Abstract
The coordination of pyridine molecules with tetrapyrrole macrocyclic complexes of manganese(III) was studied depending on the ligand structure. The coordination in toluene was ended in all cases in the formation of 1 : 1 complexes, the structure of which was established using MALDI-TOF mass spectrometry and 1H NMR spectroscopy. The numerical values of the stability constants of the coordination complexes were found to vary from 0.16 to 104 L/mol, depending on the nature of the axial anion in the manganese(III) complex, the structure of the tetrapyrrole macrocycle, and the functional substitution in the macrocycle. The data obtained are useful for selecting the structures in the design of metal porphyrin-based hybrid materials by the immobilization and supramolecular chemistry methods.





Similar content being viewed by others
REFERENCES
A. M. Meireles, A. S. Guimarães, G. R. Querino, et al., Appl. Organomet. Chem. 35 (2021). https://doi.org/10.1002/aoc.6400
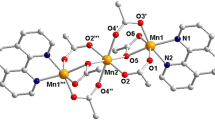
F. Gou, Q. Bian, H. Pan, et al., J. Mol. Struct. 1281, 135116 (2023). https://doi.org/10.1016/j.molstruc.2023.135116
M. Jokazi, L. S. Mpeta, and T. Nyokong, J. Electroanal. Chem. 901, 115748 (2021). https://doi.org/10.1016/j.jelechem.2021.115748
T. Lomova, Y. Tsaplev, M. Klyueva, et al., J. Organomet. Chem. 945, 121880 (2021). https://doi.org/10.1016/j.jorganchem.2021.121880
A. Žiniauskaitė, S. Ragauskas, A. K. Ghosh, et al., Ocul. Surf. 17, 257 (2019). https://doi.org/10.1016/j.jtos.2019.02.006
Y. Zheng, Y. Yuan, Y. Chai, et al., Biosens. Bioelectron. 66, 585 (2015). https://doi.org/10.1016/j.bios.2014.12.022
H.-S. Lu, M.-Y. Wang, F.-P. Ying, et al., Bioorg. Med. Chem. 35, 116090 (2021). https://doi.org/10.1016/j.bmc.2021.116090
G. Karimipour, S. Kowkabi, and A. Naghiha, Braz. Arch. Biol. Technol. 58, 431 (2015). https://doi.org/10.1590/S1516-8913201500024
K. G. Yu, D. H. Li, C. H. Zhou, et al., Chine. Chem. Lett. 20, 411 (2009). https://doi.org/10.1016/j.cclet.2008.11.030
K. A. Ashcraft, M.-K. Boss, A. Tovmasyan, et al., Int. J. Radiat. Oncol. Biol. Phys 93, 892 (2015). https://doi.org/10.1016/j.ijrobp.2015.07.2283
D. H. Weitzel, A. Tovmasyan, K. A. Ashcraft, et al., Mol. Cancer Ther. 14, 70 (2015). https://doi.org/10.1158/1535-7163.MCT-14-0343
A. V. Ezhov, A. E. Aleksandrov, K. A. Zhdanova, et al., Synth. Met. 269, 116567 (2020). https://doi.org/10.1016/j.synthmet.2020.116567
B. Fu, Y. Che, X. Yuan, et al., Dyes Pigm. 196, 109754 (2021). https://doi.org/10.1016/j.dyepig.2021.109754
E. Gacka, G. Burdzinski, B. Marciniak, et al., Phys. Chem. Chem. Phys. 22, 13456 (2020). https://doi.org/10.1039/D0CP02545C
A. S. Malyasova, P. N. Smirnova, and O. I. Koifman, Russ. J. Inorg. Chem. 67, 388 (2022). https://doi.org/10.1134/S0036023622030093
R. Chitta, D. Badgurjar, G. Reddy, et al., J. Porphyrins Phthalocyanines 25, 469 (2021). https://doi.org/10.1142/S1088424621500395
J. Janczak, Polyhedron 178, 114313 (2020). https://doi.org/10.1016/j.poly.2019.114313
Y. Li, G. Wang, X. Feng, et al., J. Mol. Struct. 1242, 130819 (2021). https://doi.org/10.1016/j.molstruc.2021.130819
L. C. Nene, M. E. Managa, D. O. Oluwole, et al., Inorg. Chim. Acta 488, 304 (2019). https://doi.org/10.1016/j.ica.2019.01.012
A. Amati, P. Cavigli, A. Kahnt, et al., J. Phys. Chem. A 121, 4242 (2017). https://doi.org/10.1021/acs.jpca.7b02973
N. Amiri, F. B. Taheur, S. Chevreux, et al., Tetrahedron 73, 7011 (2017). https://doi.org/10.1016/j.tet.2017.10.029
N. G. Bichan, E. N. Ovchenkova, A. A. Ksenofontov, et al., J. Mol. Liq. 326, 115306 (2021). https://doi.org/10.1016/j.molliq.2021.115306
K. P. Birin, I. A. Abdulaeva, D. A. Polivanovskaya, et al., Russ. J. Inorg. Chem. 66, 193 (2021). https://doi.org/10.1134/S0036023621020029
S. A. Znoiko, T. V. Kustova, E. I. Pavlova, et al., Russ. J. Gen. Chem. 91, 190 (2021). https://doi.org/10.1134/S1070363221020067
E. N. Ovchenkova, N. G. Bichan, N. O. Kudryakova, et al., Dyes Pigm. 153, 225 (2018). https://doi.org/10.1016/j.dyepig.2018.02.023
E. N. Ovchenkova, M. E. Klyueva, and T. N. Lomova, Russ. J. Inorg. Chem. 62, 1483 (2017). https://doi.org/10.1134/S0036023617110134
H. Wang, Z. Fan, T. Cao, et al., J. Alloys Compd. 887, 161462 (2021). https://doi.org/10.1016/j.jallcom.2021.161462
X. Li, K. Li, D. Wang, et al., J. Porphyrins Phthalocyanines 21, 179 (2017). https://doi.org/10.1142/S1088424616501236
N. Lahanas, P. Kucheryavy, R. A. Lalancette, et al., Acta Crystallogr., Sect. C 75, 304 (2019). .https://doi.org/10.1107/S2053229619001232
K. Kadish, K. Smith, and R. Guilard, The Porphyrin Handbook, vol. 4 (Academic Press, New York, 1999).
A. D. Adler, F. R. Longo, F. Kampas, et al., J. Inorg. Nucl. Chem. 32, 2443 (1970). https://doi.org/10.1016/0022-1902(70)80535-8
E. N. Ovchenkova, M. Hanack, and T. N. Lomova, Macroheterocycles 3, 63 (2010). https://doi.org/10.6060/mhc2010.1.63
E. N. Ovchenkova, N. G. Bichan, and T. N. Lomova, Tetrahedron 71, 6659 (2015). https://doi.org/10.1016/j.tet.2015.07.054
T. N. Lomova and B. D. Berezin, Russ. J. Coord. Chem. 27, 85 (2001). https://doi.org/10.1023/A:1009523115380
M. E. Klyueva, Doctoral Sci. (Chem.) Dissertation, Moscow, 2006.
P. Turner and M. J. Gunter, Inorg. Chem. 33, 1406 (1994). https://doi.org/10.1021/ic00085a032
A. Ikezaki and M. Nakamura, J. Porphyrins Phthalocyanines 20, 318 (2016).
A. Ikezaki and M. Nakamura, Chem. Lett. 34, 1046 (2005). https://doi.org/10.1246/cl.2005.1046
G. R. Fulmer, A. J. M. Miller, N. H. Sherden, et al., Organometallics 29, 2176 (2010). https://doi.org/10.1021/om100106e
E. N. Ovchenkova, N. G. Bichan, A. V. Lyubimtsev, et al., Russ. J. Gen. Chem. 88, 1657 (2018). https://doi.org/10.1134/S1070363218080170
K. A. Askarov, B. D. Berezin, R. P. Evstigneeva, et al., Porphyrins: Structure, Properties, Synthesis, Ed. by N. S. Enikolopyan (Nauka, Moscow, 1985) [in Russian].
T. Lomova, Appl. Organomet. Chem. 35, e6254 (2021). https://doi.org/10.1002/aoc.6254
E. N. Ovchenkova, N. G. Bichan, and T. N. Lomova, Russ. J. Phys. Chem. 93, 236 (2019). https://doi.org/10.1134/S0036024419010217
T. N. Lomova, S. V. Zaitseva, O. V. Molodkina, et al., Russ. J. Coord. Chem. 25, 397 (1999).
K. M. Kadish, L. A. Bottomley, and D. Beroiz, Inorg. Chem. 17, 1124 (1978). https://doi.org/10.1021/ic50183a006
F. A. Walker, J. Am. Chem. Soc. 95, 1150 (1973). https://doi.org/10.1021/ja00785a025
X. Q. Lin, B. Boisselier-Cocolios, and K. M. Kadish, Inorg. Chem. 25, 3242 (1986). https://doi.org/10.1021/ic00238a030
E. N. Ovchenkova, N. G. Bichan, A. S. Semeikin, et al., Macroheterocycles 11, 79 (2018). https://doi.org/10.6060/mhc170301o
ACKNOWLEDGMENTS
Performed using the research equipment of the Center for Collective Use “Upper Volga Regional Center for Physicochemical Research.”
Funding
This study was supported by the Russian Science Foundation (grant no. 21-73-20090).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors of this work declare that they have no conflicts of interest.
Additional information
Translated by Z. Svitanko
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ovchenkova, E.N., Elkhovikova, A.A. & Lomova, T.N. Coordination of Manganese(III) Porphyrins with Pyridine as a Model of Formation of Donor–Acceptor Dyads with Fullerene Acceptors. Russ. J. Inorg. Chem. (2024). https://doi.org/10.1134/S0036023623602726
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1134/S0036023623602726