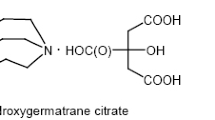
Abstract
Among germatranes, 1-germatranol hydrate is the most well-studied compound with a broad spectrum of biological activity. It is synthesized in one step by the reaction of tris(2-hydroxyethyl)amine with germanium dioxide in an aqueous medium without the use of organic solvents. Using this approach, new potentially biologically active compounds of germanium with hydroxyalkylamines and sulfonic and amino acids containing hydroxyalkyl groups were synthesized. The products were considered as new analogues of biologically active 1-germatranol hydrate. Their formation was confirmed by the results of elemental analysis, IR and NMR spectroscopy. In silico ADME and Pass analysis were used to assess the potential bioavailability and pharmacological activity profiles for new compounds. The results of in vitro study of the antiviral activity (influenza virus A/Aichi/2/68 (H3N2)) of the synthesized compounds are presented.

Similar content being viewed by others
Notes
Characteristics of IR bands: br is broadened; vs is very strong; s is strong, m is medium, w is weak.
REFERENCES
V. G. Rebrov and O. A. Gromova, Vitamins and Microelements (Moscow, 2003) [in Russian].
S. S. Karlov and G. S. Zaitseva, Chem. Heterocycl. Compd. 37, 1325 (2001). https://doi.org/10.1023/A:1017942932503
E. Lukevics and L. Ignatovich, Biological Activity of Organogermanium Compounds, PATAI’S Chemistry of Functional Groups, Ed. by Z. Rappoport (2009). https://doi.org/10.1002/9780470682531.pat0275
E. Lukevics, L. Ignatovich, T. Shul’ga, et al., J. Organomet. Chem. 659, 165 (2002).https://doi.org/10.1016/S0022-328X02.01723-0
R. T. Mertens, S. Parkin, and S. G. Awuah, Inorg. Chim. Acta 503, 119375 (2020). https://doi.org/10.1016/j.ica.2019.119375
E. N. Nikolaevskaya, P. G. Shangin, A. A. Starikova, et al., Inorg. Chim. Acta 495, 119007 (2019). https://doi.org/10.1016/j.ica.2019.119007
J. Pi, J. Zeng, J.-J. Luo, et al., Bioorg. Med. Chem. Lett. 23, 2902 (2013). https://doi.org/10.1016/j.bmcl.2013.03.061
A. A. Vishtorskaya, E. A. Saverina, V. M. Pechennikov, et al., J. Organomet. Chem. 858, 8 (2018). https://doi.org/10.1016/j.jorganchem.2018.01.004
T. Nabiyeva, B. Roufosse, M. Odachowski, et al., ACS Omega 6, 19252 (2021). https://doi.org/10.1021/acsomega.1c02665
A. V. Kadomtseva, G. M. Mochalov, and O. V. Kuzina, Russ. J. Org. Chem. 57, 879 (2021). https://doi.org/10.1134/S1070428021060026
L. G. Menchikov, A. A. Shestov, and A. V. Popov, Biochemistry (Moscow) 88, S1 (2023). https://doi.org/10.1134/S0006297923140018
M. Awais, A. Aizaz, A. Nazneen, et al., Prosthesis 4, 263 (2022). https://doi.org/10.3390/prosthesis4020026
J. M. Cho, J. Chae, S. R. Jeong, et al., PLoS One 15, e0240358 (2020). .https://doi.org/10.1371/journal.pone.0240358
L. G. Menchikov and M. A. Ignatenko, Pharm. Chem. J. 46, 635 (2013). https://doi.org/10.1007/s11094-013-0860-2
M. G. Voronkov, Z. A. Ovchinnikova, and V. P. Baryshok, Izv. AN SSSR, Ser. Khim. 4, 880 (1987).
V. P. Baryshok, K. A. Abzaeva, M. M. Rasulov, et al., Izv. VUZov, Prikl. Khim. Biotekhnol. 8, 153 (2018). https://doi.org/10.21285/2227-2925-2018-8-1-153-158
M. M. Rasulov, P. A. Storozhenko, T. A. Snisarenko, et al., Patent RF 2553986 C1, published June 20, 2015. Byull. Izobret. no. 17.
A. M. Shigarova, O. I. Grabelnych, V. P. Baryshok, et al., Appl. Biochem. Microbiol. 52, 429 (2016). https://doi.org/10.1134/S0003683816040153
A. P. Rachin, R. M. Rasulov, V. P. Baryshok, et al., Patent RF 2741229 C1, published January 22, 2021. Byull. Izobret. no. 3.
A. D. Isaev, S. A. Bashkirova, and K. V. Pavlov, Patent RF 2272624 C1, published March 27, 2006. Byull. Izobret. no. 9.
Yu. A. Kondratenko and T. A. Kochina, Russ. J. Gen. Chem. 91, 2331 (2021). https://doi.org/10.1134/S107036322112001X
M. N. Akhtar, Y. C. Chen, and M. A. Aldamen, Dalton Trans. 46, 116 (2017). https://doi.org/10.1039/C6DT03843C
Y. A. Kondratenko, D. A. Zavyalova, M. Y. Arsentev, et al., Cryst. Growth Des. 22, 6886 (2022). https://doi.org/10.1021/acs.cgd.2c00331
D. A. Zav’yalova, Yu. A. Kondratenko, A. A. Zolotarev, et al., Russ. J. Coord. Chem. 49, 486 (2023). https://doi.org/10.1134/S1070328423600389
Y. Y. Karabach, A. M. Kirillov, M. Haukka, et al., J. Inorg. Biochem. 102, 1190 (2008). https://doi.org/10.1016/j.jinorgbio.2007.11.007
X. Qin, Y. Dong, M. Wang, et al., ACS Sens. 4, 2351 (2019). https://doi.org/10.1021/acssensors.9b00914
X. Qin, C. Gu, M. Minghan Wang, et al., Anal. Chem. 90, 2826 (2018). https://doi.org/10.1021/acs.analchem.7b04952
Z. Jia, H. Sun, and Q. Gu, Colloids Surf. A 419, 174 (2013). https://doi.org/10.1016/j.colsurfa.2012.12.003
T. L. Simonenko, N. P. Simonenko, P. Y. Gorobtsov, et al., Russ. J. Inorg. Chem. 67, 622 (2022). https://doi.org/10.1134/S0036023622050175
Y. Al-Hadeethi, E. M. Mkawi, O. Al-Hartomy, et al., Int. J. Energy Res. 46, 7239 (2022). https://doi.org/10.1002/er.7632
O. A. Shilova, V. V. Khalaman, A. Y. Komendantov, et al., Glass Phys. Chem. 46, 620 (2020). https://doi.org/10.1134/S1087659620060243
Y. A. Kondratenko, N. K. Golubeva, T. A. Kochina, et al., Glass Phys. Chem. 49, 69 (2023). https://doi.org/10.1134/S1087659622600818
M. Hang, X. Zhou, J. Wang, et al., Case Stud. Constr. Mater. 18, e01907 (2023). https://doi.org/10.1016/j.cscm.2023.e01907
N. E. Good, G. D. Winget, W. Winter, et al., Biochemistry 5, 467 (1966). https://doi.org/10.1021/bi00866a011
P. Pannuru, A. Rani, P. Venkatesu, M.-J. Lee, et al., Int. J. Biol. Macromol. 112, 720 (2018). https://doi.org/10.1016/j.ijbiomac.2018.01.203
M. Taha, F. A. e Silva, M. V. Quental, et al., Green Chem. 16, 3149 (2014). https://doi.org/10.1039/C4GC00328D
D. Antuganov, M. Nadporojskii, D. Sysoev, et al., ChemistrySelect 5, 10953 (2020). https://doi.org/10.1002/slct.202002891
Y. A. Kondratenko, D. O. Antuganov, A. A. Zolotarev, et al., ChemistrySelect 7, e202200660 (2022). https://doi.org/10.1002/slct.202200660
Y. A. Kondratenko, J. S. Shilova, V. A. Gavrilov, et al., Pharmaceutics 15, 694 (2023). https://doi.org/10.3390/pharmaceutics15020694
Y. A. Kondratenko, O. N. Makovskaya, D. O. Antuga-nov, et al., J. Mol. Liq. 363, 119891 (2022). https://doi.org/10.1016/j.molliq.2022.119891
Y. A. Kondratenko, I. S. Ignatyev, D. V. Lezov, et al., J. Organomet. Chem. 958, 122188 (2022). https://doi.org/10.1016/j.jorganchem.2021.122188
V. F. Mironov, T. K. Gar, N. Yu. Khromova, et al., Zh. Obshch. Khim. 56, 638 (1986).
H. E. Gotltieb, V. Kotlyar, and A. Nudelman, Org. Chem. 62, 7512 (1997). https://doi.org/10.1021/jo971176v
L. J. Reed and H. Muench, Am. J. Epidemiol 27, 493 (1938).
A. Daina, O. Michielin, and V. Zoete, Sci. Rep. 7, 42717 (2017). https://doi.org/10.1038/srep42717
A. Lagunin, A. Stepanchikova, D. Filimonov, et al., Bioinformatics 16, 747 (2000). https://doi.org/10.1093/bioinformatics/16.8.747
V. V. Poroikov, D. A. Filimonov, T. A. Gloriozova, et al., Russ. Chem. Bull. 68, 2143 (2019). https://doi.org/10.1007/s11172-019-2683-0
I. S. Ignatyev and T. Sundius, Spectrochim. Acta A 95, 37 (2012). https://doi.org/10.1016/j.saa.2012.04.037
I. S. Ignat’ev, M. G. Voronkov, T. A. Kochina, et al., J. Struct. Chem. 55, 431 (2014). https://doi.org/10.1134/S0022476614030068
B. Glowacki, M. Lutter, W. Hiller, et al., Inorg. Chem. 58, 4244 (2019). https://doi.org/10.1021/acs.inorgchem.8b03312
V. P. Baryshok and G. N. T. Le, Russ. J. Gen. Chem. 91, 2430 (2021). https://doi.org/10.1134/S1070363221120112
H. Pajouhesh and G. R. Lenz, Neurotherapeutics 2, 541 (2005). https://doi.org/10.1602/neurorx.2.4.541
S. Bazhan, D. Antonets, E. Starostina, et al., Vaccines 8, 448 (2020). https://doi.org/10.3390/vaccines8030448
W. Wu, R. Li, X. Li, et al., Viruses 8, 6 (2016). https://doi.org/10.3390/v8010006
S. M. Lee, K. S. Sim, and K. M. Lo, Inorg. Chim. Acta 429, 195 (2015). https://doi.org/10.1016/j.ica.2015.01.017
G. Varga, N. Lajkó, M. Ugocsai, et al., Eur. J. Pharmacol. 781, 181 (2016). https://doi.org/10.1016/j.ejphar.2016.04.019
A. Bhattacharya, B. Chattopadhyay, S. Chakraborty, et al., J. Pharm. Biomed. Anal. 70, 280 (2012). https://doi.org/10.1016/j.jpba.2012.07.011
Y. A. Kondratenko, A. A. Nikonorova, A. A. Zolotarev, et al., Inorg. Chim. Acta 530, 120705 (2022). https://doi.org/10.1016/j.ica.2021.120705
Funding
This study was carried out within the state assignment of the I.V. Grebenshchikov Institute of Silicate Chemistry, Russian Academy of Sciences, and was supported by the Ministry of Education and Science of the Russian Federation (subject no. 0081-2022-0005).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors of this work declare that they have no conflicts of interest.
Additional information
Translated by Z. Svitanko
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kondratenko, Y.A., Lezov, D.V., Shtro, A.A. et al. Hypercoordinate Germanium Complexes with Ligands Containing Hydroxyalkyl Groups. Russ. J. Inorg. Chem. (2024). https://doi.org/10.1134/S0036023623602672
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1134/S0036023623602672