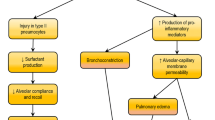
Abstract
Acute lung injury (ALI) and acute respiratory distress syndrome (ARDS) are life-threatening diseases in critically ill patients. Although pathophysiology of ALI/ARDS has been investigated in many studies, effective therapeutic strategies are still limited. Mesenchymal stem cell (MSC)-based therapy is emerging as a promising therapeutic intervention for patients with ALI. During the last two decades, researchers have focused on the efficacy and mechanism of MSC application in ALI animal models. MSC derived from variant resources exhibited therapeutic effects in preclinical studies of ALI with different mechanisms. Based on this, clinical studies on MSC treatment in ALI/ARDS has been tried recently, especially in COVID-19 caused lung injury. Emerging clinical trials of MSCs in treating COVID-19-related conditions have been registered in past two years. The advantages and potential of MSCs in the defense against COVID-19-related ALI or ARDS have been confirmed. This review provides a brief overview of recent research progress in MSC-based therapies in preclinical study and clinical trials in ALI treatment, as well as the underlying mechanisms.
Similar content being viewed by others
Abbreviations
- ALI:
-
Acute lung injury
- ARDS:
-
Acute respiratory distress syndrome
- MSC:
-
Mesenchymal stem cell
- Sca-1:
-
Stem cell antigen-1
- Stro-1:
-
Stromal cell surface marker-1
- BLM:
-
Bleomycin
- ISH:
-
In situ hybridization
- SDF-1:
-
Stromal cell-derived factor-1
- SLC:
-
Secondary lymphoid tissue chemokine
- CXCR4:
-
CXC chemokine receptor 4
- CCR7:
-
CC-chemokine receptor 7
- LPS:
-
Lipopolysaccharide
- TNF-α:
-
Tumor necrosis factor-α
- IL-1:
-
Interleukin-1
- TGF-β:
-
Transforming growth factor-beta
- IFN-γ:
-
Interferon-gamma
- COVID-19:
-
Coronavirus disease
- SARS:
-
Severe acute respiratory syndrome
- MODS:
-
Multiple organ dysfunction syndrome;
- ACE2:
-
Angiotensin-converting enzyme 2
References
Abedi F, Hayes AW, Reiter R, Karimi G (2020) Acute lung injury: the therapeutic role of Rho kinase inhibitors. Pharmacol Res 155:104736. https://doi.org/10.1016/j.phrs.2020.104736
Ahmadian-Moghadam H, Sadat-Shirazi M-S, Zarrindast M-R (2020) Therapeutic potential of stem cells for treatment of neurodegenerative diseases. Biotechnol Lett 42:1073–1101. https://doi.org/10.1007/s10529-020-02886-1
Asmussen S, Ito H, Traber DL et al (2014) Human mesenchymal stem cells reduce the severity of acute lung injury in a sheep model of bacterial pneumonia. Thorax 69:819–825. https://doi.org/10.1136/thoraxjnl-2013-204980
Butt Y, Kurdowska A, Allen TC (2016) Acute lung injury: a clinical and molecular review. Arch Pathol Lab Med 140:345–350. https://doi.org/10.5858/arpa.2015-0519-RA
Chan MCW, Kuok DIT, Leung CYH et al (2016) Human mesenchymal stromal cells reduce influenza A H5N1-associated acute lung injury in vitro and in vivo. Proc Natl Acad Sci U S A 113:3621–3626. https://doi.org/10.1073/pnas.1601911113
Chen C, Lou Y, Li X-Y et al (2020) Mapping current research and identifying hotspots on mesenchymal stem cells in cardiovascular disease. Stem Cell Res Ther 11:498. https://doi.org/10.1186/s13287-020-02009-7
Cosenza S, Toupet K, Maumus M et al (2018) Mesenchymal stem cells-derived exosomes are more immunosuppressive than microparticles in inflammatory arthritis. Theranostics 8:1399–1410. https://doi.org/10.7150/thno.21072
Cui E, Zhang L, Pan X et al (2022) RNA-Sequencing approach for exploring the therapeutic effect of umbilical cord mesenchymal stem/stromal cells on lipopolysaccharide-induced acute lung injury. Front Immunol 13:1021102. https://doi.org/10.3389/fimmu.2022.1021102
Cui E, Lv L, Chen W et al (2023) Mesenchymal stem/stromal cell-based cell-free therapy for the treatment of acute lung injury. J Cell Biochem 124:1241–1248. https://doi.org/10.1002/jcb.30469
Devaney J, Horie S, Masterson C et al (2015) Human mesenchymal stromal cells decrease the severity of acute lung injury induced by E. coli in the rat. Thorax 70:625–635. https://doi.org/10.1136/thoraxjnl-2015-206813
Ding D-C, Chang Y-H, Shyu W-C, Lin S-Z (2015) Human umbilical cord mesenchymal stem cells: a new era for stem cell therapy. Cell Transplant 24:339–347. https://doi.org/10.3727/096368915X686841
Doi D, Magotani H, Kikuchi T et al (2020) Pre-clinical study of induced pluripotent stem cell-derived dopaminergic progenitor cells for Parkinson’s disease. Nat Commun 11:3369. https://doi.org/10.1038/s41467-020-17165-w
Dominici M, Le Blanc K, Mueller I et al (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The international society for cellular therapy position statement. Cytotherapy 8:315–317. https://doi.org/10.1080/14653240600855905
Eom YW, Shim KY, Baik SK (2015) Mesenchymal stem cell therapy for liver fibrosis. Korean J Intern Med 30:580–589. https://doi.org/10.3904/kjim.2015.30.5.580
Fan E, Brodie D, Slutsky AS (2018) Acute respiratory distress syndrome: advances in diagnosis and treatment. JAMA 319:698–710. https://doi.org/10.1001/jama.2017.21907
Fang J, Wei H, Wang H et al (2023) Human placenta-derived mesenchymal stem cell administration protects against acute lung injury in a mouse model. J Cell Biochem 124:1249–1258. https://doi.org/10.1002/jcb.30445
Fathi-Kazerooni M, Fattah-Ghazi S, Darzi M et al (2022) Safety and efficacy study of allogeneic human menstrual blood stromal cells secretome to treat severe COVID-19 patients: clinical trial phase I and II. Stem Cell Res Ther 13:96. https://doi.org/10.1186/s13287-022-02771-w
Feng Y, Huang J, Wu J et al (2020) Safety and feasibility of umbilical cord mesenchymal stem cells in patients with COVID-19 pneumonia: A pilot study. Cell Prolif 53:e12947. https://doi.org/10.1111/cpr.12947
Ford E, Pearlman J, Ruan T et al (2020) Human pluripotent stem cells-based therapies for neurodegenerative diseases: current status and challenges. Cells 9:2517. https://doi.org/10.3390/cells9112517
Gupta N, Su X, Popov B et al (2007) Intrapulmonary delivery of bone marrow-derived mesenchymal stem cells improves survival and attenuates endotoxin-induced acute lung injury in mice. J Immunol 179:1855–1863. https://doi.org/10.4049/jimmunol.179.3.1855
Hashimoto N, Jin H, Liu T et al (2004) Bone marrow-derived progenitor cells in pulmonary fibrosis. J Clin Invest 113:243–252. https://doi.org/10.1172/JCI18847
Hayes M, Masterson C, Devaney J et al (2015) Therapeutic efficacy of human mesenchymal stromal cells in the repair of established ventilator-induced lung injury in the rat. Anesthesiology 122:363–373. https://doi.org/10.1097/ALN.0000000000000545
Hughes KT, Beasley MB (2017) Pulmonary manifestations of acute lung injury: more than just diffuse alveolar damage. Arch Pathol Lab Med 141:916–922. https://doi.org/10.5858/arpa.2016-0342-RA
Huppert LA, Matthay MA, Ware LB (2019) Pathogenesis of acute respiratory distress syndrome. Semin Respir Crit Care Med 40:31–39. https://doi.org/10.1055/s-0039-1683996
Khalili TM, Fleshner PR, Hiatt JR et al (1998) Colorectal cancer: comparison of laparoscopic with open approaches. Dis Colon Rectum 41:832–838. https://doi.org/10.1007/BF02235361
Köhnke R, Ahlers MO, Birkelbach MA et al (2021) Temporomandibular joint osteoarthritis: regenerative treatment by a stem cell containing advanced therapy medicinal product (ATMP)-an in vivo animal trial. Int J Mol Sci 22:443. https://doi.org/10.3390/ijms22010443
Kotton DN, Ma BY, Cardoso WV et al (2001) Bone marrow-derived cells as progenitors of lung alveolar epithelium. Development 128:5181–5188. https://doi.org/10.1242/dev.128.24.5181
Kozlowska U, Krawczenko A, Futoma K et al (2019) Similarities and differences between mesenchymal stem/progenitor cells derived from various human tissues. World J Stem Cells 11:347–374. https://doi.org/10.4252/wjsc.v11.i6.347
Lanzoni G, Linetsky E, Correa D et al (2021) Umbilical cord mesenchymal stem cells for COVID-19 acute respiratory distress syndrome: a double-blind, phase 1/2a, randomized controlled trial. Stem Cells Transl Med 10:660–673. https://doi.org/10.1002/sctm.20-0472
Leng Z, Zhu R, Hou W, et al (2020) Transplantation of ACE2-mesenchymal stem cells improves the outcome of patients with COVID-19 Pneumonia, Aging Dis, 11: 216–228. https://doi.org/10.14336/AD.2020.0228
Lian J, Lin J, Zakaria N, Yahaya BH (2020) Acute lung injury: disease modelling and the therapeutic potential of stem cells. Adv Exp Med Biol 1298:149–166. https://doi.org/10.1007/5584_2020_538
Lightner AL, Sengupta V, Qian S et al (2023) Bone marrow mesenchymal stem cell-derived extracellular vesicle infusion for the treatment of respiratory failure from COVID-19: a randomized, placebo-controlled dosing clinical trial. Chest 164:1444–1453. https://doi.org/10.1016/j.chest.2023.06.024
Liu F-B, Lin Q, Liu Z-W (2016) A study on the role of apoptotic human umbilical cord mesenchymal stem cells in bleomycin-induced acute lung injury in rat models. Eur Rev Med Pharmacol Sci 20:969–982
Liu Y, Ma Y, Du B et al (2020) Mesenchymal stem cells attenuated blood-brain barrier disruption via downregulation of aquaporin-4 expression in EAE mice. Mol Neurobiol 57:3891–3901. https://doi.org/10.1007/s12035-020-01998-z
Mahmoudifar N, Doran PM (2015) Mesenchymal stem cells derived from human adipose tissue. Methods Mol Biol 1340:53–64. https://doi.org/10.1007/978-1-4939-2938-2_4
Matthay MA, Zemans RL, Zimmerman GA et al (2019) Acute respiratory distress syndrome. Nat Rev Dis Primers 5:18. https://doi.org/10.1038/s41572-019-0069-0
Mokrá D (2020) Acute lung injury - from pathophysiology to treatment. Physiol Res 69:353–366
Moodley Y, Atienza D, Manuelpillai U et al (2009) Human umbilical cord mesenchymal stem cells reduce fibrosis of bleomycin-induced lung injury. Am J Pathol 175:303–313. https://doi.org/10.2353/ajpath.2009.080629
Mowery NT, Terzian WTH, Nelson AC (2020) Acute lung injury. Curr Probl Surg 57:100777. https://doi.org/10.1016/j.cpsurg.2020.100777
Neuringer IP, Randell SH (2004) Stem cells and repair of lung injuries. Respir Res 5:6. https://doi.org/10.1186/1465-9921-5-6
Nurkovic J, Dolicanin Z, Mustafic F et al (2016) Mesenchymal stem cells in regenerative rehabilitation. J Phys Ther Sci 28:1943–1948. https://doi.org/10.1589/jpts.28.1943
Ortiz LA, Gambelli F, McBride C et al (2003) Mesenchymal stem cell engraftment in lung is enhanced in response to bleomycin exposure and ameliorates its fibrotic effects. Proc Natl Acad Sci U S A 100:8407–8411. https://doi.org/10.1073/pnas.1432929100
Ortiz LA, Dutreil M, Fattman C et al (2007) Interleukin 1 receptor antagonist mediates the antiinflammatory and antifibrotic effect of mesenchymal stem cells during lung injury. Proc Natl Acad Sci U S A 104:11002–11007. https://doi.org/10.1073/pnas.0704421104
Pan W, Gu L, Yang H et al (2023) Repeat-dose toxicity study of human umbilical cord mesenchymal stem cells in cynomolgus monkeys by intravenous and subcutaneous injection. Front Cell Dev Biol 11:1273723. https://doi.org/10.3389/fcell.2023.1273723
Park B-N, Kim J-H, Lim TS et al (2020) Therapeutic effect of mesenchymal stem cells in an animal model of Alzheimer’s disease evaluated by β-amyloid positron emission tomography imaging. Aust N Z J Psychiatry 54:883–891. https://doi.org/10.1177/0004867420917467
Parmar M, Grealish S, Henchcliffe C (2020) The future of stem cell therapies for Parkinson disease. Nat Rev Neurosci 21:103–115. https://doi.org/10.1038/s41583-019-0257-7
Patel VJ, Biswas Roy S, Mehta HJ et al (2018) Alternative and natural therapies for acute lung injury and acute respiratory distress syndrome. Biomed Res Int 2018:2476824. https://doi.org/10.1155/2018/2476824
Phinney DG, Pittenger MF (2017) Concise review: MSC-derived exosomes for cell-free therapy. Stem Cells 35:851–858. https://doi.org/10.1002/stem.2575
Phinney DG, Di Giuseppe M, Njah J et al (2015) Mesenchymal stem cells use extracellular vesicles to outsource mitophagy and shuttle microRNAs. Nat Commun 6:8472. https://doi.org/10.1038/ncomms9472
Rani S, Ryan AE, Griffin MD, Ritter T (2015) Mesenchymal stem cell-derived extracellular vesicles: toward cell-free therapeutic applications. Mol Ther 23:812–823. https://doi.org/10.1038/mt.2015.44
Ren H, Zhang Q, Wang J, Pan R (2018) Comparative effects of umbilical cord- and menstrual blood-derived MSCs in repairing acute lung injury. Stem Cells Int 2018:7873625. https://doi.org/10.1155/2018/7873625
Rojas M, Xu J, Woods CR et al (2005) Bone marrow-derived mesenchymal stem cells in repair of the injured lung. Am J Respir Cell Mol Biol 33:145–152. https://doi.org/10.1165/rcmb.2004-0330OC
Rojas M, Cárdenes N, Kocyildirim E et al (2014) Human adult bone marrow-derived stem cells decrease severity of lipopolysaccharide-induced acute respiratory distress syndrome in sheep. Stem Cell Res Ther 5:42. https://doi.org/10.1186/scrt430
Sage EK, Loebinger MR, Polak J, Janes SM (2008) The role of bone marrow-derived stem cells in lung regeneration and repair. In: StemBook. Harvard Stem Cell Institute, Cambridge (MA)
Shao J, Xia L, Ye Z et al (2023) A repeat-dose toxicity study of human umbilical cord mesenchymal stem cells in NOG mice by intravenous injection. Expert Opin Drug Metab Toxicol 19:857–866. https://doi.org/10.1080/17425255.2023.2279243
Shu L, Niu C, Li R et al (2020) Treatment of severe COVID-19 with human umbilical cord mesenchymal stem cells. Stem Cell Res Ther 11:361. https://doi.org/10.1186/s13287-020-01875-5
Si Y-L, Zhao Y-L, Hao H-J et al (2011) MSCs: biological characteristics, clinical applications and their outstanding concerns. Ageing Res Rev 10:93–103. https://doi.org/10.1016/j.arr.2010.08.005
Soetjahjo B, Malueka RG, Nurudhin A et al (2023) Effectiveness and safety of normoxic allogenic umbilical cord mesenchymal stem cells administered as adjunctive treatment in patients with severe COVID-19. Sci Rep 13:12520. https://doi.org/10.1038/s41598-023-39268-2
Song Y, Du H, Dai C et al (2018) Human adipose-derived mesenchymal stem cells for osteoarthritis: a pilot study with long-term follow-up and repeated injections. Regen Med 13:295–307. https://doi.org/10.2217/rme-2017-0152
Takao S, Nakashima T, Masuda T et al (2021) Human bone marrow-derived mesenchymal stromal cells cultured in serum-free media demonstrate enhanced antifibrotic abilities via prolonged survival and robust regulatory T cell induction in murine bleomycin-induced pulmonary fibrosis. Stem Cell Res Ther 12:506. https://doi.org/10.1186/s13287-021-02574-5
Tang J, Xu L, Zeng Y, Gong F (2021) Effect of gut microbiota on LPS-induced acute lung injury by regulating the TLR4/NF-kB signaling pathway. Int Immunopharmacol 91:107272. https://doi.org/10.1016/j.intimp.2020.107272
Trohatou O, Roubelakis MG (2017) Mesenchymal stem/stromal cells in regenerative medicine: past, present, and future. Cell Reprogram 19:217–224. https://doi.org/10.1089/cell.2016.0062
Vishnupriya S, Priya Dharshini LC, Sakthivel KM, Rasmi RR (2020) Autophagy markers as mediators of lung injury-implication for therapeutic intervention. Life Sci 260:118308. https://doi.org/10.1016/j.lfs.2020.118308
Wang W, Qian L-L, Liu H-P, Sun B (2009) Engraftment of bone marrow stromal cells in lipopolysaccharide-injured mouse lungs. Zhongguo Dang Dai Er Ke Za Zhi 11:321–327
Wang Y, Tian M, Wang F et al (2019) Understanding the immunological mechanisms of mesenchymal stem cells in allogeneic transplantation: from the aspect of major histocompatibility complex class I. Stem Cells Dev 28:1141–1150. https://doi.org/10.1089/scd.2018.0256
Wong S-P, Rowley JE, Redpath AN et al (2015) Pericytes, mesenchymal stem cells and their contributions to tissue repair. Pharmacol Ther 151:107–120. https://doi.org/10.1016/j.pharmthera.2015.03.006
Xiao K, He W, Guan W et al (2020) Mesenchymal stem cells reverse EMT process through blocking the activation of NF-κB and Hedgehog pathways in LPS-induced acute lung injury. Cell Death Dis 11:863. https://doi.org/10.1038/s41419-020-03034-3
Yamanaka S (2020) Pluripotent stem cell-based cell therapy-promise and challenges. Cell Stem Cell 27:523–531. https://doi.org/10.1016/j.stem.2020.09.014
Zhang H, Cui Y, Zhou Z et al (2019) Alveolar type 2 epithelial cells as potential therapeutics for acute lung injury/acute respiratory distress syndrome. Curr Pharm Des 25:4877–4882. https://doi.org/10.2174/1381612825666191204092456
Zhu H, Xiong Y, Xia Y et al (2017) Therapeutic effects of human umbilical cord-derived mesenchymal stem cells in acute lung injury mice. Sci Rep 7:39889. https://doi.org/10.1038/srep39889
Funding
This work was supported by the Key Technologies R&D Program of Zhejiang Province (grant number 2019C03041), and National Key Research and Development Program of China (grant number 2016YFC1000810).
Author information
Authors and Affiliations
Contributions
JFL drafted the manuscript. WYD contributed to specific sections. SHX performed literature search and analysis in revised process. FFW reviewed the literature. EHC and RLP revised the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Confict of interest
The authors declared no potential conflicts of interest.
Ethical approval
Not applicable
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liang, J., Dai, W., Xue, S. et al. Recent progress in mesenchymal stem cell-based therapy for acute lung injury. Cell Tissue Bank (2024). https://doi.org/10.1007/s10561-024-10129-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10561-024-10129-0