Abstract
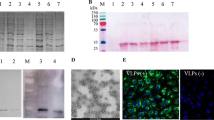
Porcine circovirus type 4 (PCV4), first identified in 2019 as a newly emerging pathogen, has been found in several provinces of China, as well as in Korea and Thailand. Since PCV4 is not included in immunization programs, epidemiological investigations should be conducted for detection of anti-PCV4 antibodies. Virus-like particles (VLPs) are frequently used for serological analysis of pathogen infections. However, there have been no reports on using PCV4 VLPs for serological investigation of PCV4 infection. In this study, we generated self-assembled PCV4 VLPs using an E. coli expression system, purified them using a two-step process, and used them to develop an indirect ELISA. This ELISA method was found to be highly specific, sensitive, and repeatable, making it suitable for PCV4 antibody detection in serum samples. Finally, the ELISA was used to analyze 422 serum samples collected from across several regions in China, 134 of which tested positive. Thus, the PCV4-VLP-based ELISA can effectively detect antibodies against PCV4 in serum samples, making it a useful tool for PCV4 epidemiology.







Similar content being viewed by others
Data availability
The datasets generated and analyzed in the current study are available from the corresponding author upon reasonable request.
References
Allan GM, McNeilly F, Cassidy JP et al (1995) Pathogenesis of porcine circovirus; experimental infections of colostrum deprived piglets and examination of pig foetal material. Vet Microbiol 44:49–64. https://doi.org/10.1016/0378-1135(94)00136-K
Meng X-J (2012) Spread like a wildfire—the omnipresence of porcine circovirus type 2 (PCV2) and its ever-expanding association with diseases in pigs. Virus Res 164:1–3. https://doi.org/10.1016/j.virusres.2011.12.005
Allan G, Krakowka S, Ellis J, Charreyre C (2012) Discovery and evolving history of two genetically related but phenotypically different viruses, porcine circoviruses 1 and 2. Virus Res 164:4–9. https://doi.org/10.1016/j.virusres.2011.09.013
Opriessnig T, Karuppannan AK, Castro AMMG, Xiao C-T (2020) Porcine circoviruses: current status, knowledge gaps and challenges. Virus Res 286:198044. https://doi.org/10.1016/j.virusres.2020.198044
Klaumann F, Correa-Fiz F, Franzo G et al (2018) Current knowledge on porcine circovirus 3 (PCV-3): a novel virus with a yet unknown impact on the swine industry. Front Vet Sci 5:315. https://doi.org/10.3389/fvets.2018.00315
Zhang H-H, Hu W-Q, Li J-Y et al (2020) Novel circovirus species identified in farmed pigs designated as Porcine circovirus 4, Hunan province, China. Transbound Emerg Dis 67:1057–1061. https://doi.org/10.1111/tbed.13446
Nguyen V-G, Do H-Q, Huynh T-M-L et al (2022) Molecular-based detection, genetic characterization and phylogenetic analysis of porcine circovirus 4 from Korean domestic swine farms. Transbound Emerg Dis 69:538–548. https://doi.org/10.1111/tbed.14017
Sirisereewan C, Nguyen TC, Piewbang C et al (2023) Molecular detection and genetic characterization of porcine circovirus 4 (PCV4) in Thailand during 2019–2020. Sci Rep 13:1–10. https://doi.org/10.1038/s41598-023-32382-1
Niu G, Zhang X, Ji W et al (2022) Porcine circovirus 4 rescued from an infectious clone is replicable and pathogenic in vivo. Transbound Emerg Dis 69:e1632–e1641. https://doi.org/10.1111/tbed.14498
Wang D, Mai J, Yang Y et al (2022) Current knowledge on epidemiology and evolution of novel porcine circovirus 4. Vet Res 53:38. https://doi.org/10.1186/s13567-022-01053-w
Zhang D, Bai C, Ge K et al (2020) Establishment of an SYBR Green-based real-time PCR assay for porcine circovirus type 4 detection. J Virol Methods 285:113963. https://doi.org/10.1016/j.jviromet.2020.113963
Chen N, Xiao Y, Li X et al (2021) Development and application of a quadruplex real-time PCR assay for differential detection of porcine circoviruses (PCV1 to PCV4) in Jiangsu province of China from 2016 to 2020. Transbound Emerg Dis 68:1615–1624. https://doi.org/10.1111/tbed.13833
Sun W, Du Q, Han Z et al (2021) Detection and genetic characterization of porcine circovirus 4 (PCV4) in Guangxi, China. Gene 773:145384. https://doi.org/10.1016/j.gene.2020.145384
Ge M, Hu W-Q, Ning K-M et al (2021) The seroprevalence of the newly identified porcine circovirus type 4 in China investigated by an enzymed-linked immunosorbent assay. Transbound Emerg Dis 68:2910–2914. https://doi.org/10.1111/tbed.14184
Hu X, Ding Z, Li Y et al (2022) Serum investigation of antibodies against porcine circovirus 4 Rep and Cap protein in Jiangxi Province, China. Front Microbiol 13:944679. https://doi.org/10.3389/fmicb.2022.944679
Lian Z, Liu J, Liu P et al (2021) Development and application of an indirect ELISA for the detection of antibody to porcine circovirus 4 in pigs. Transbound Emerg Dis 68:2975–2979. https://doi.org/10.1111/tbed.14267
Zhang Y, Wang Z, Zhan Y et al (2016) Generation of E. coli-derived virus-like particles of porcine circovirus type 2 and their use in an indirect IgG enzyme-linked immunosorbent assay. Arch Virol 161:1485–1491. https://doi.org/10.1007/s00705-016-2816-9
Trible BR, Suddith AW, Kerrigan MA et al (2012) Recognition of the different structural forms of the capsid protein determines the outcome following infection with porcine circovirus type 2. J Virol 86:13508–13514. https://doi.org/10.1128/jvi.01763-12
Meng X-J (2013) Porcine circovirus type 2 (PCV2): pathogenesis and interaction with the immune system. Annu Rev Anim Biosci 1:43–64. https://doi.org/10.1146/annurev-animal-031412-103720
Hirschberg S, Bauer H, Kamhieh-Milz J et al (2022) SARS-CoV-2 virus-like particles (VLPs) specifically detect humoral immune reactions in an ELISA-based platform. Antibodies (Basel) 11:76. https://doi.org/10.3390/antib11040076
Kirnbauer R, Hubbert NL, Wheeler CM et al (1994) A virus-like particle enzyme-linked immunosorbent assay detects serum antibodies in a majority of women infected with human papillomavirus type 16. JNCI J Natl Cancer Inst 86:494–499. https://doi.org/10.1093/jnci/86.7.494
Du P, Brendle S, Milici J et al (2015) Comparisons of VLP-based ELISA, neutralization assays with native HPV, and neutralization assays with PsV in detecting HPV antibody responses in HIV-infected women. J AIDS Clin Res 6:433. https://doi.org/10.4172/2155-6113.1000433
Mali DN, Bondre VP (2022) Japanese encephalitis genotype I virus-like particles stably expressed in BHK-21 cells serves as potential antigen in JE IgM ELISA. Appl Microbiol Biotechnol 106:1945. https://doi.org/10.1007/s00253-022-11825-1
Purdy DE, Noga AJ, Chang G-JJ (2004) Noninfectious recombinant antigen for detection of St. Louis encephalitis virus-specific antibodies in serum by enzyme-linked immunosorbent assay. J Clin Microbiol 42:4709–4717. https://doi.org/10.1128/JCM.42.10.4709-4717.2004
Bai M, Wang R, Sun S et al (2021) Development and validation of a competitive ELISA based on virus-like particles of serotype Senecavirus A to detect serum antibodies. AMB Expr 11:7. https://doi.org/10.1186/s13568-020-01167-4
Nainys J, Lasickiene R, Petraityte-Burneikiene R et al (2014) Generation in yeast of recombinant virus-like particles of porcine circovirus type 2 capsid protein and their use for a serologic assay and development of monoclonal antibodies. BMC Biotechnol 14:100. https://doi.org/10.1186/s12896-014-0100-1
Wang Y, Wang G, Duan W-T et al (2020) Self-assembly into virus-like particles of the recombinant capsid protein of porcine circovirus type 3 and its application on antibodies detection. AMB Expr 10:3. https://doi.org/10.1186/s13568-019-0940-0
Xu T, Hou C-Y, Zhang Y-H et al (2022) Simultaneous detection and genetic characterization of porcine circovirus 2 and 4 in Henan province of China. Gene 808:145991. https://doi.org/10.1016/j.gene.2021.145991
Hou C-Y, Zhang L-H, Zhang Y-H et al (2022) Phylogenetic analysis of porcine circovirus 4 in Henan Province of China: a retrospective study from 2011 to 2021. Transbound Emerg Dis 69:1890–1901. https://doi.org/10.1111/tbed.14172
Blanchard P, Mahé D, Cariolet R et al (2003) Protection of swine against post-weaning multisystemic wasting syndrome (PMWS) by porcine circovirus type 2 (PCV2) proteins. Vaccine 21:4565–4575. https://doi.org/10.1016/S0264-410X(03)00503-6
Wang D, Mai J, Lei B et al (2021) Structure, antigenic properties, and highly efficient assembly of PCV4 capsid protein. Front Vet Sci 8:695466. https://doi.org/10.3389/fvets.2021.695466
Huang L, Sun Z, Xia D et al (2020) Neutralization mechanism of a monoclonal antibody targeting a porcine circovirus type 2 cap protein conformational epitope. J Virol 94:e0183619. https://doi.org/10.1128/jvi.01836-19
Li X, Chen S, Niu G et al (2022) Porcine circovirus type 4 strains circulating in China are relatively stable and have higher homology with mink circovirus than other porcine circovirus types. Int J Mol Sci 23:3288. https://doi.org/10.3390/ijms23063288
Wu H, Hou C, Wang Z et al (2022) First complete genomic sequence analysis of porcine circovirus type 4 (PCV4) in wild boars. Vet Microbiol 273:109547. https://doi.org/10.1016/j.vetmic.2022.109547
Xu T, Chen X-M, Fu Y et al (2022) Cross-species transmission of an emerging porcine circovirus (PCV4): First molecular detection and retrospective investigation in dairy cows. Vet Microbiol 273:109528. https://doi.org/10.1016/j.vetmic.2022.109528
Zhang L-H, Wang T-X, Fu P-F et al (2023) First molecular detection and genetic analysis of a novel porcine circovirus (porcine circovirus 4) in dogs in the world. Microbiol Spectrum 11:e0433322. https://doi.org/10.1128/spectrum.04333-22
Wang Y, Yan S, Ji Y et al (2022) First identification and phylogenetic analysis of porcine circovirus type 4 in fur animals in Hebei, China. Animals 12:3325. https://doi.org/10.3390/ani12233325
Funding
This study was supported by a grant from the Heilongjiang Provincial Key Research and Development Program (JD22A023).
Author information
Authors and Affiliations
Contributions
Y.T. and H.W. conceptualized the work and designed the studies; Z.F., M.S., and S.W. performed the experiments; Y.T. and S.W. analyzed the data; Z.F. and H.W. wrote the manuscript; and X.C. and T.A. revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Handling Editor: Roman Pogranichniy.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Zheng Fang and Yabin Tu both authors are co-first authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fang, Z., Tu, Y., Sun, M. et al. Generation of porcine circovirus type 4 virus-like particles and their use to detect serum antibodies. Arch Virol 169, 67 (2024). https://doi.org/10.1007/s00705-024-05997-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00705-024-05997-6