Abstract
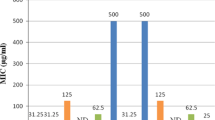
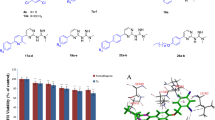
In this work, a novel series of racemic Schiff bases bearing the biologically active [1,2,4]triazolo[5,1-b]quinazoline scaffold has been synthesized in good to excellent yields, and fully characterized by FTIR, 1H NMR and 13C NMR, as well as elemental analyses. The in vitro antimalarial activities of these derivatives have been evaluated against Plasmodium falciparum (P. falciparum) K1 strain and exhibited IC50 values ranged from 15.4 to 44.4 µM. Among them, 4e, 4j, 4 l, and 4f lead to the best results with IC50 of 15.4, 15.8, 16.8, and 17.3 µM, respectively. The observed activity was further substantiated by docking study on wild-type P. falciparum dihydrofolate reductase-thymidylate synthase (pf-DHFR-TS). According to docking results, (S) stereoisomer of these synthesized compounds has the best score among them. The (S) stereoisomer shows probably more stable binding with the active site of the enzyme and can be considered as a candidate for stereoselective synthesis and biological evaluation in future studies. Moreover, among four synthetic derivatives that showed prominent antimalarial activity (4e, 4j, 4 l and 4f), 4e has the minimum Gibbs binding energy, and it is consistent with the biological activity. The new compounds were also accomplished against both Gram-positive and Gram-negative bacteria, but no promising effect was observed, even at concentrations up to 400 µg/mL.





Similar content being viewed by others
References
E.A. Ashley, A.P. Phyo, Drugs 78, 861 (2018)
K. Haldar, S. Bhattacharjee, I. Safeukui, Nat. Rev. Microbiol. 16, 156 (2018)
S. Thota, R. Yerra, Curr. Protein Pept. Sci. 17, 275 (2016)
T.M. Belete, Drug Des. Devel. Ther. 14, 3875 (2020)
M. Mishra, V.K. Mishra, V. Kashaw, A.K. Iyer, S.K. Kashaw, Eur. J. Med. Chem. 125, 1300 (2017)
P. Guyant, V. Corbel, P.J. Guérin, A. Lautissier, F. Nosten, S. Boyer, M. Coosemans, A.M. Dondorp, V. Sinou, S. Yeung, N. White, Malar. J. 14, 279 (2015)
J. Achan, J. Mwesigwa, C.P. Edwin, U. D’alessandro, Expert. Rev. Clin. Pharmacol. 11, 61 (2018)
T.N.C. Wells, R.H. van Huijsduijnen, W.C. Van Voorhis, Nat. Rev. Drug Discov. 14, 424 (2015)
H. Antony, S. Parija, Trop. Parasitol. 6, 30 (2016)
M. Lödige, L. Hiersch, Int. J. Med. Chem. 2015, 458319 (2015)
S. Alven, B. Aderibigbe, Molecules 24, 3601 (2019)
L.-H. Chen, T.-W. Chung, B.D. Narhe, C.-M. Sun, A.C.S. Comb, Sci. 18, 162 (2016)
N.A.A. El-Fatah, A.F. Darweesh, A.A. Mohamed, I.A. Abdelhamid, A.H.M. Elwahy, Monatshefte Für Chemie - Chem. Mon. 148, 2107 (2017)
M. Driowya, J. Leclercq, V. Verones, A. Barczyk, M. Lecoeur, N. Renault, N. Flouquet, A. Ghinet, P. Berthelot, N. Lebegue, Eur. J. Med. Chem. 115, 393 (2016)
F.E. Held, A.A. Guryev, T. Fröhlich, F. Hampel, A. Kahnt, C. Hutterer, M. Steingruber, H. Bahsi, C. von Bojničić-Kninski, D.S. Mattes, T.C. Foertsch, A. Nesterov-Mueller, M. Marschall, S.B. Tsogoeva, Nat. Commun. 8, 15071 (2017)
D. Xie, J. Shi, A. Zhang, Z. Lei, G. Zu, Y. Fu, X. Gan, L. Yin, B. Song, D. Hu, Bioorg. Chem. 80, 433 (2018)
R. Luo, Z. Wang, D. Luo, Y. Qin, C. Zhao, D. Yang, T. Lu, Z. Zhou, Z. Huang, J. Enzyme Inhib. Med. Chem. 36, 2170 (2021)
T. Wu, Q. Qin, R. Lv, N. Liu, W. Yin, C. Hao, Y. Sun, C. Zhang, Y. Sun, D. Zhao, M. Cheng, Eur. J. Med. Chem. 238, 114451 (2022)
F. Hakim, R. Salfi, D. Bhikshapathi, A. Khan, Anticancer. Agents Med. Chem. 22, 926 (2022)
Z.Y. Ibrahim, A. Uzairu, G.A. Shallangwa, S.E. Abechi, S. Isyaku, S.N. Appl, Sci. 3, 767 (2021)
S.H. Megahed, S. Rasheed, J. Herrmann, E.M. El-Hossary, Y.I. El-Shabrawy, A.H. Abadi, M. Engel, R. Müller, M. Abdel-Halim, M.M. Hamed, Bioorg. Med. Chem. Lett. 59, 128531 (2022)
I. Nosulenko, G. Berest, D. Skoryna, O. Voskoboinik, S. Kovalenko, J. Res. Pharm. 26(1), 1045 (2022)
O.F. Ibrahim, E.A. Bakhite, S.A.M. Metwally, Y.A. El-Ossaily, H.H.M. Abdu-Allah, E.A. Al-Taifi, M. Kandel, Russ. J. Bioorganic Chem. 47, 918 (2021)
M.M. Gineinah, M.A. El-Sherbeny, M.N. Nasr, A.R. Maarouf, Arch. Pharm. (Weinheim). 335, 556 (2002)
H.A. Abuelizz, R. El Dib, M. Marzouk, E.-H. Anouar, Y.A. Maklad, H.N. Attia, R. Al-Salahi, Molecules 22, 1094 (2017)
C.-B. Zhang, C.-W. Yang, X.-Q. Deng, Z.-S. Quan, Med. Chem. Res. 21, 3294 (2012)
K.A. Shaikh, U.N. Chaudhar, A.C.T.A. Chem, IASI 29, 15 (2021)
M.R. Mousavi, M.T. Maghsoodlou, N. Hazeri, S.M. Habibi-Khorassani, J. Iran. Chem. Soc. 12, 1419 (2015)
N. Seyyedi, F. Shirini, M.S.N. Langarudi, S. Jashnani, J. Iran. Chem. Soc. 14, 1859 (2017)
D.M. Patel, R.M. Vala, M.G. Sharma, D.P. Rajani, H.M. Patel, ChemistrySelect 4, 1031 (2019)
R.D. Patil, S. Adimurthy, Asian J. Org. Chem. 2, 726 (2013)
E. Raczuk, B. Dmochowska, J. Samaszko-Fiertek, J. Madaj, Molecules 27, 787 (2022)
A. Jarrahpour, D. Khalili, E. De Clercq, C. Salmi, J. Brunel, Molecules 12, 1720 (2007)
Z. Zebbiche, S. Tekin, H. Küçükbay, F. Yüksel, B. Boumoud, Arch. Pharm. (Weinheim). 354, 2000377 (2021)
G. Sui, D. Xu, T. Luo, H. Guo, G. Sheng, D. Yin, L. Ren, H. Hao, W. Zhou, Bioorg. Med. Chem. Lett. 30, 126774 (2020)
S. Celik, F. Ozkok, A.E. Ozel, E. Cakir, S. Akyuz, J. Mol. Struct. 1236, 130288 (2021)
A.S. Ressurreição, D. Gonçalves, A.R. Sitoe, I.S. Albuquerque, J. Gut, A. Góis, L.M. Gonçalves, M.R. Bronze, T. Hanscheid, G.A. Biagini, P.J. Rosenthal, M. Prudêncio, P. O’Neill, M.M. Mota, F. Lopes, R. Moreira, J. Med. Chem. 56, 7679 (2013)
E. Riazimontazer, H. Sadeghpour, H. Nadri, A. Sakhteman, T. Tüylü Küçükkılınç, R. Miri, N. Edraki, Bioorg. Chem. 89, 103006 (2019)
S. Murtaza, N. Kausar, U. Arshad, S. Ahmed, A. Tatheer, J. Najeeb, A. Tawab, J. Mol. Struct. 1264, 133271 (2022)
S. Kaya, S. Erkan, D. Karakaş, Spectrochim Acta Part A Mol. Biomol. Spectrosc. 244, 118829 (2021)
D.C. Zimmermann-Franco, B. Esteves, L.M. Lacerda, I. de O. Souza, J. A. dos Santos, N. de C. C. Pinto, E. Scio, A. D. da Silva, and G. C. Macedo, Bioorg. Med. Chem. 26, 4898 (2018)
J. Lu, C. Li, Y.-F. Chai, D.-Y. Yang, C.-R. Sun, Bioorg. Med. Chem. Lett. 22, 5744 (2012)
M. Bashiri, A. Jarrahpour, B. Rastegari, A. Iraji, C. Irajie, Z. Amirghofran, S. Malek-Hosseini, M. Motamedifar, M. Haddadi, K. Zomorodian, Z. Zareshahrabadi, E. Turos, Monatsh. Chem. 151, 821 (2020)
A. Jarrahpour, J. Sheikh, I. El Mounsi, H. Juneja, T. Ben Hadda, Med. Chem. Res. 22, 1203 (2013)
A. Jarrahpour, J. Fathi, M. Mimouni, T. Ben Hadda, J. Sheikh, Z. Chohan, A. Parvez, Med. Chem. Res. 21, 1984 (2012)
A. Jarrahpour, P. Shirvani, H. Sharghi, M. Aberi, V. Sinou, C. Latour, J.M. Brunel, Med. Chem. Res. 24, 4105 (2015)
W. Trager, J.B. Jensen, Science. 193, 673 (1976)
C. Lambros, J.P. Vanderberg, J. Parasitol. 65, 418 (1979)
R. Nakweti, V. Sinou, S. Ndiku, F. Sabot, C. Franche, Eur. Med. Plants 18, 1 (2017)
H. Le Nagard, C. Vincent, F. Mentré, J. Le Bras, Comput. Methods Programs Biomed. 104, 10 (2011)
Acknowledgements
Funding for this research was generously provided by the Shiraz University Research Council (Grant No. 93-GR-SC-23).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jowkar, Z., Aboonajmi, J., Heiran, R. et al. Synthesis, docking and biological evaluation of some novel [1,2,4] triazolo [5,1-b] quinazoline Schiff base derivatives. J IRAN CHEM SOC 21, 1055–1067 (2024). https://doi.org/10.1007/s13738-024-02975-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-024-02975-2