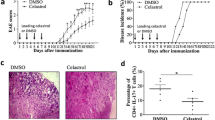
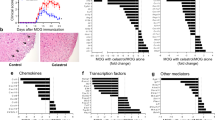
Abstract
Multiple sclerosis (MS) is an inflammatory demyelinating disease of the central nervous system (CNS) mediated by immune cells, in which auto-reactive CD4+ T cells have been implicated as a major driver in the pathogenesis of the disease. In this study, we aimed to investigate whether the artemisinin derivative TPN10475 could alleviate experimental autoimmune encephalomyelitis (EAE), a commonly used animal model of MS and its possible mechanisms. TPN10475 effectively resisted the reduction of TGF-β signal transduction induced by TCR stimulation, suppressed the activation and function of effector CD4+ T cells in vitro, and restricted the differentiation of pathogenic Th1 and Th17 cells. It was also found to negatively regulate the inflammatory response in EAE by reducing the peripheral activation drive of auto-reactive helper T lymphocytes, inhibiting the migration of inflammatory cells into the CNS to attenuate EAE. The above results suggested that the upregulation of TGF-β signal transduction may provide new ideas for the study of MS pathogenesis and have positive implications for the development of drugs for the treatment of autoimmune diseases.
Graphical Abstract
TPN10475 promotes TGF-β signaling under TCR stimulation, restricts the proliferation and activation of effector CD4+ T cells, inhibits the differentiation of pathogenic Th1 and Th17 cells, negatively regulates the inflammatory response and suppresses the migration of inflammatory cells to the CNS, thereby alleviating EAE. The figure was created with Biorender.com (Agreement number: ME25MXMB52).










Similar content being viewed by others
Data Availability
The material and data used to support the findings of the current study are available from the corresponding author on reasonable request.
References
Bierie B, Chung CH, Parker JS, Stover DG, Cheng N, Chytil A, Aakre M, Shyr Y, Moses HL (2009) Abrogation of TGF-beta signaling enhances chemokine production and correlates with prognosis in human breast cancer. J Clin Invest 119:1571–1582
Bright JJ, Sriram S (1998) TGF-beta inhibits IL-12-induced activation of Jak-STAT pathway in T lymphocytes. J Immunol (Baltimore Md: 1950) 161:1772–1777
Cekanaviciute E, Dietrich HK, Axtell RC, Williams AM, Egusquiza R, Wai KM, Koshy AA, Buckwalter MS (2014) Astrocytic TGF-beta signaling limits inflammation and reduces neuronal damage during central nervous system Toxoplasma infection. J Immunol 193:139–149
Chapman NM, Boothby MR, Chi H (2020) Metabolic coordination of T cell quiescence and activation. Nat Rev Immunol 20:55–70
Codarri L, Gyülvészi G, Tosevski V, Hesske L, Fontana A, Magnenat L, Suter T, Becher B (2011) RORγt drives production of the cytokine GM-CSF in helper T cells, which is essential for the effector phase of autoimmune neuroinflammation. Nat Immunol 12:560–567
Constantinescu CS, Farooqi N, O’Brien K, Gran B (2011) Experimental autoimmune encephalomyelitis (EAE) as a model for multiple sclerosis (MS). Br J Pharmacol 164:1079–1106
Correale J, Gaitan MI, Ysrraelit MC, Fiol MP (2017) Progressive multiple sclerosis: from pathogenic mechanisms to treatment. Brain 140:527–546
Dendrou CA, Fugger L, Friese MA (2015) Immunopathology of multiple sclerosis. Nat Rev Immunol 15:545–558
Derynck R, Budi EH (2019) Specificity, versatility, and control of TGF-beta family signaling. Sci Signal 12:eaav5183
Dittel BN (2008) CD4 T cells: balancing the coming and going of autoimmune-mediated inflammation in the CNS. Brain Behav Immun 22:421–430
Dobson R, Giovannoni G (2019) Multiple sclerosis - a review. Eur J Neurol 26:27–40
Du C, Liu C, Kang J, Zhao G, Ye Z, Huang S, Li Z, Wu Z, Pei G (2009) MicroRNA miR-326 regulates TH-17 differentiation and is associated with the pathogenesis of multiple sclerosis. Nat Immunol 10:1252–1259
Duscha A et al (2020) Propionic Acid shapes the multiple sclerosis Disease Course by an Immunomodulatory mechanism. Cell 180:1067-1080e1016
Feng X, Li X, Liu N, Hou N, Sun X, Liu Y (2022) Glutaminolysis and CD4(+) T-cell metabolism in autoimmunity: from pathogenesis to therapy prospects. Front Immunol 13:986847
Ghoreschi K et al (2010) Generation of pathogenic T(H)17 cells in the absence of TGF-beta signalling. Nature 467:967–971
Gorelik L (2002) Mechanism of transforming growth factor β–induced inhibition of T helper type 1 differentiation. J Exp Med 195:1499–1505
Heng AHS, Han CW, Abbott C, McColl SR, Comerford I (2022) Chemokine-Driven Migration of pro-inflammatory CD4(+) T cells in CNS autoimmune disease. Front Immunol 13:817473
Hohlfeld R, Dornmair K, Meinl E, Wekerle H (2016) The search for the target antigens of multiple sclerosis, part 1: autoreactive CD4 + T lymphocytes as pathogenic effectors and therapeutic targets. Lancet Neurol 15:198–209
Hoppmann N, Graetz C, Paterka M, Poisa-Beiro L, Larochelle C, Hasan M, Lill CM, Zipp F, Siffrin V (2015) New candidates for CD4 T cell pathogenicity in experimental neuroinflammation and multiple sclerosis. Brain 138:902–917
Kamali AN, Noorbakhsh SM, Hamedifar H, Jadidi-Niaragh F, Yazdani R, Bautista JM, Azizi G (2019) A role for Th1-like Th17 cells in the pathogenesis of inflammatory and autoimmune disorders. Mol Immunol 105:107–115
Kipp M, Nyamoya S, Hochstrasser T, Amor S (2017) Multiple sclerosis animal models: a clinical and histopathological perspective. Brain Pathol 27:123–137
Krishnarajah S, Becher B (2022) T(H) cells and cytokines in Encephalitogenic disorders. Front Immunol 13:822919
Lee PW, Severin ME, Lovett-Racke AE (2017) TGF-beta regulation of encephalitogenic and regulatory T cells in multiple sclerosis. Eur J Immunol 47:446–453
Legroux L, Arbour N (2015) Multiple sclerosis and T lymphocytes: an Entangled Story. J Neuroimmune Pharmacol 10:528–546
Li X, Li TT, Zhang XH, Hou LF, Yang XQ, Zhu FH, Tang W, Zuo JP (2013) Artemisinin analogue SM934 ameliorates murine experimental autoimmune encephalomyelitis through enhancing the expansion and functions of regulatory T cell. PLoS ONE 8:e74108
Liu G, Jiang X, Han M, Lv J, Zhuang W, Xie L, Zhang Y, Wang C, Saimaier K, Yang J, Shen J, Li N, Du C (2022) Artemisinin derivative TPN10466 suppresses immune cell migration and Th1/Th17 differentiation to ameliorate disease severity in experimental autoimmune encephalomyelitis. Cell Immunol 373:104500
Luckheeram RV, Zhou R, Verma AD, Xia B (2012) CD4(+)T cells: differentiation and functions. Clin Dev Immunol 2012:925135
Lyons AB, Blake SJ, Doherty KV (2013) Flow cytometric analysis of cell division by dilution of CFSE and related dyes. Curr Protoc Cytom Chap 9:9 (11 11–19 11 12)
Mahad DH, Trapp BD, Lassmann H (2015) Pathological mechanisms in progressive multiple sclerosis. Lancet Neurol 14:183–193
Oh SA, Li MO (2013) TGF-beta: guardian of T cell function. J Immunol 191:3973–3979
Olek MJ (2021) Multiple sclerosis. Ann Intern Med 174:ITC81–ITC96
Sun X, Cui Y, Feng H, Liu H, Liu X (2019) TGF-beta signaling controls Foxp3 methylation and T reg cell differentiation by modulating Uhrf1 activity. J Exp Med 216:2819–2837
Takeuchi H (2014) Midkine and multiple sclerosis. Br J Pharmacol 171:931–935
Takimoto T, Wakabayashi Y, Sekiya T, Inoue N, Morita R, Ichiyama K, Takahashi R, Asakawa M, Muto G, Mori T, Hasegawa E, Saika S, Hara T, Nomura M, Yoshimura A (2010) Smad2 and Smad3 are redundantly essential for the TGF-beta-mediated regulation of regulatory T plasticity and Th1 development. J Immunol 185:842–855
Tu E, Chia CPZ, Chen W, Zhang D, Park SA, Jin W, Wang D, Alegre ML, Zhang YE, Sun L, Chen W (2018) T cell receptor-regulated TGF-beta type I receptor expression determines T cell quiescence and activation. Immunity 48:745-759e746
Wingerchuk DM, Carter JL (2014) Multiple sclerosis: current and emerging disease-modifying therapies and treatment strategies. Mayo Clin Proc 89:225–240
Wu Y, Tang W, Zuo J (2016) Development of artemisinin drugs in the treatment of autoimmune diseases. Sci Bull 61:37–41
Xu J, Liu D, Niu H, Zhu G, Xu Y, Ye D, Li J, Zhang Q (2017) Resveratrol reverses doxorubicin resistance by inhibiting epithelial-mesenchymal transition (EMT) through modulating PTEN/Akt signaling pathway in gastric cancer. J Exp Clin Cancer Res 36:19
Zhu J, Yamane H, Paul WE (2010) Differentiation of effector CD4 T cell populations (*). Annu Rev Immunol 28:445–489
Funding
This project was supported by grants from the National Natural Science Foundation of China (32070768, 32270754).
Author information
Authors and Affiliations
Contributions
Chun Wang performed the experiments and wrote the manuscript. Xiangrui Jiang and Changsheng Du conceived the study and designed the experiment. Jie Lv, Wei Zhuang, and Ling Xie helped with data collection. Guangyu Liu, Kaidireya Saimaier, and Sanxing Han assisted in the analysis of the data. Changjie Shi, Qiuhong Hua, and Ru Zhang provided software and resources. Changsheng Du extensively and critically reviewed the manuscript. All authors participated in the study and supported the publication of the final version.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
All experiments were conducted by Ethical Principles in Animal Experimentation and were approved by the Animal Research Ethics Committee of Tongji University (Ethics number: TJAB03320103).
Consent for Publication
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, C., Jiang, X., Lv, J. et al. TPN10475 Constrains Effector T Lymphocytes Activation and Attenuates Experimental Autoimmune Encephalomyelitis Pathogenesis by Facilitating TGF-β Signal Transduction. J Neuroimmune Pharmacol 19, 6 (2024). https://doi.org/10.1007/s11481-024-10109-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11481-024-10109-x