Abstract
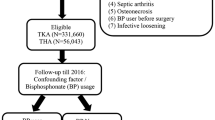
Bisphosphonates have been associated with a decreased risk of revision surgery after total joint arthroplasty of the hip or knee (TJA) because of their effects on decreased periprosthetic bone loss and prosthetic migration. However, the results in the early literature are inconsistent, and the influence of bisphosphonates on associated complications and subsequent TJA remains unknown. This study investigated the association between the use of bisphosphonates and the risk of adverse outcomes after primary TJA. This matched cohort study utilized the National Health Insurance Research Database in Taiwan to identify patients who underwent primary TJA over a 15-year period (January 2000–December 2015 inclusive). Study participants were further categorized into two groups, bisphosphonate users and nonusers, using propensity score matching. The Kaplan–Meier curve analysis and adjusted hazard ratios (aHRs) of revision surgery, adverse outcomes of primary surgery and subsequent TJA were calculated using Cox regression analysis. This study analyzed data from 6485 patients who underwent total hip arthroplasty (THA) and 20,920 patients who underwent total knee arthroplasty (TKA). The risk of revision hip and knee arthroplasty was significantly lower in the bisphosphonate users than in the nonusers (aHR, 0.54 and 0.53, respectively). Furthermore, the risk of a subsequent total joint arthroplasty, adverse events and all-cause mortality were also significantly reduced in the bisphosphonate users. This study, involving a large cohort of patients who underwent primary arthroplasties, revealed that bisphosphonate treatment may potentially reduce the risk of revision surgery and associated adverse outcomes. Furthermore, the use of bisphosphonates after TJA is also associated with a reduced need for subsequent arthroplasty.
Research Registration Unique Identifying Number (UIN): ClinicalTrials.gov Identifier—NCT05623540 (https://clinicaltrials.gov/show/NCT05623540).


Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from National Health Insurance Research Database, which belongs to Health and Welfare Data Science Center, Ministry of Health and Welfare, Taiwan. Restrictions apply to the availability of these data, which were used under license for this study. Data are only available with the permission of National Health Insurance Research Database.
References
Chen A, Gupte C, Akhtar K, Smith P, Cobb J (2012) The global economic cost of osteoarthritis: how the UK compares. Arthritis 2012:698709. https://doi.org/10.1155/2012/698709
Vos T, Flaxman AD, Naghavi M, Lozano R, Michaud C, Ezzati M et al (2012) Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the global burden of disease study 2010. Lancet 380(9859):2163–2196. https://doi.org/10.1016/s0140-6736(12)61729-2
Murray CJL (2013) The state of US health, 1990–2010: burden of diseases, injuries, and risk factors. JAMA 310(6):591. https://doi.org/10.1001/jama.2013.13805
Cross M, Smith E, Hoy D, Nolte S, Ackerman I, Fransen M et al (2014) The global burden of hip and knee osteoarthritis: estimates from the global burden of disease 2010 study. Ann Rheum Dis 73(7):1323–1330. https://doi.org/10.1136/annrheumdis-2013-204763
Zhang W, Moskowitz RW, Nuki G, Abramson S, Altman RD, Arden N et al (2008) OARSI recommendations for the management of hip and knee osteoarthritis, Part II: OARSI evidence-based, expert consensus guidelines. Osteoarthritis Cartilage 16(2):137–162. https://doi.org/10.1016/j.joca.2007.12.013
Harris WH, Sledge CB (1990) Total hip and total knee replacement (1). N Engl J Med 323(11):725–731. https://doi.org/10.1056/NEJM199009133231106
Murphy LB, Helmick CG, Schwartz TA, Renner JB, Tudor G, Koch GG et al (2010) One in four people may develop symptomatic hip osteoarthritis in his or her lifetime. Osteoarthritis Cartilage 18(11):1372–1379. https://doi.org/10.1016/j.joca.2010.08.005
Losina E, Thornhill TS, Rome BN, Wright J, Katz JN (2012) The dramatic increase in total knee replacement utilization rates in the United States cannot be fully explained by growth in population size and the obesity epidemic. J Bone Joint Surg Am 94(3):201–207. https://doi.org/10.2106/JBJS.J.01958
Kurtz S, Ong K, Lau E, Mowat F, Halpern M (2007) Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am 89(4):780–785. https://doi.org/10.2106/JBJS.F.00222
Patel A, Pavlou G, Mújica-Mota RE, Toms AD (2015) The epidemiology of revision total knee and hip arthroplasty in England and Wales: a comparative analysis with projections for the United States. A study using the National Joint Registry dataset. Bone Joint J. 97-B(8):1076–81. https://doi.org/10.1302/0301-620x.97b8.35170
Havelin LI, Engesaeter LB, Espehaug B, Furnes O, Lie SA, Vollset SE (2000) The Norwegian arthroplasty register: 11 years and 73,000 arthroplasties. Acta Orthop Scand 71(4):337–353. https://doi.org/10.1080/000164700317393321
Vasso M, Beaufils P, Cerciello S, Schiavone PA (2014) Bone loss following knee arthroplasty: potential treatment options. Arch Orthop Trauma Surg 134(4):543–553. https://doi.org/10.1007/s00402-014-1941-8
Abu-Amer Y, Darwech I, Clohisy JC (2007) Aseptic loosening of total joint replacements: mechanisms underlying osteolysis and potential therapies. Arthritis Res Ther 9(Suppl 1):S6. https://doi.org/10.1186/ar2170
Russell RGG (2011) Bisphosphonates: the first 40 years. Bone 49(1):2–19. https://doi.org/10.1016/j.bone.2011.04.022
Shanbhag AS (2006) Use of bisphosphonates to improve the durability of total joint replacements. J Am Acad Orthop Surg 14(4):215–225. https://doi.org/10.5435/00124635-200604000-00003
Hilding M, Aspenberg P (2007) Local peroperative treatment with a bisphosphonate improves the fixation of total knee prostheses: a randomized, double-blind radiostereometric study of 50 patients. Acta Orthop 78(6):795–799. https://doi.org/10.1080/17453670710014572
Hilding M, Aspenberg P (2006) Postoperative clodronate decreases prosthetic migration: 4-year follow-up of a randomized radiostereometric study of 50 total knee patients. Acta Orthop 77(6):912–916. https://doi.org/10.1080/17453670610013213
Thillemann TM, Pedersen AB, Mehnert F, Johnsen SP, Søballe K (2010) Postoperative use of bisphosphonates and risk of revision after primary total hip arthroplasty: a nationwide population-based study. Bone 46(4):946–951. https://doi.org/10.1016/j.bone.2010.01.377
Prieto-Alhambra D, Javaid MK, Judge A, Murray D, Carr A, Cooper C et al (2011) Association between bisphosphonate use and implant survival after primary total arthroplasty of the knee or hip: population based retrospective cohort study. BMJ 343:d7222. https://doi.org/10.1136/bmj.d7222
Teng S, Yi C, Krettek C, Jagodzinski M (2015) Bisphosphonate use and risk of implant revision after total hip/knee arthroplasty: a meta-analysis of observational studies. PLoS ONE 10(10):e0139927. https://doi.org/10.1371/journal.pone.0139927
Hansson U, Toksvig-Larsen S, Ryd L, Aspenberg P (2009) Once-weekly oral medication with alendronate does not prevent migration of knee prostheses: a double-blind randomized RSA study. Acta Orthop 80(1):41–45. https://doi.org/10.1080/17453670902804968
Walsh DA, Chapman V (2011) Bisphosphonates for osteoarthritis. Arthritis Res Ther 13(5):128. https://doi.org/10.1186/ar3448
von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP et al (2014) The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Int J Surg 12(12):1495–1499. https://doi.org/10.1016/j.ijsu.2014.07.013
Prieto-Alhambra D, Lalmohamed A, Abrahamsen B, Arden NK, de Boer A, Vestergaard P et al (2014) Oral bisphosphonate use and total knee/hip implant survival: validation of results in an external population-based cohort: bisphosphonate use and implant survival. Arthritis Rheumatol 66(11):3233–3240. https://doi.org/10.1002/art.38789
Kozma C, Dickson Phillips, Meletiche. (2013) Medication possession ratio: implications of using fixed and variable observation periods in assessing adherence with disease-modifying drugs in patients with multiple sclerosis. Patient Prefer Adherence. https://doi.org/10.2147/ppa.s40736
Fu S-H, Wang C-Y, Yang R-S, Wu F-LL, Hsiao F-Y (2017) Bisphosphonate use and the risk of undergoing total knee arthroplasty in osteoporotic patients with osteoarthritis: a nationwide cohort study in Taiwan. J Bone Joint Surg Am. 99(11):938–46. https://doi.org/10.2106/JBJS.16.00385
Deyo RA, Cherkin DC, Ciol MA (1992) Adapting a clinical comorbidity index for use with ICD-9-CM administrative databases. J Clin Epidemiol 45(6):613–619. https://doi.org/10.1016/0895-4356(92)90133-8
National Joint Registry. National Joint Registry for England and Wales. 2009
Paling IH (2000) Wear in total hip and knee replacements. J Bone Joint Surg-Am Volume 82(12):1806. https://doi.org/10.2106/00004623-200012000-00020
Carbone LD, Nevitt MC, Wildy K, Barrow KD, Harris F, Felson D et al (2004) The relationship of antiresorptive drug use to structural findings and symptoms of knee osteoarthritis. Arthritis Rheum 50(11):3516–3525. https://doi.org/10.1002/art.20627
Vaysbrot EE, Osani MC, Musetti M-C, McAlindon TE, Bannuru RR (2018) Are bisphosphonates efficacious in knee osteoarthritis? A meta-analysis of randomized controlled trials. Osteoarthritis Cartilage 26(2):154–164. https://doi.org/10.1016/j.joca.2017.11.013
Kiyomoto K, Iba K, Hanaka M, Ibe K, Hayakawa H, Teramoto A et al (2020) High bone turnover state under osteoporotic changes induces pain-like behaviors in mild osteoarthritis model mice. J Bone Miner Metab 38(6):806–818. https://doi.org/10.1007/s00774-020-01124-y
Fernández-Martín S, López-Peña M, Muñoz F, Permuy M, González-Cantalapiedra A (2021) Bisphosphonates as disease-modifying drugs in osteoarthritis preclinical studies: a systematic review from 2000 to 2020. Arthritis Res Ther 23(1):60. https://doi.org/10.1186/s13075-021-02446-6
Tande AJ, Patel R (2014) Prosthetic joint infection. Clin Microbiol Rev 27(2):302–345. https://doi.org/10.1128/CMR.00111-13
Verdrengh M, Carlsten H, Ohlsson C, Tarkowski A (2007) Addition of bisphosphonate to antibiotic and anti-inflammatory treatment reduces bone resorption in experimental Staphylococcus aureus-induced arthritis. J Orthop Res 25(3):304–310. https://doi.org/10.1002/jor.20317
Hiltunen AK, Vuorela PM (2014) Fallarero bisphosphonates offer protection against prosthetic joint infections caused by Staphylococcus aureus and Staphylococcus epidermidis biofilms. J Drug Deliv Sci Technol 27:302–345. https://doi.org/10.1128/CMR.00111-13
Benkovich V, Klassov Y, Mazilis B, Bloom S (2020) Periprosthetic fractures of the knee: a comprehensive review. Eur J Orthop Surg Traumatol 30(3):387–399. https://doi.org/10.1007/s00590-019-02582-5
Ramavath A, Lamb JN, Palan J, Pandit HG, Jain S (2020) Postoperative periprosthetic femoral fracture around total hip replacements: current concepts and clinical outcomes. EFORT Open Rev 5(9):558–567. https://doi.org/10.1302/2058-5241.5.200003
MacKenzie SA, Ng RT, Snowden G, Powell-Bowns MFR, Duckworth AD, Scott CEH (2019) Periprosthetic atypical femoral fractures exist and are associated with duration of bisphosphonate therapy. Bone Joint J 101(10):1285–91. https://doi.org/10.1302/0301-620X.101B10.BJJ-2019-0599.R2
Ben-Shlomo Y, Blom A, Boulton C, Brittain R, Clark E, Craig R, et al. The National Joint Registry 16th Annual Report 2019. https://pubmed.ncbi.nlm.nih.gov/32744812/
Nasser AAH, Chauhan G, Osman K, Nandra S, Nandra R, Mahmood A (2021) Study protocol for a national retrospective review of femoral periprosthetic fracture management. Is there variation in practice? J Surg Protoc Res Methodol. https://doi.org/10.1093/jsprm/snab004
Tai TW, Huang CF, Huang HK, Yang RS, Chen JF, Cheng TT et al (2023) Clinical practice guidelines for the prevention and treatment of osteoporosis in Taiwan: 2022 update. J Formos Med Assoc. https://doi.org/10.1016/j.jfma.2023.01.007
Holzer LA, Borotschnig L, Holzer G (2023) Evaluation of FRAX in patients with periprosthetic fractures following primary total hip and knee arthroplasty. Sci Rep 13(1):7145. https://doi.org/10.1038/s41598-023-34230-8
Dózsai D, Ecseri T, Csonka I, Gárgyán I, Doró P, Csonka Á (2020) Atypical periprosthetic femoral fracture associated with long-term bisphosphonate therapy. J Orthop Surg Res 15(1):414. https://doi.org/10.1186/s13018-020-01941-x
Ro C, Cooper O (2013) Bisphosphonate drug holiday: choosing appropriate candidates. Curr Osteoporos Rep 11(1):45–51. https://doi.org/10.1007/s11914-012-0129-9
Hooper GJ, Rothwell AG, Stringer M, Frampton C (2009) Revision following cemented and uncemented primary total hip replacement. J Bone Joint Surg Br Volume 91-B(4):451–8. https://doi.org/10.1302/0301-620X.91B4.21363
Okike K, Chan PH, Prentice HA, Paxton EW, Burri RA (2020) Association between uncemented vs cemented hemiarthroplasty and revision surgery among patients with hip fracture. JAMA 323(11):1077. https://doi.org/10.1001/jama.2020.1067
Konan S, Abdel MP, Haddad FS (2019) Cemented versus uncemented hip implant fixation. Bone Joint Res 8(12):604–607. https://doi.org/10.1302/2046-3758.812.BJR-2019-0337
Acknowledgements
We appreciate the Health and Welfare Data Science Center, Ministry of Health and Welfare (HWDC, MOHW), Taiwan, for providing the National Health Insurance Research Database (NHIRD).
Funding
This study was supported by the Tri-Service General Hospital Research Foundation (Grant Number: TSGH-D-110103).
Author information
Authors and Affiliations
Contributions
S-HW: Conceptualization, Supervision; J-TS: Writing—Original draft preparation; TLT: Writing—Reviewing and Editing; P-HS: Methodology; T-TY: Visualization, Investigation; C-CW: Project administration; R-YP: Validation; W-CC: Resources, Data curation; C-HC: Software, Formal analysis.
Corresponding author
Ethics declarations
Conflict of interest
Jen-Ta Shih, Timothy L Tan, Pei-Hung Shen, Tsu-Te Yeh, Chia-Chun Wu, Ru-Yu Pan, Wu-Chien Chien, Chi-Hsiang Chung, Sheng-Hao Wang have no relevant financial or non-financial interests to disclose.
Ethical Approval
The study protocol was approved by the Institutional Review Board of Tri-Service General Hospital, National Defense Medical Center (Approval No.: B-111-05). All procedures were performed in accordance with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Human and Animal Rights and Informed Consent
This database study involving human participants was in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The informed conset was waived because this study invovled no more than minimal riskand used de-idetification data. The IRB of National Defense Medical Center approved this study (approval No. B-111-05).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shih, JT., Tan, T.L., Shen, PH. et al. Postoperative Bisphosphonates Use is Associated with Reduced Adverse Outcomes After Primary Total Joint Arthroplasty of Hip and Knee: A Nationwide Population-Based Cohort Study. Calcif Tissue Int 114, 451–460 (2024). https://doi.org/10.1007/s00223-024-01192-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-024-01192-6