Abstract
Introduction
Tacrolimus-associated neurotoxicity (TAN) manifests with wide clinical spectrum, ranging from mild tremors to severe encephalopathy. The isolated involvement of the brainstem is a rarely documented presentation of TAN, and its clinical and diagnostic characteristics are unclear.
Methods
We report two cases of brainstem-isolated TAN (bi-TAN). Moreover, we performed a systematic review of the literature on bi-TAN and extracted data concerning demographics, clinical characteristics, radiological features, and management. The systematic literature search followed PRISMA guidelines and a pre-defined protocol.
Results
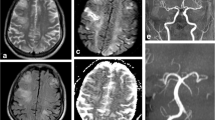
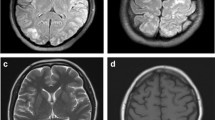
Eleven patients, including our two, were identified (mean age: 41.3 years, ± 18.8; five males, 45%). Speech disturbance was the most common clinical presentation (45%). The mean latency from Tacrolimus initiation to bi-TAN onset was 26 days (± 30.8). Tacrolimus serum level tested above the reference range in three patients (mean: 26.83 ± 5.48). Brain MRI showed T2-FLAIR hyperintensities; three showed restricted diffusion on ADC maps. Neurological symptoms resolved completely in seven patients (63%) after Tacrolimus withdrawal or dose reduction.
Conclusions
Our findings suggest that bi-TAN could represent a brainstem variant of posterior reversible encephalopathy syndrome. Recognition of bi-TAN as a potential cause of isolated brainstem lesions is crucial to disentangle the diagnostic work-up and ensure prompt withdrawal or reduction of the offending agent.



Similar content being viewed by others
Data availability
The corresponding author has full access to all of the data and has the right to publish any and all data separate and apart from any sponsor.
Code availability
Not applicable.
References
Wijdicks EFM, Wiesner RH, Dahlke LJ, Krom RAF (1994) FK506-induced neurotoxicity in liver transplantation. Ann Neurol 35(4):498–501. https://doi.org/10.1002/ana.410350422
Wu Q et al (2010) Tacrolimus-associated posterior reversible encephalopathy syndrome after solid organ transplantation. Eur Neurol 64(3):169–177. https://doi.org/10.1159/000319032
Furukuwa M, Terae S, Chu BC, Kaneko K, Kamada H, Miyasaka K (2001) MRI in seven cases of tacrolimus (FK-506) encephalopathy: utility of FLAIR and diffusion-weighted imaging. Neuroradiology 43(8):615–621. https://doi.org/10.1007/s002340100545
Hsiao CY, Ho MC, Ho CM, Wu YM, Lee PH, Hu RH (2021) Long-term tacrolimus blood trough level and patient survival in adult liver transplantation. J Pers Med. https://doi.org/10.3390/jpm11020090
Dumont FJ (2012) FK506, an immunosuppressant targeting calcineurin function. Curr Med Chem. https://doi.org/10.2174/0929867003374723
Spencer CM, Goa KL, Gillis JC (1997) Tacrolimus. An update of its pharmacology and clinical efficacy in the management of organ transplantation. Drugs. https://doi.org/10.2165/00003495-199754060-00009
Bechstein WO (2000) Neurotoxicity of calcineurin inhibitors: impact and clinical management. Transpl Int. https://doi.org/10.1111/j.1432-2277.2000.tb01004.x
Appignani BA, Bhadelia RA, Blacklow SC, Wang AK, Roland SF, Freeman RB (1996) Neuroimaging findings in patients on immunosuppressive therapy: experience with tacrolimus toxicity. Am J Roentgenol 166(3):683–688. https://doi.org/10.2214/ajr.166.3.8623651
Triplett JD, Kutlubaev MA, Kermode AG, Hardy T (2022) Posterior reversible encephalopathy syndrome (PRES): diagnosis and management. Pract Neurol. https://doi.org/10.1136/practneurol-2021-003194
McKinney AM et al (2007) Posterior reversible encephalopathy syndrome: incidence of atypical regions of involvement and imaging findings. Am J Roentgenol. https://doi.org/10.2214/AJR.07.2024
Saad AF, Chaudhari R, Wintermark M (2019) Imaging of atypical and complicated posterior reversible encephalopathy syndrome. Front Neurol. https://doi.org/10.3389/fneur.2019.00964
Wu Q, Marescaux C, Qin X, Kessler R, Yang J (2014) Heterogeneity of radiological spectrum in tacrolimus-associated encephalopathy after lung transplantation. Behav Neurol 2014(i). https://doi.org/10.1155/2014/931808
Covarrubias DJ, Luetmer PH, Campeau NG (2002) Posterior reversible encephalopathy syndrome: prognostic utility of quantitative diffusion-weighted MR images. AJNR Am J Neuroradiol. 23(6):1038–1048
Yakel JL (1997) Calcineurin regulation of synaptic function: from ion channels to transmitter release and gene transcription. Trends Pharmacol Sci. https://doi.org/10.1016/S0165-6147(97)01046-8
Lai MM, Luo HR, Burnett PE, Hong JJ, Snyder SH (2000) The calcineurin-binding protein cain is a negative regulator of synaptic vesicle endocytosis. J Biol Chem. https://doi.org/10.1074/jbc.C000429200
Lyson T, Ermel LAD, Belshaw PJ, Alberg DG, Schreiber SL, Victor RG (1993) Cyclosporine- and FK506-induced sympathetic activation correlates with calcineurin-mediated inhibition of T-cell signaling. Circ Res. https://doi.org/10.1161/01.res.73.3.596
Meyer N, Brodowski L, von Kaisenberg C, Schröder-Heurich B, von Versen-Höynck F (2021) Cyclosporine a and tacrolimus induce functional impairment and inflammatory reactions in endothelial progenitor cells. Int J Mol Sci. https://doi.org/10.3390/ijms22189696
Zhou Q, Liu H, Qiao F, Wu Y, Xu J (2010) VEGF deficit is involved in endothelium dysfunction in preeclampsia. J Huazhong Univ Sci Technol - Med Sci. https://doi.org/10.1007/s11596-010-0359-y
Tlemsani C et al (2011) Posterior reversible encephalopathy syndrome induced by anti-VEGF agents. Target Oncol. https://doi.org/10.1007/s11523-011-0201-x
Weiss N et al (2016) Cerebrospinal fluid metabolomics highlights dysregulation of energy metabolism in overt hepatic encephalopathy. J Hepatol. https://doi.org/10.1016/j.jhep.2016.07.046
Weiss N et al (2017) Modification in CSF specific gravity in acutely decompensated cirrhosis and acute on chronic liver failure independent of encephalopathy, evidences for an early blood-CSF barrier dysfunction in cirrhosis. Metab Brain Dis. https://doi.org/10.1007/s11011-016-9916-9
DiMartini A, Fontes P, Dew MA, Lotrich FE, de Vera M (2008) Age, model for end-stage liver disease score, and organ functioning predict posttransplant tacrolimus neurotoxicity. Liver Transplant. https://doi.org/10.1002/lt.21427
Jusko WJ et al (1995) Pharmacokinetics of tacrolimus in liver transplant patients. Clin Pharmacol Ther. https://doi.org/10.1016/0009-9236(95)90153-1
Wong R et al (2003) Tacrolimus-associated posterior reversible encephalopathy syndrome after allogeneic haematopoietic stem cell transplantation. Br J Haematol 122(1):128–134. https://doi.org/10.1046/j.1365-2141.2003.04447.x
Kino T et al (1987) Fk-506, a novel immunosuppressant isolated from a Streptomyces. J Antibiot (Tokyo). https://doi.org/10.7164/antibiotics.40.1249
Pirsch JD, Miller J, Deierhoi MH, Vincenti F, Filo RS (1997) A comparison of tacrolimus (FK506) and cyclosporine for immunosuppression after cadaveric renal transplantation. Transplantation. https://doi.org/10.1097/00007890-199704150-00013
Reyes J, Gayowski T, Fung J, Todo S, Alessiani M, Starzl TE (1990) Expessive dysphasia possibly related to FK506 in two liver transplant recipients. Transplantation. https://doi.org/10.1097/00007890-199012000-00028
Oliverio PJ, Restrepo L, Mitchell SA, Tornatore CS, Frankel SR (2000) Reversible tacrolimus-induced neurotoxity isolated to the brain stem. Am J Neuroradiol 21(7):1251–1254
Ravaioli M et al (2003) Speech disorder related to tacrolimus-induced pontine myelinolysis after orthotopic liver transplantation. Transplant Int Off J Eur Soc Organ Transplant. https://doi.org/10.1007/s00147-003-0620-4
Fukazawa K, Nishida S, Aguina LM, Pretto EA (2011) Central pontine myelinolysis (CPM) associated with tacrolimus (FK506) after liver transplantation. Ann Transplant 16(3):139–142. https://doi.org/10.12659/AOT.882008
Bao J, Liang H (2014) Tacrolimus-induced permanent asymptomatic pontine myelinolysis after liver transplantation. Intern Med 53(15):1713–1714. https://doi.org/10.2169/internalmedicine.53.2418
Uchida H et al (2014) Central pontine myelinolysis following pediatric living donor liver transplantation: a case report and review of literature. Pediatr Transplant 18(4):18–21. https://doi.org/10.1111/petr.12255
Silva Sieger FA, Serrano-Martínez AM, Castillo A, Vanegas Ballesteros M, Cote Florez TP (2017) Mielinolisis Póntica Central post-trasplante hepático: Reporte de Caso. Acta Neurológica Colomb 33(3):154–159
Saadi A, Schmahmann JD (2016) Pearls & Oy-sters: Tacrolimus neurotoxicity presenting as an isolated brainstem lesion. Neurology 87(13):1423. https://doi.org/10.1212/WNL.0000000000003196
Patel K, Menon J, Shanmugam N, Kalyanasundaram S, Rela M (2022) Tacrolimus-induced central pontine myelinolysis in a pediatric liver transplant recipient. J Clin Exp Hepatol 12(2):714–715. https://doi.org/10.1016/j.jceh.2021.07.011
Author information
Authors and Affiliations
Contributions
Design and conceptualization of the study: S. R., R. R., M. G.
Drafting of the manuscript: S. R., G. M. A., V. B.
Major role in acquisition and interpretation of data: S. R., G. M. A., V. B., L. F., L. S.
Analysis of neuroradiological images: L. F., L. S.
Revision of the manuscript for intellectual content: P. P., M. C. M., M. C., P. C., M. G.
Corresponding author
Ethics declarations
Ethics approval
This is a case series including two patients and a literature review. No ethical approval is required.
Informed consent
The patients included in this study provided written informed consent for the publication of anonymized data in accordance with the declaration of Helsinki.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
The participants have consented to the submission of the case report to the journal.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rossi, S., Rinaldi, R., Asioli, G.M. et al. Tacrolimus-associated neurotoxicity isolated to the brainstem: two illustrative cases and a systematic review of the literature. Neurol Sci (2024). https://doi.org/10.1007/s10072-024-07433-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10072-024-07433-8