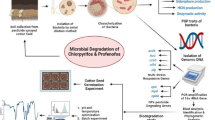
Abstract
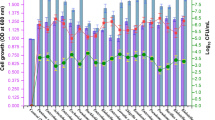
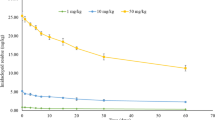
Imidacloprid (IMD), a neonicotinoid insecticide, is intensively used in agricultural fields for effective protection against aphids, cane beetles, thrips, stink bugs, locusts, etc., is causing serious environmental concerns. In recent years, seed treatment with Imidacloprid is being practiced mainly to prevent sucking insect pests. In India, due to the increase in application of this insecticide residue has been proven to have an impact on the quality of soil and water. In view of this, the current investigation is focussed on sustainable approach to minimize the residual effect of IMD in agricultural fields. The present study reveals a most promising imidacloprid resistant bacterium Lysinibacillus fusiformis IMD-Bio5 strain isolated from insecticide-contaminated soil. The isolated bacterial strain upon tested for its biodegradation potential on mineral salt medium (MSM) showed a significant survival growth at 150 g/L of IMD achieved after 3 days, whereas immobilized cells on MSM amended with 200 g/L of IMD as the sole carbon source provided degradation of 188 and 180 g/L of IMD in silica beads and sponge matrices, respectively. The liquid chromatography mass spectrometry was performed to test the metabolite responsive for IMD biodegradation potential of L. fusiformis IMD-Bio5 which showed the induced activity of the metabolite 6-Chloronicotinic acid. Furthermore, as compared to the untreated control, the Lysinibacillus fusiformis IMD-Bio5 protein profile revealed a range of patterns showing the expression of stress enzymes. Thus, results provided a most effective bacterium enabling the removal of IMD-like hazardous contaminants from the environment, which contributes to better agricultural production and soil quality, while long-term environmental advantages are restored.






Similar content being viewed by others
References
Agarwal, R., & Srinivas, R. (2007). Severe neuropsychiatric manifestations and rhabdomyolysis in a patient with imidacloprid poisoning. The American Journal of Emergency Medicine, 25, 844–845. https://doi.org/10.1016/j.ajem.2006.11.044
Costa, O. Y. A., Raaijmakers, J. M., & Kuramae, E. E. (2018). Microbial extracellular polymeric substances: Ecological function and impact on soil aggregation. Frontiers in Microbiology, 9, 1636. https://doi.org/10.3389/fmicb.2018.01636
David, D., George, I. A., & Peter, J. V. (2007). Toxicology of the newer neonicotinoid insecticides: Imidacloprid poisoning in a human. Clinical Toxicology, 45, 485–486. https://doi.org/10.1080/15563650701338229
Dzionek, A., Wojcieszy´nska, D., Hupert-Kocurek, K., Adamczyk-Habrajska, M., & Guzikc, U. (2018). Immobilization of planococcus sp. S5 strain on the loofah sponge and its application in naproxen removal. Catalysts, 8, 176. https://doi.org/10.3390/catal8050176
Gautam, P., & Dubey, S. K. (2022). Biodegradation of imidacloprid: Molecular and kinetic analysis. Bioresource Technology, 350, 126915. https://doi.org/10.1016/j.biortech.2022.126915
Gautam, P., Pandey, A. K., Gupta, A., & Dubey, S. K. (2023). Microcosm-omics centric investigation reveals elevated bacterial degradation of imidacloprid. Environmental Pollution, 324, 121402. https://doi.org/10.1016/j.envpol.2023.121402
Green, T. (1977). Significance of glucose oxidase in lignin degradation. Nature, 268, 78–80. https://doi.org/10.1038/268078a0
Gupta, S., Gajbhiye, V. T., Kalpana, K., & Agnihotri, N. P. (2002). Leaching behavior of imidacloprid formulations in soil. Bulletin of Environmental Contamination and Toxicology, 68, 502–508. https://doi.org/10.1007/s001280283
Iqbal, M., Saeed, A., Edyvean, R. G. J., O’Sullivan, B., & Styring, P. (2005). Production of fungal biomass immobilized loofa sponge (FBILS)-discs for the removal of heavy metal ions and chlorinated compounds from aqueous solution. Biotechnology Letters, 27, 1319–1323. https://doi.org/10.1007/s10529-005-0477-y
Jaiswal, S., & Shukla, P. (2020). Alternative strategies for microbial remediation of pollutants via synthetic biology. Frontiers in Microbiology, 11, 808.
Jeon, J. M., Park, S. J., Choi, T. R., Park, J. H., Yang, Y. H., & Yoon, J. J. (2021). Biodegradation of polyethylene and polypropylene by Lysinibacillus species JJY0216 isolated from soil grove. Polymer Degradation and Stability, 191, 109662. https://doi.org/10.1016/j.polymdegradstab.2021.109662
Kalia, A., & Dhanya, M. S. (2022). Evaluation of biodegradation efficiency of xylene pretreated polyethylene wastes by isolated Lysinibacillus fusiformis. Nature Environment and Pollution Technology, 21, 1375–1380. https://doi.org/10.46488/NEPT.2022.v21i03.045
Kaushal, J., Mehandia, S., Singh, G., et al. (2018). Catalase enzyme: Application in bioremediation and food industry. Biocatalysis and Agricultural Biotechnology, 16, 192–199. https://doi.org/10.1016/j.bcab.2018.07.035
Kumral, Y. A., Kumral, N. A., Kolcu &, M. B. B., & Artik. (2020). Simulation study for the degradation of some insecticides during different black table olive processes. ACS Omega, 5, 14164–14172. https://doi.org/10.1021/acsomega.0c01907
Laemmli, U. K. (1970). Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 227, 680–685. https://doi.org/10.1038/227680a0
Lenka, S., Choudhary, R., Lenka, N. K., Saha, J.K., Amat, D., Patra, A. K., Gami, V., & Singh, D. (2022) Nutrient management drives the direction and magnitude of nitrous oxide flux in crop residue-returned soil under different soil moisture. Frontiers in Environmental Science, 10, 857233. https://doi.org/10.3389/fenvs.2022.857233
Lim, K. T., Shukor, M. Y., & Wasoh, H. (2014). Physical, chemical, and biological methods for the removal of arsenic compounds. BioMed Research International, 2014, 503784. https://doi.org/10.1155/2014/503784
Lin, C., Gan, L., & Chen, Z. L. (2010). Biodegradation of naphthalene by strain Bacillus fusiformis (BFN). Journal of Hazardous Materials, 182, 771–777. https://doi.org/10.1016/j.jhazmat.2010.06.101
Lowry, O. H., Roserbrough, N. T., Farr, A. L., & Randall, R. J. (1951). Protein measurement with folin phenol reagent. Journal of Biological Chemistry, 193, 265–275.
Mendes, K., de Sousa, R. N., Goulart, M. O., & Tornisielo, V. L. (2020). Role of Raw feedstock and biochar amendments on sorption-desorption and leaching potential of three 3H- and 14C-labelled pesticides in soils. Journal of Radioanalytical and Nuclear Chemistry, 324, 1373–1386. https://doi.org/10.1007/s10967-020-07128-2
Miranda, G. R. B., Raetano, C. G., Silva, E., Daam, M. A., & Cerejeira, M. J. (2011). environmental fate of neonicotinoids and classification of their potential risks to hypogean, epygean, and surface. Water Ecosystems in Brazil Human and Ecological Risk Assessment: an International Journal, 17, 981–995. https://doi.org/10.1080/10807039.2011.588159
Mohammed, Y. M. M., & Badawy, M. E. I. (2017). Biodegradation of imidacloprid in liquid media by an isolated wastewater fungus Aspergillus terreus YESM3. Journal of Environmental Science and Health, Part B, 52, 752–761. https://doi.org/10.1080/03601234.2017.1356666
Mukherjee, S., Chowdhuri, U. R., & Kundu, P. P. (2016). Bio-degradation of polyethylene waste by simultaneous use of two bacteria: Bacillus licheniformis for production of bio-surfactant and Lysinibacillus fusiformis for bio-degradation. RSC Advances, 6, 2982–2992. https://doi.org/10.1039/C5RA25128A
Mundhe, S. A., Birajdar, S. V., Chavan, S. S., & Pawar, N. R. (2017). Imidacloprid poisoning: An emerging cause of potentially fatal poisoning. Indian Journal of Critical Care Medicine., 21, 786–788. https://doi.org/10.4103/ijccm.IJCCM_152_17
Pandey, G., Dorrian, S., Russell, R. J., & Oakeshott, J. G. (2009). Biotransformation of the neonicotinoid insecticides imidacloprid and thiamethoxam by Pseudomonas sp. 1G. Biochemical and Biophysical Research Communications, 380, 710–740. https://doi.org/10.1016/j.bbrc.2009.01.156
Pang, S., Lin, Z., Zhang, W., Mishra, S., Bhatt, P., & Chen, S. (2020). Insights into the microbial degradation and biochemical mechanisms of neonicotinoids. Frontiers in Microbiology, 11, 868. https://doi.org/10.3389/fmicb.2020.00868
Pooja (2017) Isolation and characterization of imidacloprid degrading bacteria. Doctoral dissertation. Chaudhary Charan Singh Haryana Agricultural University (CCSHAU), Haryana. http://krishikosh.egranth.ac.in/handle/1/5810031702
Preetha, G., Stanley, J., & Manoharan, T. (2018). Harvest time residues of imidacloprid in cotton seed, lint, oil and bhendi (Okra) fruits. Journal of the Entomological Research Society, 42, 391–393. https://doi.org/10.5958/0974-4576.2018.00066.X
Proenca, P., Teixeira, H., Castanheira, F., Pinheiro, J., Monsanto, P. V., et al. (2005). Two fatal intoxication cases with imidacloprid: LC/MS analysis. Forensic Science International, 153, 75–80. https://doi.org/10.1016/j.forsciint.2005.04.015
Rana, S., Jindal, V., Mandal, K., et al. (2015). Thiamethoxam degradation by Pseudomonas and bacillus strains isolated from agricultural soils. Environmental Monitoring and Assessment, 187, 300. https://doi.org/10.1007/s10661-015-4532-4
Reyes-Cervantes, A., Robles-Morales, D. L., Téllez-Jurado, A., Huerta-Ochoa, S., Jiménez-González, A., & Medina-Moreno, S. A. (2021). Evaluation in the performance of the biodegradation of herbicide diuron to high concentrations by Lysinibacillus fusiformis acclimatized by sequential batch culture. Journal of Environmental Management, 291, 112688. https://doi.org/10.1016/j.jenvman.2021.112688
Sabourmoghaddam, N., Zakaria, M. P., & Omar, D. (2015). Evidence for the microbial degradation of imidacloprid in soils of cameron highlands. Journal of the Saudi Society of Agricultural Sciences, 14, 182–188. https://doi.org/10.1016/j.jssas.2014.03.002
Sánchez-Bayo, F., & Hyne, R. V. (2014). Detection and analysis of neonicotinoids in river waters—development of a passive sampler for three commonly used insecticides. Chemosphere, 99, 143–151. https://doi.org/10.1016/j.chemosphere.2013.10.051
Sari, I., & Simarani, K. (2019). Decolorization of selected azo dye by Lysinibacillus fusiformis W1B6: Biodegradation optimization, isotherm, and kinetic study biosorption mechanism. Adsorption Science & Technology, 37, 492–508. https://doi.org/10.1177/0263617419848897
Satapute, P., Paidi, M. K., Kurjogi, M., & Jogaiah, S. (2019). Physiological adaptation and spectral annotation of arsenic and cadmium heavy metal-resistant and susceptible strain Pseudomonas Taiwanensis. Environmental Pollution, 251, 555–563. https://doi.org/10.1016/j.envpol.2019.05.054
Satapute, P., De Britto, S., Hadimani, S., Abdelrahman, M., Alarifi, S., Govind, S. R., & Jogaiah, S. (2023). Bacterial chemotaxis of herbicide atrazine provides an insight into the degradation mechanism through intermediates hydroxyatrazine, N-N-isopropylammelide, and cyanuric acid compounds. Environmental Research, 237(Pt 2), 117017. https://doi.org/10.1016/j.envres.2023.117017
Satapute, P., & Jogaiah, S. (2022). A biogenic microbial biosurfactin that degrades difenoconazole fungicide with potential antimicrobial and oil displacement properties. Chemosphere, 286, 131694. https://doi.org/10.1016/j.chemosphere.2021.131694
Schloter, M., Nannipieri, P., Sørensen, S. J., & van Elsas, J. D. (2018). Microbial indicators for soil quality. Biology and Fertility of Soils, 54, 1–10. https://doi.org/10.1007/s00374-017-1248-3
Seubert, N. Y. W. (1960). Determination of isoprenoid compounds by microorganisms. Isolation and characterization of an isoprenoid degrading bacterium. Pseudomonas citronellolis, new species. Journal of Bacteriology, 79, 426–434. https://doi.org/10.1128/jb.79.3.426-434.1960
Sharma, S., Singh, B., & Gupta, V. K. (2014). Assessment of imidacloprid degradation by soil-isolated Bacillus alkalinitrilicus. Environmental Monitoring and Assessment, 186, 7183–7193. https://doi.org/10.1007/s10661-014-3919-y
Simon-Delso, N., Amaral-Rogers, V., Belzunces, L. P., Bonmatin, J. M., Chagnon, M., Downs, C., Furlan, L., et al. (2015). Systemic insecticides (neonicotinoids and fipronil): Trends, uses, mode of action and metabolites. Environmental Science and Pollution Research, 2, 5–34. https://doi.org/10.1007/s11356-014-3470-y
Singh, J. S., & Gupta, V. K. (2018). Soil microbial biomass: A key soil driver in management of ecosystem functioning. Science of the Total Environment, 634, 497–500. https://doi.org/10.1016/j.scitotenv.2018.03.373
Singh, B. K., Walker, A., Morgan, J. A., & Wright, D. J. (2004). Biodegradation of chlorpyrifos by enterobacter strain B-14 and its use in bioremediation of contaminated soils. Applied and Environmental Microbiology., 70, 4855–4863. https://doi.org/10.1128/AEM.70.8.4855-4863
Sumon, K. A., Ritika, A. K., Peeters, E. T. H. M., Rashid, H., Bosma, R. H., Rahman, M. S., Fatema, M. K., & Van den Brink, P. J. (2018). Effects of imidacloprid on the ecology of sub-tropical freshwater microcosms. Environmental Pollution, 236, 432–441. https://doi.org/10.1016/j.envpol.2018.01.102
Supreeth, M. (2022). Enhanced remediation of pollutants by microorganisms-plant combination. International Journal of Environmental Science and Technology, 19, 4587–4598. https://doi.org/10.1007/s13762-021-03354-7
Takio, N., Yadav, M., & Yadav, H. S. (2021). Catalase-mediated remediation of environmental pollutants and potential application—a review. Biocatalysis and Biotransformation, 39, 389–407. https://doi.org/10.1080/10242422.2021.1932838
Tamura, K., Stecher, G., Peterson, D., Filipski, A., & Kumar, S. (2013). MEGA: Molecular evolutionary genentics analysis version 6.0. Molecular Biology and Evolution, 30, 2727–2729. https://doi.org/10.1093/molbev/mst197
Thompson, D. A., Lehmler, H. J., Kolpin, D. W., Hladik, M. L., Vargo, J. D., Schilling, K. E., LeFevre, G. H., Peeples, T. L., et al. (2020). A Critical review on the potential impacts of neonicotinoid insecticide use: Current knowledge of environmental fate, toxicity, and implications for human health. Environmental Science: Processes & Impacts, 22, 1315–1346. https://doi.org/10.1039/C9EM00586B
Timková, I., Sedláková-Kaduková, J., & Pristaš, P. (2018). Biosorption and bioaccumulation abilities of actinomycetes/streptomycetes isolated from metal contaminated Sites. Separations, 5, 54.
Tiwari, S., Tripathi, P., Mohan, D., & Singh, R. S. (2022). Imidacloprid biodegradation using novel bacteria Tepidibacillus decaturensis strain ST1 in batch and in situ microcosm study. Environmental Science and Pollution Research, 30, 61562–61572. https://doi.org/10.1007/s11356-022-24779-8
Tomlin, C. D. S. (2006). The pesticide manual, a world compendium (14th ed., pp. 598–599). Surry: British Crop Protection Council.
Wan-ling, Y., Bo, X., Gang-chao, R., Hui-ping, C., Peng-yue, Z., & Qi-liang, H. (2020). Application of imidacloprid controlled-release granules to enhance the utilization rate and control wheat aphid on winter wheat. Journal of Integrative Agriculture, 19, 3045–3053. https://doi.org/10.1016/S2095-3119(20)63240-3
Wismer, T. (2004). Novel insecticides. Clinical veterinary toxicology. In K. H. Plumlee (Ed.), Mosby (pp. 184–185). St. Louis.
Acknowledgements
All the authors thank Department of Environmental Science, Central University of Kerala, for this fruitful research work. The authors are always grateful to Karnatak University, Dharwad, and University of Mysore, India, for this collective collaborative research work.
Funding
There is no financial assistance from any funding agency to perform this research work.
Author information
Authors and Affiliations
Contributions
SJ contributed to research plan and study conception and wrote and edited the manuscript. PS performed experiment. PS, GN, and SJ were involved in literature survey and data compilation/interpretation and wrote the draft manuscript. PS and SJ contributed to figures drawn. All authors have read and approved the revised manuscript.
Corresponding author
Ethics declarations
Conflict of interest
We declare that we do not have any kind of conflict of interest pertaining to this research work.
Ethical approval
This work does not involve any human or animal participants. Hence, we do not require any ethical approval.
Consent to participate
Not applicable.
Consent to publish
All the authors hereby give full consent for publishing this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Satapute, P., Nagaraja, G. & Jogaiah, S. Microbial-based metabolites associated with degradation of imidacloprid and its impact on stress-responsive proteins. Environ Geochem Health 46, 114 (2024). https://doi.org/10.1007/s10653-024-01892-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10653-024-01892-w