mice via the Ampk pathway. Autophagy 18(4):877–890
Liu D, Meng X, Tian Q, Cao W, Fan X, Wu L, Song M, Meng Q, Wang W, Wang Y (2022) Vitamin D and multiple health outcomes: an umbrella review of observational studies, randomized controlled trials, and mendelian randomization studies. Adv Nutr 13(4):1044–1062
Lu R, Shang M, Zhang YG, Jiao Y, Xia Y, Garrett S, Bakke D, Bäuerl C, Martinez GP, Kim CH et al (2020) Lactic acid bacteria isolated from Korean kimchi activate the vitamin D receptor-autophagy signaling pathways. Inflamm Bowel Dis 26(8):1199–1211
Martini AC, Gomez-Arboledas A, Forner S, Rodriguez-Ortiz CJ, McQuade A, Danhash E, Phan J, Javonillo D, Ha JV, Tram M et al (2019) Amyloid-beta impairs Tom1-mediated Il-1r1 signaling. Proc Natl Acad Sci U S A 116(42):21198–21206
Mattsson-Carlgren N, Janelidze S, Bateman RJ, Smith R, Stomrud E, Serrano GE, Reiman EM, Palmqvist S, Dage JL, Beach TG et al (2021) Soluble P-Tau217 reflects amyloid and tau pathology and mediates the association of amyloid with tau. EMBO Mol Med 13(6):e14022
Milà-Alomà M, Ashton NJ, Shekari M, Salvadó G, Ortiz-Romero P, Montoliu-Gaya L, Benedet AL, Karikari TK, Lantero-Rodriguez J, Vanmechelen E et al (2022) Plasma P-Tau231 and P-Tau217 as state markers of amyloid-β pathology in preclinical Alzheimer’s disease. Nat Med 28(9):1797–1801
Mizushima N, Komatsu M (2011) Autophagy: renovation of cells and tissues. Cell 147(4):728–741
Mizushima N, Yoshimori T, Ohsumi Y (2011) The role of Atg proteins in autophagosome formation. Annu Rev Cell Dev Biol 27:107–132
Morello M, Landel V, Lacassagne E, Baranger K, Annweiler C, Féron F, Millet P (2018) Vitamin d improves neurogenesis and cognition in a mouse model of Alzheimer’s disease. Mol Neurobiol 55(8):6463–6479
Morris R (1984) Developments of a water-maze procedure for studying spatial learning in the rat. J Neurosci Methods 11(1):47–60
Newcombe EA, Camats-Perna J, Silva ML, Valmas N, Huat TJ, Medeiros R (2018) Inflammation: the link between comorbidities, genetics, and Alzheimer’s disease. J Neuroinflammation 15(1):276
Pan Y, Zhang Y, Liu N, Lu W, Yang J, Li Y, Liu Z, Wei Y, Lou Y, Kong J (2021) Vitamin D attenuates Alzheimer-like pathology induced by okadaic acid. ACS Chem Neurosci 12(8):1343–1350
Patel P, Shah J (2022) Vitamin D(3) supplementation ameliorates cognitive impairment and alters neurodegenerative and inflammatory markers in scopolamine induced rat model. Metab Brain Dis 37(8):2653–2667
Phillips EA, Hendricks N, Bucher M, Maloyan A (2022) Vitamin D supplementation improves mitochondrial function and reduces inflammation in placentae of obese women. Front Endocrinol (lausanne) 13:893848
Rajasekar N, Dwivedi S, Tota SK, Kamat PK, Hanif K, Nath C, Shukla R (2013) Neuroprotective effect of curcumin on okadaic acid induced memory impairment in mice. Eur J Pharmacol 715(1–3):381–394
Sakono M, Zako T (2010) Amyloid oligomers: formation and toxicity of abeta oligomers. Febs J 277(6):1348–1358
Shehata M, Matsumura H, Okubo-Suzuki R, Ohkawa N, Inokuchi K (2012) Neuronal stimulation induces autophagy in hippocampal neurons that is involved in ampa receptor degradation after chemical long-term depression. J Neurosci 32(30):10413–10422
Shen W, Ganetzky B (2009) Autophagy promotes synapse development in Drosophila. J Cell Biol 187(1):71–79
Shi L, Xiao C, Zhang Y, Xia Y, Zha H, Zhu J, Song Z (2022) Vitamin D/vitamin D receptor/Atg16l1 axis maintains podocyte autophagy and survival in diabetic kidney disease. Ren Fail 44(1):694–705
Su H, Liu N, Zhang Y, Kong J (2021) Vitamin D/Vdr regulates peripheral energy homeostasis via central renin-angiotensin system. J Adv Res 33:69–80
Wang TT, Tavera-Mendoza LE, Laperriere D, Libby E, MacLeod NB, Nagai Y, Bourdeau V, Konstorum A, Lallemant B, Zhang R et al (2005) Large-scale in silico and microarray-based identification of direct 1,25-dihydroxyvitamin D3 target genes. Mol Endocrinol 19(11):2685–2695
Wang X, Zhou X, Li G, Zhang Y, Wu Y, Song W (2017a) Modifications and trafficking of app in the pathogenesis of Alzheimer’s disease. Front Mol Neurosci 10:294
Wang YC, Lauwers E, Verstreken P (2017b) Presynaptic protein homeostasis and neuronal function. Curr Opin Genet Dev 44:38–46
Wang J, Guo MN, Liu ZZ, Ma SF, Liu WJ, Qian JJ, Zhang WN (2021) Pgc-1α reduces amyloid-Β deposition in Alzheimer’s disease: effect of increased Vdr expression. Neurosci Lett 744:135598
Wang W, Li Y, Meng X (2023) Vitamin D and neurodegenerative diseases. Heliyon 9(1):e12877
Wu TY, Zhao LX, Zhang YH, Fan YG (2022) Activation of vitamin D receptor inhibits tau phosphorylation is associated with reduction of iron accumulation in App/Ps1 transgenic mice. Neurochem Int 153:105260
Xie J, Van Hoecke L, Vandenbroucke RE (2021) The impact of systemic inflammation on Alzheimer’s disease pathology. Front Immunol 12:796867
Yang Z, Klionsky DJ (2010) Eaten alive: a history of macroautophagy. Nat Cell Biol 12(9):814–822
Yang J, Zhang Y, Pan Y, Sun C, Liu Z, Liu N, Fu Y, Li X, Li Y, Kong J (2021) The protective effect of 1,25(Oh)(2)D(3) on myocardial function is mediated via sirtuin 3-regulated fatty acid metabolism. Front Cell Dev Biol 9:627135
Zhang H, Wang X, Xu P, Ji X, Chi T, Liu P, Zou L (2020) Tolfenamic acid inhibits Gsk-3β and Pp2a mediated tau hyperphosphorylation in Alzheimer’s disease models. J Physiol Sci 70(1):29
Zhang H, Wei W, Zhao M, Ma L, Jiang X, Pei H, Cao Y, Li H (2021a) Interaction between Aβ and tau in the pathogenesis of Alzheimer’s disease. Int J Biol Sci 17(9):2181–2192
Zhang Z, Yang X, Song YQ, Tu J (2021b) Autophagy in Alzheimer’s disease pathogenesis: therapeutic potential and future perspectives. Ageing Res Rev 72:101464
Zhou A, Hyppönen E (2023) Vitamin D deficiency and C-reactive protein: a bidirectional mendelian randomization study. Int J Epidemiol 52(1):260–271
Acknowledgements
None.
Funding
We are very grateful to the Yichang Central People’s Hospital for its financial support for this project.
Author information
Authors and Affiliations
Contributions
Zhixiong Huang contributed to the conception of the study; Zhixiong Huang, Wei Ang, Hefei Huang, Yanyan wang performed the experiment; Zhixiong Huang and Wei Ang contributed significantly to analysis and manuscript preparation; Zhixiong Huang performed the data analyses and wrote the manuscript; Wei Ang, Hefei Huang, Yanyan Wang helped perform the analysis with constructive discussions. Zhixiong Huang reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Zhixiong Huang, Wei Ang, Hefei Huang, and Yanyan Wang declare that they have no conflict of interest.
Ethical approval
All the experimental procedures were performed in accordance with the ethical standards stipulated by the International Committee of Medical Journal Editors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, Z., Ang, W., Huang, H. et al. VD/VDR-mediated ATG16L1 activation reduces Alzheimer’s disease-like pathology and cognitive decline. Mol. Cell. Toxicol. (2024). https://doi.org/10.1007/s13273-024-00429-7
Accepted:
Published:
DOI: https://doi.org/10.1007/s13273-024-00429-7