Abstract
Introduction
Infections associated with cardiac implantable electronic devices (CIEDs) are a multifactorial disease that leads to increased morbidity and mortality.
Objective
The aim was to analyze patient-, disease- and treatment-related characteristics including microbiological and bacterial spectrum according to survival status and to identify risk factors for 1- and 3-year mortality in patients with local and systemic CIED infection.
Methods
In a retrospective cohort study, we analyzed data from patients with CIED-related local or systemic infection undergoing successful transvenous lead extraction (TLE). Survival status as well as incidence and cause of rehospitalization were recorded. Microbiology and antibiotics used as first-line therapy were compared according to mortality. Independent risk factors for 1- and 3-year mortality were determined.
Results
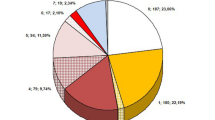
Data from 243 Patients were analyzed. In-hospital mortality was 2.5%. Mortality rates at 30 days, 1- and 3 years were 4.1%, 18.1% and 30%, respectively. Seventy-four (30.5%) patients had systemic bacterial infection. Independent risk factors for 1-year mortality included age (OR 1.05 [1.01–1.10], p = 0.014), NT-proBNP at admission (OR 4.18 [1.81–9.65], p = 0.001), new onset or worsened tricuspid regurgitation after TLE (OR 6.04 [1.58–23.02], p = 0.009), and systemic infection (OR 2.76 [1.08–7.03], p = 0.034), whereas systemic infection was no longer an independent risk factor for 3-year mortality. Staphylococcus aureus was found in 18.1% of patients who survived and in 25% of those who died, p = 0.092. There was a high proportion of methicillin-resistant strains among coagulase-negative staphylococci (16.5%) compared to Staphylococcus aureus (1.2%).
Conclusions
Staphylococci are the most common causative germs of CIED-infection with coagulase-negative staphylococci showing higher resistance rates to antibiotics. The independent risk factors for increased long-term mortality could contribute to individual risk stratification and well-founded treatment decisions in clinical routine. Especially the role of tricuspid regurgitation as a complication after TLE should be investigated in future studies.







Similar content being viewed by others
Data availability
The data underlying this article will be shared on reasonable request to the corresponding author.
References
Mond HG, Proclemer A (2011) The 11th world survey of cardiac pacing and implantable cardioverter-defibrillators: calendar year 2009—a World Society of Arrhythmia’s project. Pacing Clin Electrophysiol 34:1013–1027
Bongiorni MG, Burri H, Deharo JC, Starck C, Kennergren C, Saghy L, Rao A, Tascini C, Lever N, Kutarski A, Fernandez Lozano I, Strathmore N, Costa R, Epstein L, Love C, Blomstrom-Lundqvist C, ESC Scientific Document Group (2018) 2018 EHRA expert consensus statement on lead extraction: recommendations on definitions, endpoints, research trial design, and data collection requirements for clinical scientific studies and registries: endorsed by APHRS/HRS/LAHRS. Europace 20:1217
Miyagi Y, Sakamoto SI, Kawase Y, Oomori H, Watanabe Y, Kurita J, Maruyama Y, Sasaki T, Ishii Y (2021) Temporal and microbiological analysis of cardiac implantable electrical device infections—a retrospective study. Circ Rep 3:488–496
Le KY, Sohail MR, Friedman PA, Uslan DZ, Cha SS, Hayes DL, Wilson WR, Steckelberg JM, Baddour LM, Mayo Cardiovascular Infections Study Group (2011) Clinical predictors of cardiovascular implantable electronic device-related infective endocarditis. Pacing Clin Electrophysiol 34:450–459
Blomström-Lundqvist C, Traykov V, Erba PA, Burri H, Nielsen JC, Bongiorni MG, Poole J, Boriani G, Costa R, Deharo JC, Epstein LM, Saghy L, Snygg-Martin U, Starck C, Tascini C, Strathmore N, ESC Scientific Document Group (2020) European Heart Rhythm Association (EHRA) international consensus document on how to prevent, diagnose, and treat cardiac implantable electronic device infections-endorsed by the Heart Rhythm Society (HRS), the Asia Pacific Heart Rhythm Society (APHRS), the Latin American Heart Rhythm Society (LAHRS), International Society for Cardiovascular Infectious Diseases (ISCVID) and the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Europace 22:515–549
Olsen T, Jørgensen OD, Nielsen JC, Thøgersen AM, Philbert BT, Frausing MHJP, Sandgaard NCF, Johansen JB (2022) Risk factors for cardiac implantable electronic device infections: a nationwide Danish study. Eur Heart J 43:4946–4956
Krahn AD, Longtin Y, Philippon F, Birnie DH, Manlucu J, Angaran P, Rinne C, Coutu B, Low RA, Essebag V, Morillo C, Redfearn D, Toal S, Becker G, Degrâce M, Thibault B, Crystal E, Tung S, LeMaitre J, Sultan O, Bennett M, Bashir J, Ayala-Paredes F, Gervais P, Rioux L, Hemels MEW, Bouwels LHR, van Vlies B, Wang J, Exner DV, Dorian P, Parkash R, Alings M, Connolly SJ (2018) Prevention of arrhythmia device infection trial: the PADIT trial. J Am Coll Cardiol 72:3098–3109
Tarakji KG, Mittal S, Kennergren C, Corey R, Poole JE, Schloss E, Gallastegui J, Pickett RA, Evonich R, Philippon F, McComb JM, Roark SF, Sorrentino D, Sholevar D, Cronin E, Berman B, Riggio D, Biffi M, Khan H, Silver MT, Collier J, Eldadah Z, Wright DJ, Lande JD, Lexcen DR, Cheng A, Wilkoff BL, WRAP-IT Investigators (2019) Antibacterial envelope to prevent cardiac implantable device infection. N Engl J Med 380:1895–1905
Brunner MP, Yu C, Hussein AA, Tarakji KG, Wazni OM, Kattan MW, Wilkoff BL (2015) Nomogram for predicting 30-day all-cause mortality after transvenous pacemaker and defibrillator lead extraction. Heart Rhythm 12:2381–2386
Deshmukh A, Patel N, Noseworthy PA, Patel AA, Patel N, Arora S, Kapa S, Noheria A, Mulpuru S, Badheka A, Fischer A, Coffey JO, Cha YM, Friedman P, Asirvatham S, Viles-Gonzalez JF (2015) Trends in use and adverse outcomes associated with transvenous lead removal in the United States. Circulation 132:2363–2371
Mehta VS, Elliott MK, Sidhu BS, Gould J, Kemp T, Vergani V, Kadiwar S, Shetty AK, Blauth C, Gill J, Bosco P, Rinaldi CA (2021) Long-term survival following transvenous lead extraction: importance of indication and comorbidities. Heart Rhythm 18:1566–1576
Oszczygieł E, Kutarski A, Oszczygieł A, Mańkowska-Załuska B, Chudzik M, Wranicz JK, Cygankiewicz I (2017) Risk score to assess mortality risk in patients undergoing transvenous lead extraction. Pacing Clin Electrophysiol 40:918–923
Sood N, Martin DT, Lampert R, Curtis JP, Parzynski C, Clansy J (2018) Incidence and predictors of perioperative complications with transvenous lead extractions: real-world experience with national cardiovascular data registry. Circ Arrhythm Electrophysiol 11(2):e004768
Wazni O, Epstein LM, Carrillo RG, Love C, Adler SW, Riggio DW, Karim SS, Bashir J, Greenspon AJ, DiMarco JP, Cooper JM, Onufer JR, Ellenbogen KA, Kutalek SP, Dentry-Mabry S, Ervin CM, Wilkoff BL (2010) Lead extraction in the contemporary setting: the LExICon study: an observational retrospective study of consecutive laser lead extractions. J Am Coll Cardiol 55:579–586
El-Chami MF, Liu Y, Griffiths RI, Knight BP, Weiss R, Mark GE, Biffi M, Probst V, Lambiase PD, Miller MA, Jacobsen CM, Baddour LM (2022) Device-related infection associated with increased mortality risk in de novo transvenous implantable cardioverter-defibrillator medicare patients. J Cardiovasc Electrophysiol 33:725–730
Rizwan Sohail M, Henrikson CA, Jo Braid-Forbes M, Forbes KF, Lerner DJ (2015) Increased long-term mortality in patients with cardiovascular implantable electronic device infections. Pacing Clin Electrophysiol 38:231–239
Chung DU, Burger H, Kaiser L, Osswald B, Bärsch V, Nägele H, Knaut M, Reichenspurner H, Gessler N, Willems S, Butter C, Pecha S, Hakmi S, GALLERY investigators (2023) Transvenous lead extraction in patients with systemic cardiac device-related infection-Procedural outcome and risk prediction: a GALLERY subgroup-analysis. Heart Rhythm 20(2):181–189
Sandoe JA, Barlow G, Chambers JB, Gammage M, Guleri A, Howard P, Olson E, Perry JD, Prendergast BD, Spry MJ, Steeds RP, Tayebjee MH, Watkin R, British Society for Antimicrobial Chemotherapy, British Heart Rhythm Society, British Cardiovascular Society, British Heart Valve Society, British Society for Echocardiography (2015) Guidelines for the diagnosis, prevention and management of implantable cardiac electronic device infection. Report of a joint Working Party project on behalf of the British Society for Antimicrobial Chemotherapy (BSAC, host organization), British Heart Rhythm Society (BHRS), British Cardiovascular Society (BCS), British Heart Valve Society (BHVS) and British Society for Echocardiography (BSE). J Antimicrob Chemother 70:325–359
Polyzos KA, Konstantelias AA, Falagas ME (2015) Risk factors for cardiac implantable electronic device infection: a systematic review and meta-analysis. Europace 17:767–777
Palmisano P, Accogli M, Zaccaria M, Luzzi G, Nacci F, Anaclerio M, Favale S (2013) Rate, causes, and impact on patient outcome of implantable device complications requiring surgical revision: large population survey from two centres in Italy. Europace 15:531–540
Prutkin JM, Reynolds MR, Bao H, Curtis JP, Al-Khatib SM, Aggarwal S, Uslan DZ (2014) Rates of and factors associated with infection in 200 909 Medicare implantable cardioverter-defibrillator implants: results from the National Cardiovascular Data Registry. Circulation 130:1037–1043
Rao A, Garner D, Starck C, Kirkfeldt RE, Dagres N, Didier K, Montano N, Heidbuchel H (2020) Knowledge gaps, lack of confidence, and system barriers to guideline implementation among European physicians managing patients with CIED lead or infection complications: a European Heart Rhythm Association/European Society of Cardiology educational needs assessment survey. Europace 22:1743–1753
Bongiorni MG, Kennergren C, Butter C, Deharo JC, Kutarski A, Rinaldi CA, Romano SL, Maggioni AP, Andarala M, Auricchio A, Kuck KH, Blomström-Lundqvist C, ELECTRa Investigators, (2017) The European Lead Extraction ConTRolled (ELECTRa) study: a European Heart Rhythm Association (EHRA) Registry of transvenous lead extraction outcomes. Eur Heart J 38:2995–3005
Baman TS, Gupta SK, Valle JA, Yamada E (2009) Risk factors for mortality in patients with cardiac device-related infection. Circ Arrhythm Electrophysiol 2:129–134
Athan E, Chu VH, Tattevin P, Selton-Suty C, Jones P, Naber C, Miró JM, Ninot S, Fernández-Hidalgo N, Durante-Mangoni E, Spelman D, Hoen B, Lejko-Zupanc T, Cecchi E, Thuny F, Hannan MM, Pappas P, Henry M, Fowler VG Jr, Crowley AL, Wang A, ICE-PCS Investigators (2012) Clinical characteristics and outcome of infective endocarditis involving implantable cardiac devices. JAMA 307:1727–1735
Deharo JC, Quatre A, Mancini J, Khairy P, Le Dolley Y, Casalta JP, Peyrouse E, Prévôt S, Thuny F, Collart F, Raoult D, Habib G, Franceschi F (2012) Long-term outcomes following infection of cardiac implantable electronic devices: a prospective matched cohort study. Heart 98:724–731
Habib A, Le KY, Baddour LM, Friedman PA, Hayes DL, Lohse CM, Wilson WR, Steckelberg JM, Sohail MR, Mayo Cardiovascular Infections Study Group (2013) Predictors of mortality in patients with cardiovascular implantable electronic device infections. Am J Cardiol 111:874–879
Diemberger I, Biffi M, Lorenzetti S, Martignani C, Raffaelli E, Ziacchi M, Rapezzi C, Pacini D, Boriani G (2018) Predictors of long-term survival free from relapses after extraction of infected CIED. Europace 20:1018–1027
Klug D, Lacroix D, Savoye C, Goullard L, Grandmougin D, Hennequin JL, Kacet S, Lekieffre J (1997) Systemic infection related to endocarditis on pacemaker leads: clinical presentation and management. Circulation 95:2098–2107
Le KY, Sohail MR, Friedman PA, Uslan DZ, Cha SS, Hayes DL, Wilson WR, Steckelberg JM, Baddour LM, Mayo Cardiovascular Infections Study Group (2011) Impact of timing of device removal on mortality in patients with cardiovascular implantable electronic device infections. Heart Rhythm 8:1678–1685
von Eiff C, Peters G, Heilmann C (2002) Pathogenesis of infections due to coagulasenegative staphylococci. Lancet Infect Dis 2:677–685
Becker K, Heilmann C, Peters G (2014) Coagulase-negative staphylococci. Clin Microbiol Rev 27:870–926
Polewczyk A, Jacheć W, Polewczyk AM, Tomasik A, Janion M, Kutarski A (2017) Infectious complications in patients with cardiac implantable electronic devices: risk factors, prevention, and prognosis. Pol Arch Intern Med 127:597–607
Santaguida PL, Don-Wauchope AC, Oremus M, McKelvie R, Ali U, Hill SA, Balion C, Booth RA, Brown JA, Bustamam A, Sohel N, Raina P (2014) BNP and NT-proBNP as prognostic markers in persons with acute decompensated heart failure: a systematic review. Heart Fail Rev 19:453–470
Li H, Zeng Z, Cheng J, Hu G, Li Y, Wei L, Zhou Y, Ran P (2020) Prognostic role of NT-proBNP for in-hospital and 1-year mortality in patients with acute exacerbations of COPD. Int J Chron Obstruct Pulmon Dis 15:57–67
Birgersdotter-Green U, Dawood FZ (2018) Transvenous extraction of pacemaker and defibrillator leads and the risk of tricuspid valve regurgitation: making a case for thoughtful lead management. JACC Clin Electrophysiol 4:1429–1430
Funding
Funded by the Brandenburg Medical School (MHB) publication fund supported by DFG.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors have no conflicts of interest to declare.
Ethical approval
The study was performed in compliance with the principals outlined in the Declaration of Helsinki and approved by the ethics committee of the Brandenburg Medical School (MHB) (E-01-20200809).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
Laboratory results according to 1-year mortality
Survived n = 199 | Died n = 44 | p-Value | |
|---|---|---|---|
At admission | |||
Haemoglobin [mmol/L] | 7.9 (7.2–8.8) | 7.4 (6.2–8.3) | 0.009 |
Thrombocytes [103/µL] | 221 (169–273) | 204 (142–263) | 0.209 |
Leukocytes [Gpt/L] | 8.1 (6.7–10.0) | 8.2 (6.8–11.9) | 0.301 |
CRP [mg/L] | 9.0 (3.0–33.9) | 37.2 (10.1–99.8) | < 0.001 |
NT-proBNP [pg/mL] | 1109 (356–3425) | 3803 (1254–9063) | < 0.001 |
Creatinine [µmol/L] | 96 (79.0–131.0) | 112.5 (73.0–178.0) | 0.136 |
eGFR [mL/min/1.73] | 66 (48–89) | 54 (32–84) | 0.040 |
At discharge | |||
Haemoglobin [mmol/L] | 6.6 (6.0–7.4) | 6.4 (5.6–7.1) | 0.063 |
Thrombocytes [103/µL] | 215 (166–281) | 209 (142–306) | 0.734 |
Leukocytes [Gpt/L] | 7.2 (6.0–9.1) | 8.7 (7.1–11.4) | 0.003 |
CRP [mg/L] | 26.4 (13.3–59.0) | 49.7 (18.5–76.1) | 0.018 |
Creatinine [µmol/L] | 92 (75–134) | 130 (88–190) | 0.004 |
eGFR [mL/min/1.73] | 69 (46–86) | 46 (28–77) | 0.003 |
Difference in laboratory results from admission to discharge | |||
△Haemoglobin [mmol/L] | 1.2 (0.6–1.8) | 1 (0.45–1.55) | 0.160 |
△ Thrombocytes [103/µL] | −7 (−46–43) | 15 (−37–63) | 0.421 |
△Leukocytes [Gpt/L] | 0.7 (−0.5–2.1) | 0 (−1.7–2.0) | 0.136 |
△CRP [mg/L] | −11.2 (−29.8–5.8) | 1.3 (−17.5–26.8) | 0.013 |
△eGFR [mL/min/1.73] | 0.0 (−9.0–6.0) | 1 (−8–14) | 0.360 |
△Creatinine [µmol/L] | 4.0 (−9.0–17) | −1.0 (−38.5–17.5) | 0.180 |
Antibiotics used as first-line therapy according to 1-year mortality
Survived n = 199 | Died n = 44 | p-Value | |
|---|---|---|---|
Flucloxacillin | 39 (19.6%) | 9 (20.5%) | 0.897 |
Amoxicillin | 2 (1.0%) | 1 (2.3%) | 0.491 |
Ampicillin/sulbactam | 16 (8.0%) | 3 (6.8%) | 0.785 |
Piperacillin/tazobactam | 11 (5.5%) | 4 (9.1%) | 0.374 |
Cefuroxime | 50 (25.1%) | 7 (15.9%) | 0.192 |
Ceftriaxone | 2 (1.0%) | 3 (6.8%) | 0.014 |
Meropenem | 6 (3.0%) | 4 (9.1%) | 0.066 |
Ciprofloxacin | 7 (3.5%) | 1 (2.3%) | 0.675 |
Vancomycin | 36 (18.1%) | 12 (27.3%) | 0.166 |
Gentamicin | 13 (6.5%) | 4 (9.1%) | 0.547 |
Clindamycin | 27 (13.6%) | 3 (6.8%) | 0.218 |
Linezolid | 4 (2.0%) | 1 (2.3%) | 0.912 |
Daptomycin | 5 (2.5%) | 3 (6.8%) | 0.147 |
Rifampicin | 16 (8.0%) | 5 (11.4%) | 0.478 |
Cotrimoxazole | 3 (1.5%) | 0 (0%) | 0.412 |
Mortality table
Interval [days] | Number of patients entering Interval | Number of deaths | Cumulative proportion surviving at end of interval |
|---|---|---|---|
0 | 243 | 16 | 0.93 |
50 | 227 | 8 | 0.90 |
100 | 218 | 3 | 0.89 |
150 | 215 | 4 | 0.87 |
200 | 211 | 3 | 0.86 |
250 | 208 | 5 | 0.84 |
300 | 203 | 1 | 0.84 |
350 | 202 | 4 | 0.82 |
400 | 198 | 6 | 0.79 |
450 | 191 | 4 | 0.78 |
500 | 187 | 0 | 0.78 |
550 | 187 | 2 | 0.77 |
600 | 185 | 1 | 0.76 |
650 | 184 | 0 | 0.76 |
700 | 184 | 2 | 0.76 |
750 | 182 | 1 | 0.75 |
800 | 181 | 0 | 0.75 |
850 | 181 | 1 | 0.75 |
900 | 180 | 1 | 0.74 |
950 | 179 | 4 | 0.73 |
1000 | 175 | 2 | 0.72 |
1050 | 172 | 5 | 0.70 |
1100 | 164 | 0 | 0.70 |
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Schipmann, L.C., Moeller, V., Krimnitz, J. et al. Outcome and microbiological findings of patients with cardiac implantable electronic device infection. Heart Vessels (2024). https://doi.org/10.1007/s00380-024-02380-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00380-024-02380-y