Abstract
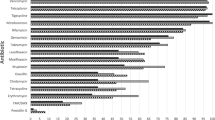
Methicillin-resistant Staphylococcus aureus (MRSA) is a highly infectious pathogen that poses a serious threat to human life and health. This study aimed to provide a scientific basis for the rational clinical use of antimicrobial drugs for treating MRSA infections and inform the development of preventive and control measures by analyzing the clinical distribution and resistance characteristics of MRSA in a hospital in Hebei China. To accomplish this, bacterial identification and drug sensitivity experiments were performed with 1858 Staphylococcus aureus (S. aureus) strains collected from a hospital from January 2018 to December 2022 using a phoenixTM-100 bacterial identification drug sensitivity analyzer. The experimental data were analyzed using WHONET 5.6 software, and the MRSA strains detected were analyzed for their clinical distribution and drug resistance. Of the 1858 S. aureus strains isolated, 429 were MRSA. Sputum samples had the highest MRSA detection rates (52.45%). Critical care medicine had the highest rate of MRSA (12.59%), followed by dermatology (9.79%). MRSA resistance to tetracycline increased by 13.9% over 5 years; resistance to quinupristin/dalfopristin also increased but remained low (1.9%). Resistance decreased to gentamicin, rifampicin, ciprofloxacin, and cotrimoxazole, though most significantly to erythromycin and clindamycin, exceeding 77% and 83%, respectively. No strains were resistant to vancomycin, teicoplanin, or linezolid, and drug resistance was most prevalent in patients ≥ 60 years old. This study will aid in improving the diagnosis and treatment of MRSA infections.


Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are not publicly available due to privacy and ethical reasons but are available from the corresponding author on reasonable request.
References
Baraz A, Chowers M, Nevo D, Obolski U (2023) The time-varying association between previous antibiotic use and antibiotic resistance. Clin Microbiol Infect 29:390.e1-390.e4. https://doi.org/10.1016/j.cmi.2022.10.021
Algammal AM, Hetta HF, Elkelish A et al (2020) Methicillin-resistant Staphylococcus aureus (MRSA): one health perspective approach to the bacterium epidemiology, virulence factors, antibiotic-resistance, and zoonotic impact. Infect Drug Resist 13:3255–3265. https://doi.org/10.2147/IDR.S272733
Guo Y, Song G, Sun M, Wang J, Wang Y (2020) Prevalence and therapies of antibiotic-resistance in Staphylococcus aureus. Front Cell Infect Microbiol 10:107. https://doi.org/10.3389/fcimb.2020.00107
Turner NA, Sharma-Kuinkel BK, Maskarinec SA et al (2019) Methicillin-resistant Staphylococcus aureus: an overview of basic and clinical research. Nat Rev Microbiol 17:203–218. https://doi.org/10.1038/s41579-018-0147-4
Cassini A, Högberg LD, Plachouras D et al (2019) Attributable deaths and disability-adjusted life-years caused by infections with antibiotic-resistant bacteria in the EU and the European Economic Area in 2015: a population-level modelling analysis. Lancet Infect Dis 19:56–66. https://doi.org/10.1016/S1473-3099(18)30605-4
Rupp ME, Fitzgerald T, Hayes K et al (2017) Effect of cessation of contact isolation for endemic methicillin-resistant Staphylococcus aureus and vancomycin-resistant Enterococci. Infect Control Hosp Epidemiol 38:1005–1007. https://doi.org/10.1017/ice.2017.122
Tabaja H, Hindy JR, Kanj SS (2021) Epidemiology of methicillin-resistant Staphylococcus aureus in Arab countries of the Middle East and North African (MENA) region. Mediterr J Hematol Infect Dis 13:e2021050. https://doi.org/10.4084/MJHID.2021.050
Gajdács M (2019) The continuing threat of methicillin-resistant Staphylococcus aureus. Antibiotics 8:52. https://doi.org/10.3390/antibiotics8020052
Koulenti D, Tsigou E, Rello J (2017) Nosocomial pneumonia in 27 ICUs in Europe: perspectives from the EU-VAP/CAP study. Eur J Clin Microbiol Infect Dis 36:1999–2006. https://doi.org/10.1007/s10096-016-2703-z
Teerawattanapong N, Panich P, Kulpokin D et al (2018) A systematic review of the burden of multidrug-resistant healthcare-associated infections among intensive care unit patients in Southeast Asia: the rise of multidrug-resistant Acinetobacter baumannii. Infect Control Hosp Epidemiol 39:525–533. https://doi.org/10.1017/ice.2018.58
Wang LC, Zhou KH, Chen W, Yu Y, Feng SF (2019) Epidemiology and risk factors for nosocomial infection in the respiratory intensive care unit of a teaching hospital in China: a prospective surveillance during 2013 and 2015. BMC Infect Dis 19:145. https://doi.org/10.1186/s12879-019-3772-2
Gastmeier P, Geffers C, Herrmann M et al (2016) Nosocomial infections and infections with multidrug-resistant pathogens: frequency and mortality. Dtsch Med Wochenschr 141:421–426. https://doi.org/10.1055/s-0041-106299
Clinical and Laboratory Standards Institute (2022) M100: performance standards for antimicrobial susceptibility testing, 32nd edn. Clinical and Laboratory Standards Institute, Wayne (PA)
Hassoun A, Linden PK, Friedman B (2017) Incidence, prevalence, and management of MRSA bacteremia across patient populations-a review of recent developments in MRSA management and treatment. Crit Care 21:211. https://doi.org/10.1186/s13054-017-1801-3
Koike Y, Nishiura H (2021) Recovery of antimicrobial susceptibility in methicillin-resistant Staphylococcus aureus (MRSA): a retrospective, epidemiological analysis in a secondary care hospital, Sapporo, Japan. PeerJ 9:e11644. https://doi.org/10.7717/peerj.11644
Schöneweck F, Schmitz RPH, Rißner F et al (2021) The epidemiology of bloodstream infections and antimicrobial susceptibility patterns in Thuringia, Germany: a five-year prospective, state-wide surveillance study (AlertsNet). Antimicrob Resist Infect Control 10:132. https://doi.org/10.1186/s13756-021-00997-6
Campanile F, Bongiorno D, Perez M et al (2015) Epidemiology of Staphylococcus aureus in Italy: first nationwide survey, 2012. J Glob Antimicrob Resist 3:247–254. https://doi.org/10.1016/j.jgar.2015.06.006
Juayang AC, de Los Reyes GB, de la Rama AJ, Gallega CT (2014) Antibiotic resistance profiling of Staphylococcus aureus isolated from clinical specimens in a tertiary hospital from 2010 to 2012. Interdiscip Perspect Infect Dis 2014:898457. https://doi.org/10.1155/2014/898457
Tadesse S, Alemayehu H, Tenna A et al (2018) Antimicrobial resistance profile of Staphylococcus aureus isolated from patients with infection at Tikur Anbessa Specialized Hospital, Addis Ababa, Ethiopia. BMC Pharmacol Toxicol 19:24. https://doi.org/10.1186/s40360-018-0210-9
Qodrati M, SeyedAlinaghi S, Dehghan Manshadi SA, Abdollahi A, Dadras O (2022) Antimicrobial susceptibility testing of Staphylococcus aureus isolates from patients at a tertiary hospital in Tehran, Iran, 2018–2019. Eur J Med Res 27:152. https://doi.org/10.1186/s40001-022-00778-w
Huang L, Zhang R, Hu Y et al (2019) Epidemiology and risk factors of methicillin-resistant Staphylococcus aureus and vancomycin-resistant enterococci infections in Zhejiang China from 2015 to 2017. Antimicrob Resist Infect Control 8:90. https://doi.org/10.1186/s13756-019-0539-x
Latha T, Anil B, Manjunatha H et al (2019) MRSA: the leading pathogen of orthopedic infection in a tertiary care hospital, South India. Afr Health Sci 19:1393–1401. https://doi.org/10.4314/ahs.v19i1.12
Tuon FF, Cieslinski J, Ono AFM et al (2019) Microbiological profile and susceptibility pattern of surgical site infections related to orthopaedic trauma. Int Orthop 43:1309–1313. https://doi.org/10.1007/s00264-018-4076-7
Ishak B, Abdul-Jabbar A, Moss GB et al (2021) De novo methicillin-resistant Staphylococcus aureus vs. methicillin-sensitive Staphylococcus aureus infections of the spine, similar clinical outcome, despite more severe presentation in surgical patients. Neurosurg Rev 44:2111–2118. https://doi.org/10.1007/s10143-020-01376-2
Kandel SN, Adhikari N, Dhungel B et al (2020) Characteristics of Staphylococcus aureus isolated from clinical specimens in a tertiary care hospital, Kathmandu. Nepal Microbiol Insights 13:1178636120972695. https://doi.org/10.1177/1178636120972695
Lakhundi S, Zhang K (2018) Methicillin-resistant Staphylococcus aureus: molecular characterization, evolution, and epidemiology. Clin Microbiol Rev 31:e00020-e118. https://doi.org/10.1128/CMR.00020-18
Lim WW, Wu P, Bond HS et al (2019) Determinants of methicillin-resistant Staphylococcus aureus (MRSA) prevalence in the Asia-Pacific region: a systematic review and meta-analysis. J Glob Antimicrob Resist 16:17–27. https://doi.org/10.1016/j.jgar.2018.08.014
Sami Awayid H, Qassim Mohammad S (2022) Prevalence and antibiotic resistance pattern of methicillin-resistant Staphylococcus aureus isolated from Iraqi hospitals. Arch Razi Inst 77:1147–1156. https://doi.org/10.22092/ARI.2022.357391.2031
Liu C, Bayer A, Cosgrove SE et al (2011) Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis 52:e18–e55. https://doi.org/10.1093/cid/ciq146
Youssef CRB, Kadry AA, El-Ganiny AM (2022) Investigating the relation between resistance pattern and type of Staphylococcal cassette chromosome mec (SCCmec) in methicillin-resistant Staphylococcus aureus. Iran J Microbiol 14:56–66. https://doi.org/10.18502/ijm.v14i1.8802
Chen JP, Battini N, Ansari MF, Zhou CH (2021) Membrane active 7-thiazoxime quinolones as novel DNA binding agents to decrease the genes expression and exert potent anti-methicillin-resistant Staphylococcus aureus activity. Eur J Med Chem 217:113340. https://doi.org/10.1016/j.ejmech.2021.113340
Cassir N, Rolain JM, Brouqui P (2014) A new strategy to fight antimicrobial resistance: the revival of old antibiotics. Front Microbiol 5:551. https://doi.org/10.3389/fmicb.2014.00551
Schweizer ML, Richardson K, Vaughan Sarrazin MS (2021) Comparative effectiveness of switching to daptomycin versus remaining on vancomycin among patients with methicillin-resistant Staphylococcus aureus (MRSA) bloodstream infections. Clin Infect Dis 72:S68–S73. https://doi.org/10.1093/cid/ciaa1572
Habib G, Gul H, Ahmad P et al (2023) Teicoplanin associated gene tcaA inactivation increases persister cell formation in Staphylococcus aureus. Front Microbiol 14:1241995. https://doi.org/10.3389/fmicb.2023.1241995
Shariati A, Dadashi M, Chegini Z et al (2020) The global prevalence of daptomycin, tigecycline, quinupristin/dalfopristin, and linezolid-resistant Staphylococcus aureus and coagulase-negative staphylococci strains: a systematic review and meta-analysis. Antimicrob Resist Infect Control 9:56. https://doi.org/10.1186/s13756-020-00714-9
Azzam A, Khaled H, Mosa M et al (2023) Epidemiology of clinically isolated methicillin-resistant Staphylococcus aureus (MRSA) and its susceptibility to linezolid and vancomycin in Egypt: a systematic review with meta-analysis. BMC Infect Dis 23:263. https://doi.org/10.1186/s12879-023-08202-2
Perlaza-Jiménez L, Tan KS, Piper SJ et al (2022) A structurally characterized staphylococcus aureus evolutionary escape route from treatment with the antibiotic linezolid. Microbiol Spectr 10:e0058322. https://doi.org/10.1128/spectrum.00583-22
Mahmoudi S, Mamishi S, Mohammadi M et al (2019) Phenotypic and genotypic determinants of mupirocin resistance among Staphylococcus aureus isolates recovered from clinical samples of children: an Iranian hospital-based study. Infect Drug Resist 12:137–143. https://doi.org/10.2147/IDR.S185610
Acknowledgements
This study was supported by the Hebei Provincial Medical Science Research Project (No. 20210366). We appreciate the assistance of the hospital staff and the technical assistance provided by the Department of Microbiology and the Antimicrobial Resistance Surveillance Team of the Infection Management Division. We would like to thank Editage (www.editage.cn) for English language editing.
Funding
This study was supported by the Hebei Provincial Medical Science Research Project (No. 20210366).
Author information
Authors and Affiliations
Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising, or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical Approval
This study was approved by the Ethics Committee of the First Affiliated Hospital of Hebei North University and complied with the ethical guidelines of the Declaration of Helsinki. As this was an observational study, isolated MRSA strains from residual samples were anonymized, ensuring compliance with the Declaration of Helsinki and therefore meeting the criteria for exemption. Formal ethical review approval was obtained after consultation with the IRB of the First Affiliated Hospital of Hebei North University (Approval number: W2023014). Written informed consent from the patients was not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lan, T., Zhang, B., Liu, J.L. et al. Prevalence and Antibiotic Resistance Patterns of Methicillin-Resistant Staphylococcus aureus (MRSA) in a Hospital Setting: A Retrospective Study from 2018 to 2022. Indian J Microbiol (2024). https://doi.org/10.1007/s12088-024-01228-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12088-024-01228-3