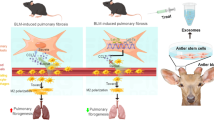
Abstract
Idiopathic pulmonary fibrosis (IPF) is a chronic pulmonary fibrosis disease that is fatal. Mesenchymal stem cells (MSCs)-secreted exosomes (exos) have been linked to improving PF. Moreover, exosomal microRNAs (miRs) can control the growth of numerous diseases, including lung disorders. Our bioinformatics analysis showed that miR-30b was downregulated in tissue samples from surgical remnants of biopsies or lungs explanted from patients with IPF who underwent pulmonary transplantation. This suggests that miR-30b plays an important role in both the pathogenesis and treatment of IPF. Herein, this research was designed to ascertain the mechanism of MSCs-exos-packaged miR-30b in alleviating PF. The serum was harvested from idiopathic PF (IPF) patients with interstitial pneumonia caused by dermatomyositis and the MLE12 lung epithelial cell fibrosis model was built with TGF-β1 (10 ng/mL), followed by miR-30b expression determination. TGF-β1-stimulated MLE12 cells were co-incubated with exos from MSCs with or without Spred2 or Runx1 overexpression, followed by measurement of cell viability and apoptosis. After establishing the IPF mouse model with bleomycin and injecting exos and/or silencing and overexpressing adenovirus vectors, fibrosis evaluation was conducted. In mice and cells, the expression of TGF-β1, TNF-α, and IL-1β was tested via ELISA, and the levels of E-cad, ZO-1, α-SMA, and collagen type I via western blot analysis. The promoters of miR-30b, Runx1, and Spred2 were investigated. miR-30b was downregulated in the serum of IPF patients and TGF-β1-stimulated MLE12 cells. Mechanistically, miR-30b inhibited Spred2 transcription by negatively targeting Runx1. MSCs-exos or MSCs-exo-miR-30b decreased the apoptosis, inflammation, and fibrosis while increasing their viability in TGF-β1-stimulated MLE12 cells, which was annulled by overexpressing Runx1 or Spred2. Exo-miR-30b decreased Runx1 expression to downregulate Spred2, reducing fibrosis and inflammation in IPF mice. Our results indicated that MSCs-exos-encapsulated miR-30b had a potential function to inhibit PF and part of its function may be achieved by targeting RUNX1 to reduce the Spred2 transcription level. Moreover, this work offered evidence and therapeutic targets for therapeutic strategies for managing clinical PF in patients.







Similar content being viewed by others
Data availability
Data will be made available on request.
References
Abreu SC, Lopes-Pacheco M, Weiss DJ, Rocco PRM (2021) Mesenchymal stromal cell-derived extracellular vesicles in lung diseases: current status and perspectives. Front Cell Dev Biol 9:600711
Ashcroft T, Simpson JM, Timbrell V (1988) Simple method of estimating severity of pulmonary fibrosis on a numerical scale. J Clin Pathol 41:467–470
Chioma OS, Drake WP (2017) Role of microbial agents in pulmonary fibrosis. Yale J Biol Med 90:219–227
Dubey S, Dubey PK, Umeshappa CS, Ghebre YT, Krishnamurthy P (2022) Inhibition of RUNX1 blocks the differentiation of lung fibroblasts to myofibroblasts. J Cell Physiol 237:2169–2182
Eom YW, Shim KY, Baik SK (2015) Mesenchymal stem cell therapy for liver fibrosis. Korean J Intern Med 30:580–589
Ghafouri-Fard S, Niazi V, Hussen BM, Omrani MD, Taheri M, Basiri A (2021) The emerging role of exosomes in the treatment of human disorders with a special focus on mesenchymal stem cells-derived exosomes. Front Cell Dev Biol 9:653296
Guiot J, Moermans C, Henket M, Corhay JL, Louis R (2017) Blood biomarkers in idiopathic pulmonary fibrosis. Lung 195:273–280
Ji Q, Hou J, Yong X, Gong G, Muddassir M, Tang T, Xie J, Fan W, Chen X (2021) Targeted dual small interfering ribonucleic acid delivery via non-viral polymeric vectors for pulmonary fibrosis therapy. Adv Mater 33:e2007798
Kawara A, Mizuta R, Fujisawa M, Ito T, Li C, Nakamura K, Sun C, Kuwabara M, Kitabatake M, Yoshimura T, Matsukawa A (2020) Spred2-deficiency enhances the proliferation of lung epithelial cells and alleviates pulmonary fibrosis induced by bleomycin. Sci Rep 10:16490
Li W, Li C, Zhou T, Liu X, Liu X, Li X, Chen D (2017) Role of exosomal proteins in cancer diagnosis. Mol Cancer 16:145
Li JW, Wei L, Han Z, Chen Z (2019) Mesenchymal stromal cells-derived exosomes alleviate ischemia/reperfusion injury in mouse lung by transporting anti-apoptotic miR-21-5p. Eur J Pharmacol 852:68–76
Lin S, Zhang R, Xu L, Ma R, Xu L, Zhu L, Hu J, An X (2020) LncRNA Hoxaas3 promotes lung fibroblast activation and fibrosis by targeting miR-450b-5p to regulate Runx1. Cell Death Dis 11:706
Nakashima R, Imura Y, Kobayashi S, Yukawa N, Yoshifuji H, Nojima T, Kawabata D, Ohmura K, Usui T, Fujii T, Okawa K, Mimori T (2010) The RIG-I-like receptor IFIH1/MDA5 is a dermatomyositis-specific autoantigen identified by the anti-CADM-140 antibody. Rheumatology (oxford) 49:433–440
Nalysnyk L, Cid-Ruzafa J, Rotella P, Esser D (2012) Incidence and prevalence of idiopathic pulmonary fibrosis: review of the literature. Eur Respir Rev 21:355–361
O’Hare M, Amarnani D, Whitmore HAB, An M, Marino C, Ramos L, Delgado-Tirado S, Hu X, Chmielewska N, Chandrahas A, Fitzek A, Heinrich F, Steurer S, Ondruschka B, Glatzel M, Krasemann S, Sepulveda-Falla D, Lagares D, Pedron J, Bushweller JH, Liu P, Arboleda-Velasquez JF, Kim LA (2021) Targeting runt-related transcription factor 1 prevents pulmonary fibrosis and reduces expression of severe acute respiratory syndrome coronavirus 2 host mediators. Am J Pathol 191:1193–1208
Ohkura T, Yoshimura T, Fujisawa M, Ohara T, Marutani R, Usami K, Matsukawa A (2019) Spred2 regulates high fat diet-induced adipose tissue inflammation, and metabolic abnormalities in mice. Front Immunol 10:17
Oldham JM, Ma SF, Martinez FJ, Anstrom KJ, Raghu G, Schwartz DA, Valenzi E, Witt L, Lee C, Vij R, Huang Y, Strek ME, Noth I, Investigators IP (2015) TOLLIP, MUC5B, and the response to N-acetylcysteine among individuals with idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 192:1475–1482
Perera UE, Derseh HB, Dewage SNV, Stent A, Wijayarathna R, Snibson KJ (2021) Evaluation of microRNA expression in a sheep model for lung fibrosis. BMC Genomics 22:827
Sgalla G, Iovene B, Calvello M, Ori M, Varone F, Richeldi L (2018) Idiopathic pulmonary fibrosis: pathogenesis and management. Respir Res 19:32
Sharif R (2017) Overview of idiopathic pulmonary fibrosis (IPF) and evidence-based guidelines. Am J Manag Care 23:S176–S182
Srour N, Thebaud B (2015) Mesenchymal stromal cells in animal bleomycin pulmonary fibrosis models: a systematic review. Stem Cells Transl Med 4:1500–1510
Tang X, Sun L, Jin X, Chen Y, Zhu H, Liang Y, Wu Q, Han X, Liang J, Liu X, Liang Z, Wang G, Luo F (2017) Runt-related transcription factor 1 regulates LPS-induced acute lung injury via NF-kappaB signaling. Am J Respir Cell Mol Biol 57:174–183
Tang X, Sun L, Wang G, Chen B, Luo F (2018) RUNX1: a regulator of NF-kB signaling in pulmonary diseases. Curr Protein Pept Sci 19:172–178
Tkach M, Thery C (2016) Communication by extracellular vesicles: where we are and where we need to go. Cell 164:1226–1232
Willis GR, Fernandez-Gonzalez A, Anastas J, Vitali SH, Liu X, Ericsson M, Kwong A, Mitsialis SA, Kourembanas S (2018) Mesenchymal stromal cell exosomes ameliorate experimental bronchopulmonary dysplasia and restore lung function through macrophage immunomodulation. Am J Respir Crit Care Med 197:104–116
Wolters PJ, Blackwell TS, Eickelberg O, Loyd JE, Kaminski N, Jenkins G, Maher TM, Molina-Molina M, Noble PW, Raghu G, Richeldi L, Schwarz MI, Selman M, Wuyts WA, Schwartz DA (2018) Time for a change: is idiopathic pulmonary fibrosis still idiopathic and only fibrotic? Lancet Respir Med 6:154–160
Xiao K, He W, Guan W, Hou F, Yan P, Xu J, Zhou T, Liu Y, Xie L (2020) Mesenchymal stem cells reverse EMT process through blocking the activation of NF-kappaB and Hedgehog pathways in LPS-induced acute lung injury. Cell Death Dis 11:863
Xie T, Wang Y, Deng N, Huang G, Taghavifar F, Geng Y, Liu N, Kulur V, Yao C, Chen P, Liu Z, Stripp B, Tang J, Liang J, Noble PW, Jiang D (2018) Single-cell deconvolution of fibroblast heterogeneity in mouse pulmonary fibrosis. Cell Rep 22:3625–3640
Xu C, Zhao J, Li Q, Hou L, Wang Y, Li S, Jiang F, Zhu Z, Tian L (2020) Exosomes derived from three-dimensional cultured human umbilical cord mesenchymal stem cells ameliorate pulmonary fibrosis in a mouse silicosis model. Stem Cell Res Ther 11:503
Yagishita M, Kondo Y, Terasaki T, Terasaki M, Shimizu M, Honda F, Oyama A, Takahashi H, Yokosawa M, Asashima H, Hagiwara S, Tsuboi H, Matsumoto I, Sumida T (2018) Clinically amyopathic dermatomyositis with interstitial pneumonia that was successfully treated with plasma exchange. Intern Med 57:1935–1938
Yi X, Wei X, Lv H, An Y, Li L, Lu P, Yang Y, Zhang Q, Yi H, Chen G (2019) Exosomes derived from microRNA-30b-3p-overexpressing mesenchymal stem cells protect against lipopolysaccharide-induced acute lung injury by inhibiting SAA3. Exp Cell Res 383:111454
Yu B, Zhang X, Li X (2014) Exosomes derived from mesenchymal stem cells. Int J Mol Sci 15:4142–4157
Zhang Z, Ge L, Zhang S, Wang J, Jiang W, Xin Q, Luan Y (2020) The protective effects of MSC-EXO against pulmonary hypertension through regulating Wnt5a/BMP signalling pathway. J Cell Mol Med 24:13938–13948
Zhang Y, Cai Y, Zhang H, Zhang J, Zeng Y, Fan C, Zou S, Wu C, Fang S, Li P, Lin X, Wang L, Guan M (2021) Brown adipose tissue transplantation ameliorates diabetic nephropathy through the miR-30b pathway by targeting Runx1. Metabolism 125:154916
Zhang Q, Ban J, Chang S, Qu H, Chen J, Liu F (2023) The aggravate role of exosomal circRNA11:120406118|12040782 on macrophage pyroptosis through miR-30b-5p/NLRP3 axis in silica-induced lung fibrosis. Int Immunopharmacol 114:109476
Zhu L, Chen M, Wang W, Zhu J, Wu H (2023) microRNA-141-3p mediates epithelial cell proliferation, apoptosis, and epithelial-mesenchymal transition and alleviates pulmonary fibrosis in mice via Spred2. Histol Histopathol 38:1269–1282
Acknowledgements
Not applicable.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
ZL and XY contributed equally to this research. ZL and XY conceived the ideas. ZL, XY, WJQ, ZY, and WXH designed the experiments. ZL, XY, WJQ, ZY, and ZJW performed the experiments. ZL, XY, and WXH analyzed the data. ZL and XY provided critical materials. ZY, XY, WJQ, and ZY wrote the manuscript. WHX supervised the study. All the authors have read and approved the final version for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare there is no conflict of interests.
Ethical approval
This study was approved by the Ethics Committee of the Second Affiliated Hospital, Zhejiang University School of Medicine and fully complied with the declaration of Helsinki. All participants were signed informed consent forms.
Additional information
Communicated by Martine Collart.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhu, L., Xu, Y., Wang, J. et al. Mesenchymal stem cells-derived exosomes carrying microRNA-30b confer protection against pulmonary fibrosis by downregulating Runx1 via Spred2. Mol Genet Genomics 299, 33 (2024). https://doi.org/10.1007/s00438-024-02116-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00438-024-02116-7